![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
42 Cards in this Set
- Front
- Back
|
Every substance is either an element or a(n)
|
compound
|
|
|
Most molecular compounds are composed of two or more
|
non-metals
|
|
|
Molecules Consisting of two atoms are _____ molecules
|
diatomic
|
|
|
The chemical formula of a molecular compound is a
|
molecular formula
|
|
|
Molecular compounds tend to have ____ melting and boiling points.
|
low
|
|
|
Ionic compounds tend to have _____ melting and boiling points
|
high
|
|
|
A molecular formula shows how many ____ of each element contains
|
atoms
|
|
|
A molecular formula does not indicate the _____ of the molecule
|
structure
|
|
|
A compound is either ____ or ionic in nature
|
covalent
|
|
|
What is an electrically neutral group of atoms joined together by covalent bonds
|
molecule
|
|
|
What is composed of molecules?
|
molecular compound
|
|
|
What joins atoms held together by sharing electrons?
|
covalent bond
|
|
|
What is a molecule consisting of two atoms?
|
diatomic molecule
|
|
|
What shows the kinds and numbers present in a molecule of a compound?
|
molecular formula
|
|
|
Is CH4 an atom or molecule?
|
molecule
|
|
|
Is Ne an atom or molecule?
|
atom
|
|
|
Is O2 and atom or molecule?
|
molecule
|
|
|
When atoms share electrons to gain the electron configuration of a noble gase, the bonds are ____
|
covalent
|
|
|
A ____ pair of valence electrons constitutes a single covalent bond.
|
shared
|
|
|
Pairs of valence electrons that are not shared between atoms are called
|
unshared pairs
|
|
|
When it is possible to write two or more valid electron dot formulas for a molecule or ion, each formula is referred to as a ____
|
resinate structure
|
|
|
The chemical formulas of molecular compounds show
|
the number and types of atoms in each molecule
|
|
|
A molecule of bromine has ___ unshared pairs of electrons
|
6
|
|
|
What is a chemical formula that shows the arrangement of atoms in a molecule or a polyatomic ion?
|
Structural Formula
|
|
|
Is H20 polar or nonpolar?
|
polar
|
|
|
Is NH3 polar or nonpolar?
|
polar
|
|
|
Is CO2 polar or nonpolar?
|
nonpolar
|
|
|
Is HCl polar or nonpolar?
|
polar
|
|
|
what determines the physical properties of a compound?
|
types of bonds
|
|
|
What causes dispersion?
|
motion of electrons
|
|

|
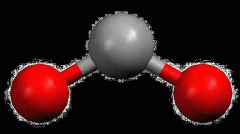
Linear
|
|
|
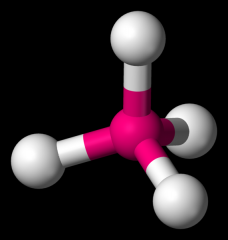
Trigonal Pyramidal
|
|
|
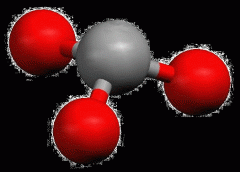
Bent
|
|
|
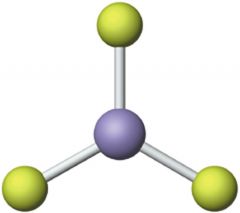
Trigonal Plannar
|
|
|
Tetrahedral
|
|
|
How many covalent bonds are in a nitrogen molecule?
|
3
|
|
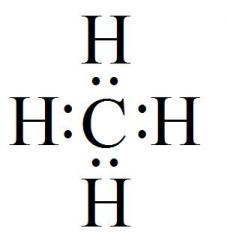
Which Molecule Is This?
|
CH4
|
|
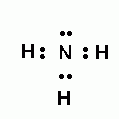
Which Molecule Is This?
|
NH3
|
|
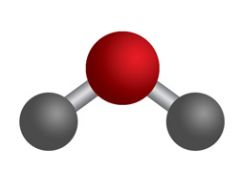
What Type Of Model Is This
|
Ball and Stick Model
|
|

What Type Of Model Is This?
|
Space Filling Molecular Model
|
|
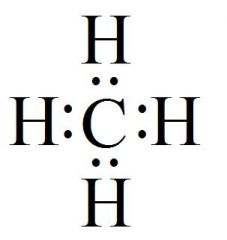
What Kind Of Formula Is This?
|
Structural Formula
|
|
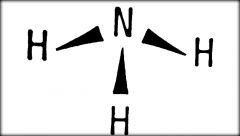
What Type Of Model Is This?
|
Perspective Drawing
|

