![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
24 Cards in this Set
- Front
- Back
|
A compound hat has a carbonyl group bonded to at least one hydrogen RCHO
|
Aldehyde
|
|
|
A compound that has a carbonyl group bonded to two carbons in organic groups that can be the same or different R2C=O, RCOR
|
Ketone
|
|
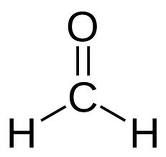
What is the name of this compound
|
Formaldehyde
|
|
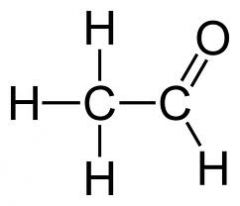
What is the name of this compound?
|
Acetaldehyde
|
|
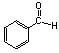
What is the name of this compound?
|
Benzaldehyde
|
|
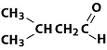
What is the name of this compound?
|
3-methybutanal
|
|
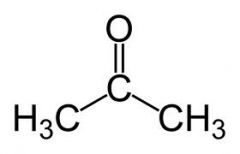
What is the name of this compound?
common systematic |
Acetone
2-propanone |
|
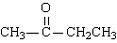
What is the name of this compound?
common systematic |
methyl ethyl ketone
2-Butanone |
|
|
What is the name of this compound?
|
2-Pentanone
|
|
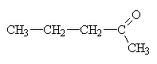
What is the name of this compound?
|
2-pentanone
|
|
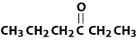
What is the name of this compound?
common systematic |
ethyl propyl ketone
3-hexanone |
|
|
What type of rxns do Aldehydes and ketones undergo?
|
Addition reactions
|
|
|
What are the reactants to form a Hemiacetal?
|
An aldehyde or ketone and an alcohol
|
|
|
What does it take to form an acetal?
|
An aldehyde or ketone and an alcohol AND an acid catalyst
|
|
|
What is the other product formed by Acetal Formation
|
H2O
|
|
|
A compound that has two ether-like -OR groups bonded to the same carbon
|
An acetal
|
|
|
A compound that with both an alcohol-like -OH and an ether-like -OR group bonded to the same carbon atom
|
Hemicacetal
|
|
|
What type of reaction is Acetal formation?
|
substitution
|
|
|
What is the product of a mild oxidation of an Aldehyde?
|
Carboxylic acid
|
|
|
What is the product of oxidation of a ketone?
|
No reaction
|
|
|
What would you use to determine between an Aldehyde and a ketone? Why?
|
Tollen's Reagent
It react with Aldehyde producing the silver mirror |
|
|
What is the product of Reduction of an Aldehyde?
|
Primary alcohol
|
|
|
What is the product of Reduction of a Ketone?
|
Secondary alchohol
|
|
|
What is the reducing agent used in aldehyde and ketone reduction?
|
H3O+
|

