![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
58 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
When a protein molecule in eggs and meat change shape it is called? |
Denaturing |
|
|
|
Why does the texture of egg/meat change when cooked? |
The shape of the protein molecule is PERMANENTLY CHANGED. It is irreversible |
|
|
|
Describe what an emulsifier looks like |
Hydrophilic part that attracts water Hydrophobic part that attracts oil. |
|
|
|
Why do emulsifiers work? |
Because they hydrophobic end binds with the water molecules and the hydrophobic end bonds with the oil. This means that they cannot separate. |
|
|
|
What is the balanced symbol equation for the decomposition of sodium hydrogencarbonate |
2NaHCO3 (goes to) Na2CO3 +H20 + CO2 |
|
|
|
Basic equation for making an ester? |
Alcohol + acid (goes to) ester + water |
|
|
|
Experiment to make am ester? |
Xjsjsje |
|
|
|
Why do perfumes need the following properties?: Easily evaporate? |
So that the perfume particles can easily reach the nose |
|
|
|
Non-toxic? |
So it doesn't poison you! #essential |
|
|
|
Not reactant with water |
Otherwise it would react with sweat |
|
|
|
Non irritant |
Otherwise you would not be able to apply it to the skin |
|
|
|
Insoluble in water |
Otherwise it would wash off easily |
|
|

Must be volatile |
This means they evaporate easily. Molecules of perfume are held together by weak forces of attraction. The molecules that have lots of energy can easily overcome the weak forces and escape |
|
|
|
What can esters be used as? |
Solvents |
|
|
|
What is a solution? |
A mixture of solvent and solute that does not separate out. |
|
|
|
Why does water not dissolve nail varnish? |
+ attraction between water molecules = stronger than the attraction between water molecules and particles in the nail varnish + attraction between particles in the nail varnish = stronger than the attraction between water molecules and particles in nail varnish |
|
|
|
Advantage of animal testing? |
Prevents humans from being harmed |
|
|
|
Disadvantages of animal testing |
Cruel to animals They do not have the same body chemistry as humans so the results may not even be applicable |
|
|
|
Animals testing has been |
BANNED IN THE EU DUE TO ANIMAL WELFARE |
|
|
|
Paint is a colloid, can you describe this? |
A colloid is where the particles are mixed well (dispersed but not dissolved) with liquid particles |
|
|
|
Paint is a mixture of: |
Pigment giving the paint it's colour Binding medium, sticks the pigment onto the surface it's being painted onto Solvent thins the binding medium making it easier to cost the surface |
3 components |
|
|
How do most paints dry? |
1. Paint is applied as a thin layer 2. The solvent evaporates |
|
|
|
How do oil based paints dry? |
Oil based paint such as gloss paint dries and two stages step 1 the solvent evaporates step 2 the Binding medium reacts with oxygen in the air as it dries to form a hard layer |
|
|
|
Thermochromic paints suited to a given use? |
They change colour when heated or cooled. Cutlery for babies as a warning if the food is too hot Kettles and cups to indicate temperature |
|
|
|
What can be added to thermochromic pigments? |
Acrylic paints can be added so that it experiences even more colour change. |
|
|
|
Why do phosphorescent pigments glow in the dark? |
1. They ABSORB and STORE energy 2. Slowly release it as light energy over a period of time when it is dark. |
|
|
|
What is the alternative to phosphorescent pigments? Why is that worse? |
The alternative is radioactive substances. They are dangerous and expose people to too much radiation. The first watched were made of this. Phosphorescent pigments are not radioactive so are much safer |
|
|
|
Factors that need to be considered when choosing a fuel? Choose 5. |
Energy value - how much energy is released for a gram of that fuel? Availability - how easy is it to get the fuel? Ease of storage - how easy is it to store the fuel? Cost - how much fuel do you get for your money Toxicity - is the fuel, or products from its combustion toxic? Pollution - do the combustion products cause pollution? Ease of use - is it easy to control, is special equipment needed? |
PEETAC |
|
|
Why is the amount of fossil fuels being burnt increasing? |
Increasing world population Increase of use in developing countries such as China and India |
|
|
|
Complete combustion, symbol equation? Products? |

Methane + oxygen ---> carbon dioxide + water CH4 + 2O2 ---> CO2 + 2H2O |
|
|
|
Incomplete combustion. Products, symbol equation? |

Methane + oxygen ---> carbon monoxide + water 2CH4 + 3O2 ---> 2CO + 4H20 |
|
|
|
Why is complete combustion better than incomplete combustion? |
Becuase it releases more heat energy Incomplete combustion releases carbon monoxide which is poisonous. That is why gas appliances have to be regularly checked. |
|
|
|
Why does incomplete combustion occur? |
It is when a fuel is burnt without enough oxygen |
|
|
|
The composition of the air in percentages: |
78% nitrogen 21% oxygen 0.035% is carbon dioxide |
|
|
|
Carbon cycle? |
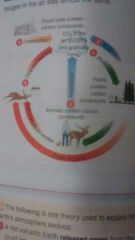
Plants photosynthesise ( and respire) Animals eat the plants Animal (and plants) dies leading to decay and respiration Combustion also adds to the carbon dioxide levels in the atmosphere |
|
|
|
Atmosphere evolution? |
Ammonia and carbon dioxide in the atmosphere as they were released from volcanoes Photosynthesising plants increase the amount of oxygen in the atmosphere |
|
|
|
A theory to explain HOW the atmosphere evolved? |
1. Degassing of early volcanoes producing an atmosphere rich in carbon dioxide, ammonia and eater vapour
2. The water vapour condensed to form the oceans.
3. These newly formed bodies of water dissolved some of the carbon dioxide in the atmosphere
4. Increase in nitrogen levels due to its unreactivity and nitrifying bacteria releasing it. 5. Development of primitive plants that could photosynthesise allowing the oxygen levels to increase and carbon dioxide levels to decrease even more. |
|
|
|
Evaluate human influences on the composition of the air |
Burning fossil fuels - carbon dioxide Deforestation - photosynthesis is reduced so carbon dioxide levels increase Increase in population - more demand for space and energy |
|
|
|
Effects of sulfur dioxide? |
Acid rain: Plant and aquatic life death Stone work and iron work corroded |
|
|
|
Effects of carbon monoxide? |
A poisonous gas that comes from incomplete combustion |
|
|
|
Effects of oxides of nitrogen? |
Causes photochemical smog and acid rain |
|
|
|
Hot car engine allows? |
Nitrogen and oxygen to react to make nitrogen monoxide and nitrogen dioxide |
|
|
|
Why is it important that atmospheric pollution is controlled? |
Because it can affect people's health and damage the surroundings |
|
|
|
what does a catalytic converter do and how does it work? |
Catalytic converters turn polluting chemicals in the exhaust like carbon monoxide carbon monoxide to react with oxygen to create less harmful gases like carbon dioxide instead. This reduces the amount of pollutants in the atmosphere. Carbon monoxide + nitrogen oxide ---> nitrogen + carbon dioxide 2CO + 2NO ---> N2 + 2CO2 |
|
|
|
Why are fossil fuels finite and non-renewable? |
Becuase they are being used up faster than new ones can be formed. It will run out and be used up in the future |
|
|
|
What is crude oil? |
A mixture of hydrocarbons |
|
|
|
Problems associated with using crude oil? |
Finding replacements It will run out Conflict between making petrochemicals and fuels |
|
|
|
What is a hydrocarbon? |
A molecule made of hydrogen and carbon ONLY |
|
|
|
Describe fractional distillation |

Fractional distillation is the separation of crude oil into different fractions 1. Crude oil is heated 2. A fractioning column is used that has a temperature gradient 3. Fractions with low boiling points exit from the TOP 4. Fractions with high boiling points exit from the BOTTOM of the column 5. Hydrocarbons with similar boiling points exit from the same fraction |
|
|
|
Explain why crude oil can be separated by fractional distillation in terms of intermolecular forces |
There are a mixture of hydrocarbons, some long, some short Long hydrocarbon = strong intermolecular forces = more energy needed to overcome the forces = higher temperature needed Short hydrocarbon = weaker intermolecular forces = less energy needed to break the bonds = lower temperature needed to break the bonds Intermolecular forces, ones between different hydrocarbon molecules are broken. Covalent bonds, the bonds within one hydrocarbon, are not broken. |
|
|
|
Problems with the transportation of crude oil? |
Accidents can cause oil spills: Damage to birds feathers causing death Damage to sea life Detergents used to clear up oil slicks have chemicals that harm wildlife. |
|
|
|
Political problems with the exploitation of crude oil? |
A lot of the UK supply of crude oil comes from politically unstable countries There are future supply issues |
|
|
|
What can hydrocarbon molecules be described as? |
Alkanes and alkenes |
|
|
|
What is cracking? |
Cracking is the process used to convert large alkene molecules into into smaller alkane and alkene molecules. It makes useful alkene molecules that can be used to make polymers. There isn't petrol in crude oil to meet demands. So parts that cannot be used can be made into petrol. |
|
|
|
Describe the properties of an alkene? |

- they are unsaturated. - have single COVALENT bonds. -they have a maximum amount of hydrogen atoms per carbon atom in the molecule. |
U S A |
|
|
Describe the properties of alkenes? |
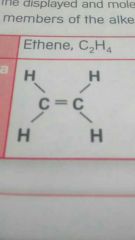
- they are unsaturated - double covalent bonds with the carbon atoms - the carbon atom isn't bonded to the maximum number of hydrogen atoms. -double bonds involve two pairs of shared electrons. |
|
|
|
How to distinguish between alkenes and alkanes? |
Bromine water test! Alkenes - turn bromine water colourless. It is an addition reaction between bromine water and c=c to make a colourless dibromo compound Alkanes - don't change bromine water, it remains orange |
|
|
|
How to distinguish between alkenes and alkanes? |
Bromine water test! Alkenes - turn bromine water colourless. It is an addition reaction between bromine water and c=c to make a colourless dibromo compound Alkanes - don't change bromine water, it remains orange |
|

