![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
45 Cards in this Set
- Front
- Back
|
A change in matter that does NOT involve changing the identity of the a substance is a ____ change. |
physical |
|
|
A change matter that changes the identity of a substance is a ____ change. |
chemical |
|
|
Substances that interact during a chemical reaction are called ____ |
reactants |
|
|
Substances that are produced in a chemical reaction are called ____ |
products |
|
|
_______ yield, produce, or equal _____ |
reactants, products |
|
|
A _____ can speed up a chemical reaction. |
catalyst |
|
|
The study of matter and how it changes is called.... |
chemistry |
|
|
The smallest particle of an element that still retains the property of that element is called an.... |
atom |
|
|
An atom contains ____, ____, and ____ in it. We call them subatomic particles. |
protons, neutrons, & electrons |
|
|
An ____ is a pure substance that only contains one type of atom. |
element |
|
|
Anything that takes up space and has mass is called ____. |
matter |
|
|
Elements combined together to make new substances are called.... |
compounds |
|
|
A single unit of an element is called an ____ while a single unit of a compound is called a ____. |
atom, molecule |
|
|
Compounds have more than ____ element in them. |
one |
|
|
A column on the periodic table is called a ___ or family. |
group |
|
|
There are ____ columns on the periodic table, and thus ____ groups. |
18 18 |
|
|
Elements in the same group on the periodic table have the same.... |
number of electrons in their valence level |
|
|
The outermost level of electrons in an atom is called the ____ electrons |
valence |
|
|
The number of valence electrons in an atom is the same as the ____ number on the periodic table |
group |
|
|
The periodic table has ____ rows which are called _____. |
7 periods |
|
|
In addition to the same number of valence electrons, members of a group have the same.... |
characteristics |
|
|
The charge on an electron is ____. The charge on a proton is ____. The charge on a neutron is ____ |
-1 +1 0 |
|
|
The ___ & ___ are located in the center of the atom, which is called a ____. |
protons & neutrons nucleus |
|
|
Iron has an atomic number of 26 and an atomic mass of 56. An atom of iron, Fe, has ____ protons, ____ electrons, and ____ neutrons in it. |
26, 26, 30 |
|
|
Copper, Cu, has an atomic number of 29 and an atomic mass of 64. An atom of copper has ___ protons, ____ electrons, and ____ neutrons |
29, 29, 35 |
|
|
The elements Mg, Ca, & Sr are in the same ____. |
group |
|
|
The elements Na, Mg, & Al are in the same ____. |
period |
|
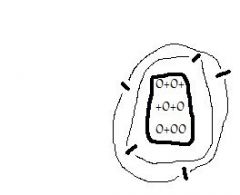
This atom has ___ protons, ___ electrons, & ___neutrons |
5, 5, 7 |
|
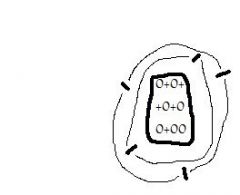
The atomic number of this element is ____. Its atomic mass is ___. |
5, 12 |
|
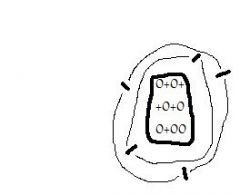
Since this atom, like all atoms, has an equal number of protons & electrons, we say it is electrically ____. |
neutral |
|
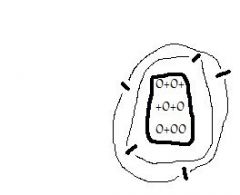
What element is this? |
Boron (B) |
|
|
Ice melting is an example of a ____ change. |
physical |
|
|
Paper burning is an example of a ___ change. |
chemical |
|
|
Water turning into water vapor is an example of a ___ change. |
physical |
|
|
An iron nail getting rust on the outside of it is an example of a ____ change. |
chemical |
|
|
Neon light turning orange when electrified is a ____ change. |
physical |
|
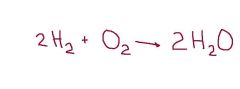
In this reaction of making water, the elements on the left side of the arrow are called ___. |
reactants |
|
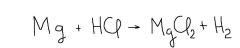
In this reaction of Magnesium with hydrochloric acid, the substances on the right of the arrow are called ____. |
products |
|
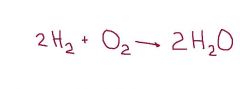
The arrow in this balanced equation is called a ____ sign and it means "equals" or "produces". |
yields |
|

This element has ___ protons, ___ electrons, and ____ neutrons |
79, 79, 118 |
|

The mass number of this element is ____. The number of protons & neutrons together in the nucleus is ____. |
39 39 |
|
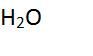
Is this an element? Is it a compound? One unit of it is called a ____. |
No Yes molecule |
|
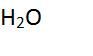
This compound has ___ atoms in it. |
3 |
|
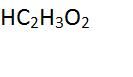
This multi-atom symbol is an example of a ____ . |
compound |
|
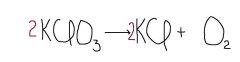
The "2" in front of the K symbols are numbers that help ____ the equation. What number should be in front of Oxygen on the product side? |
balance 3 |

