![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
67 Cards in this Set
- Front
- Back
|
Rate of reaction measures what? |
Rate of reaction measure how much product is formed in a fixed period of time |
|
|
How do you work out the rate of reaction be measured on graphs? (Show the equation too) |
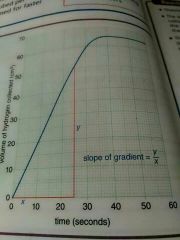
* choose part of the graph where there's a straight line (not curved line) * Measure the value of y and x Gradient = y --- X |
|
|
Whats limiting reactant? |
Limiting reactant is the reactant not in excess that gets used up by the end of the reaction |
|
|
If amount of limiting reactant doubles, what doubles as well? |
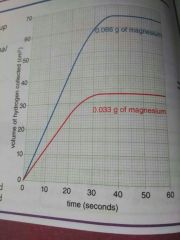
Amount of product formed doubles |
|
|
When does reactions occur ? |
When particles collide together |
|
|
Remember |
Reactions occur when particles collide together. Therefore, if the number of reacting particles of one reactant is limited, The number of collisions by particles of that reactant is limited |
|
|
Chemical reactions take place when particles do what? |
Collide to each other |
|
|
The rate of reaction depends on what? |
Number of collisions between the reacting particles. |
|
|
The higher the number of collisions the faster is what? |
Reaction |
|
|
Apart from the fact of particles collision, by what three things can rate of reaction be increased by? |
Increase in... Temperature -particles move faster as they gain kinetic energy Concentration - particles are more crowded so more successful collisions Pressure - forces particles to be closer together aka more frequent collisions |
|
|
The rate of reaction depends on what? |
Collision frequency |
|
|
Tip |
The rate of reaction depends on the collision frequency: this describes the number of successful collisions between reacting particles each second |
|
|
Exam tip |
When asked to explain the reaction rate changes , always use the idea of the rate of successful collisions of particles |
|
|
What powders can cause explosions ? |
Combustible powders can cause explosions |
|
|
The owners of a factory using combustible powders such as flour, custard powder or sulfur must be very careful. Why? |
Because if they have contact with open atmosphere, they may react with oxygen causing explosion as well as they need to ensure that the chances of spark being produced near the powders is very small |
|
|
A powdered reactant has a much larger surface area than the same mass of reactant in block form. Why? |
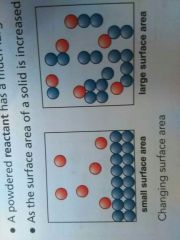
In a block, collisions can only occur with the particles on the surface. Most of the particles in the block are trapped on the inside so they are not available to react. Powdered reactant has more particles to react. |
|
|
How to make a block or something have bigger surface area? |
By breaking it up into pieces |
|
|
Reactions occur when reactant particles collide with what? |
Sufficient energy |
|
|
A catalyst changes the rate of reaction but it doesn't change what at the end? |

Doesn't change the end in terms of how much products were formed. |
|
|
Percentage yield is calculated by what formula? |
% yield = actual yield ---------------------- X100 Predicted yield |
|
|
What is catalyst? |
Chemical that speeds up the reaction |
|
|
Industrial processes need to have as high a percentage yield as possible so that they.... |
- reduce the amount of reactants waste, which is wasteful and costly - reduce their costs by ensuring that enough reactants are used, as too little reduces the amount of product - |
|
|
Atom economy can be found using what formula? |

Atom economy = Mr of desired products ---------------------------------- Sum and of Mr of all products
X100 |
|
|
Bond breaking is what process? |
Endothermic process |
|
|
Endothermic process is what? |
Bond breaking |
|
|
Bond making is what process? |
Exothermic process |
|
|
Exothermic process does what? |
Bond making |
|
|
Remember! |
All chemical reactions involve bond making (Exothermic ) and bond breaking (Endothermic) |
|
|
Diagrams can be used to represent bond energy changed. |

|
|
|
In chemical reaction, if more energy is released than needed then what process/reaction is that? |
Exothermic |
|
|
If a chemical reaction has more energy needed than released, what process/reaction is this? |
Endothermic |
|
|
The energy transferred by a fuel is calculated using what formula? |
Energy transfer (in J) = MxCx /\T M = mass in water heated (g) C = specific heat capacity of water (J/g °C) /\T = temperature change (°C)
|
|
|
Chemicals needed in large quantities, such as ammonia and sulfuric acid, are made using by batch or continuous process? |
Continuous process |
|
|
Give 2 facts of continuous process ? |
- makes large amount of the product 24 hours a day, seven days a week - takes less energy to maintain - as long as the process can be kept running than to stop and start the process. |
|
|
Chemicals needed in small quantities, such as medicines, are made using continuous or batch process? |
Batch process |
|
|
Batch processes make 2 things easier. What are they? |
Makes it easy to change a new batch when needed Makes it easy to change the production to a different product |
|
|
Batch process makes a ____ amount. |
Batch process makes a fixed amount |
|
|
Batch process allows what 2 things? |
Allows batches to be made and store until needed Allows quantities to be made it hat can be sold within a given time as many drugs have a "sell by" date |
|
|
Advantages of continuous process? |
- large quantity produced in one time - the cost per tonne is very small |
|
|
Disadvantages of continuous process? |
- the process is inefficient if not in constant use - there is a very high initial building and setup cost for these chemical plants |
|
|
Advantages of batch process? |
Batches can be made and then stored until needed Flexibility Easy to change the production |
|
|
Disadvantages of batch process? |
- each batch needs to be supervised so it is labour intensive and very costly - Time is needed for cleaning if the product line is changed - it is ineffective as the production is not in use all the time |
|
|
How many years does it usually take to develop and test new drug? |
10 years |
|
|
How do you extract a chemical from a plant? 4 steps |
- crushing to disrupt and break cell walls - boiling in a suitable solvent to dissolve the compounds - chromatography to separate and identify individual compounds - isolating, purifying and testing potentially useful compounds |
|
|
Pure compounds have what melting point and boiling point? |
Definite |
|
|
Why is it difficult and costly to get a licence for a new drug? |
- thousands of compounds often need to be tested to find effective ones - need to be tested on living tissues to ensure safety - long term trials on humans are needed to identify possible side effects - many similar compounds needs to be developed to reduce side effects - recommended doses need to be shown effective - research needs to be independently verified - required to know expire date |
|
|
Three allotropes of carbon? |
Diamond Graphite Fullerenes |
|
|
What are allotropes? |
Allotropes are different structures of the same element |
|
|
Fullerenes are carbon structures that form what? |
Tubes or spheres |
|
|
Fullerenes are carbon structures that form spheres or tubes and can be used for what? |
To carry and deliver drug molecules around the body To trap dangerous substances in the body and remove them |
|
|
Diamonds and graphites are both what structures? |
Diamonds and graphites are both giant covalent structures of carbon atoms |
|
|
2 examples of giant covalent structures of carbon atom? |
Diamond and graphites |
|
|
Diamond structure? |
Every carbon atom in diamond makes strong covalent bonds in different directions |
|
|
Graphite structure? |
The carbon atoms bond to make layers |
|
|
Diamonds are what kind of natural substance and what melting and boiling point do they have? |
Diamonds are the hardest natural substances known and has very high melting point and boiling point |
|
|
The imperfection in diamond is what? |
Naturally occur in diamonds form cleaving plates which allow them to be shaped |
|
|
Good uses of diamonds? |
Cutting tools Jewellery Facets reflect light |
|
|
Graphite have what boiling point and melting point? |
High |
|
|
Graphite disadvantage ? |
Since graphites structure is layered, the layers can slide over each other |
|
|
Uses of graphites? |
Used in pencils High temperature lubricants |
|
|
Giant covalent bonding involves what? |
Electron sharing |
|
|
Every carbon atom in diamond is _____ bonded. |
Every carbon atom in diamond is covalently bonded |
|
|
The structure of diamonds give it what specific properties? (2) |
- strong covalent bonds in all directions make diamond hard. Large amounts of energy are required to break the bonds, giving High melting point - An absence of free electrons, meaning diamond doesn't conduct electricity |
|
|
Every carbon atom in graphite is covalently bonded to ___others in flat hexagonal layers |
Every carbon atom in graphite is covalently bonded to three others in flat hexagonal layers |
|
|
Graphites structure gives it specific properties which are.... |
- the strong covalent bonds give graphite a melting point similar to diamonds - delocalised electrons make it a good electrical conductor - when force is applied, the weak forces between the layers slide over each other; this slippery nature makes it an ideal high temperature lubricant |
|
|
Nanotubes can be used in what systems? |
Catalyst systems |
|
|
remember |
Nanotubes can be used in catalyst systems. Atoms of the catalyst can be attached to the large surface areas on the Nanotubes |

