![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
12 Cards in this Set
- Front
- Back
|
water: what are the stages of filtration? |
reservoir-sedimentation-filtration-chlorination- fluoridation- households |
|
|
water: why is fluorine and chlorine added to water? |
fluorine = to improve dental health chlorine = to kill harmful micro organisms like Bacteria |
|
|
water: why do we need water? |
drinking cooling towers in factories growing food on farms hygiene |
|
|
water: how can we decrease our water use ? |
have a shower instead of a bath use waste water for plants stop dripping taps turn off water while brushing teeth |
|
|
water: what is desalination? |
desalination is where large amounts of salt water is boiled to turn it into viable drinking water. because it need to be boiled, large amounts of energy is required so it is not suitable for many areas |
|
|
water: what is a fractional distillation column? |
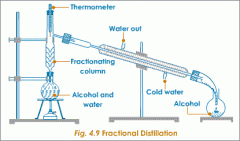
|
|
|
what is chromatography? |
It is the technique for separating the components of a mixture. |
|
|
Types of chromatography? |
paper chromatography - used to separate inks gas chromatography - find the amount of banned substances in athletes blood. atomic spectroscopy - used to tell what atoms are presents in a sample |
|
|
What is hard water? |
Hard water is water that has passed over limestone, this causes it to contain calcium ions. the water can also flow over gypsum which causes water to have magnesium ions. Water can be permanently hard or temporary hard. |
|
|
What is soft water? |
soft water is water that hasn't got calcium or magnesium ions. |
|
|
How is water defined? |
Hard water is classified as water that does not produce a lather, but soft water can produce a lather. |
|
|
How can hard water be turned soft? |
adding sodium carbonate (washing powder) ion exchange coloumn |

