![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
37 Cards in this Set
- Front
- Back
|
WHMIS? |
Workplace hazardous materials information system |
|
|
What are the three main requirements of WHMIS? |
-labels -material safety data sheet (msds) -worker education |
|
|
What are labels used for? |
-alert you of the type of hazardous material in the container -the dangers of the product -how to use the product safely |
|
|
Any chemical has three hazards associated with it, what are they? |
-flammability -reactivity -health |
|

? |
Compressed Gas -gas under high pressure and may explode easily |
|

? |
Flammable and Combustible -can catch fire easily |
|

? |
Oxidizing -contains highly reactive oxygen |
|
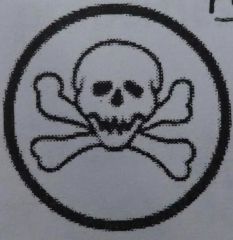
? |
Poisonous and Infectious Causing Immediate Serious Toxic Effects -very toxic, may be fatal |
|
|
HHPS? |
Hazardous Household Product Symbols |
|

? |
Danger (red) |
|

? |
Warning (orange) |
|

? |
Caution (yellow) |
|
|
Physical property? |
A characteristic of a substance |
|
|
Physical change? |
Change that does not affect a subtances size or form |
|
|
Chemical property? |
Characteristic behaviour that occurs when a substance changes to a new substance |
|
|
Chemical change? |
When substances react and rearrange their atoms to form a new substance |
|
|
What are five indicators that a chemical change has occured? |
-color change -odor change -formation of gas -formation of percipitate -energy change |
|
|
Precipitate? |
Solid material forming in liquid |
|
|
Compound? |
Pure substances that contain two or more different elements chemically combined |
|
|
Mixture? |
A substance that contains two or more different kinds of particles combined in some way |
|
|
Heterogeneous mixture? |
You can see the different componets |
|
|
Homogeneous mixture |
You can't see the different componets |
|
|
What are the three types of special mixtures? |
-solution -dispersion -suspension |
|
|
Solution? |
-homogeneous -the solid particals don't filter out -transparent (doesn't show tyndall effect) |
|
|
Dispersion? |
-heterogeneous -the solid particles dont filter out -semi transparent (show tyndall effect) |
|
|
Suspension? |
-heterogeneous -the solid particles will filter out -not transparent (show tyndall effect) |
|
|
_______ are homogeneous mixtures, made up of a _______ (the substance being dissovled) and a _______ (the substance doing the dissolving. |
-solution -solute -solvent |
|
|
What are the three types of solutions? |
-unsaturated -saturated -super saturated |
|
|
______________ is the process that occurs at saturation point, for every crystal at the bottom that forces its way into the solution, one must come out to make room for it. |
Dynamic equilibrium |
|
|
Melting? |
-solid to liquid -volume ⬆ -density ⬇ -heat must be added -water is an exception |
|
|
Freezing? |
-liquid to solid -volume ⬇ -density ⬆ -heat must be removed |
|
|
Modern atomic theory? #5 |
The law of definite porportions states when atoms combine to form compounds they do so in the ratio of small whole numbers. |
|
|
Modern atomic theory? #6 |
The law of multiple porportions states that sometimes atoms of the same element can combine to form different compounds. |
|
|
Modern atomic theory? #6 |
The law of multiple porportions states that sometimes atoms of the same element can combine to form different compounds. |
|
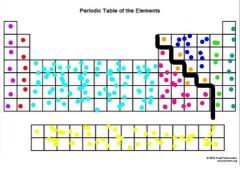
What are the groups? |
Purple - alkali metals Red - alkaline earth metals Light Blue - transition metals Yellow - inner transition metals Pink - metals Orange - metalloids Dark Blue - non metals Green - halogens Teal - noble gases |
|
|
Atoms are _____. The number of _____ = the number of _____. They will give up neutrality to become stable this is called the ___________. |
Neutral Electrons Protons Octet Rule |
|

? |
Dangerously Reactive -may react violently with almost any chemical to produce potentially dangerour reactions |

