![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
12 Cards in this Set
- Front
- Back
|
COMPOUND |
a substance that is composed of two or more different elements; a mixture. Ex. H2O, CO2 |
|
|
ELEMENT |
A substance that cannot be broken down into simpler substances by chemical means
|
|
|
MOLECULE |
The smallest piece of a substance. Ex. H2O, O2, CO2 |
|
|
SYMBOLS |

A chemical symbol is a code for a chemical element. It is usually derived from the name of the element, often in Latin. |
|
|
CHEMICAL REACTIONS |
In a chemical change where there is a chemical reaction, a new substance is formed and energy is either given off or absorbed. |
|
|
PHYSICAL CHANGE |

A physical change in a substance doesn't change what the substance is. |
|
|
REACTANT |
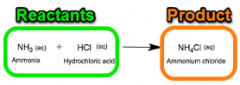
A substance that takes part in and undergoes change during a reaction. |
|
|
SOLUTION |
A mixture composed of 2 or more substances. |
|
|
PRECIPITATE |
A precipitate is a solid that emerges from a liquid solution. |
|
|
CHEMICAL FORMULA |
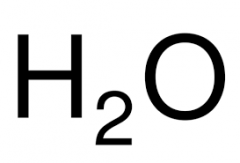
A recipe using coefficients and subscripts to explain what a substance is made of. |
|
|
COEFFICIENT
|
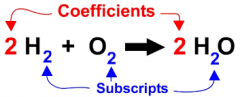
A number or symbol multiplied with a variable or an unknown quantity in an algebraic term. |
|
|
SUBSCRIPT |
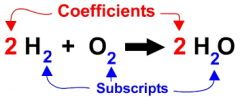
A type of lettering form written lower than the things around it. In chemical formulas the number of atoms in a molecule is written as a subscript, so we write H2O for water which has two atoms of hydrogen for each one of oxygen. |

