![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
11 Cards in this Set
- Front
- Back
|
How many P.S.I. Are equal to 1 atmosphere |
14.7 |
|
|
What is boyle's law |
P1 X V1 = P2 X V2 |
|
|
What is charle's law |
V1/T1 = V2/T2 |
|
|
Gas constant |
0.08206(atm x L)/(n x T) |
|
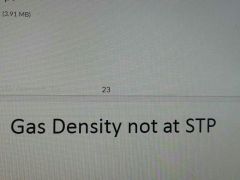
|

|
|
|
1 ATM = ???mm Hg |
760 mm Hg |
|
|
1 mm Hg = ??? torr |
1mm Hg= 1 torr |
|
|
Formula for pressure |
P=F/A |
|
|
Kinetic energy formula |
KE= 1/2mv² |
|
|
Standard molar volume of gas |
22.4 L = 1 mole of a gas |
|
|
= joule |
1Kg m²/s² |

