![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
46 Cards in this Set
- Front
- Back
|
Cations are formed from _____ or _____.
|
metals
polyatomic ions (those with a positive charge) |
|
|
Some metals have predictable charges. What are these called?
|
Trustworthy Metals
|
|
|
Why are trustworthy metals considered predictable?
|
The charge can be predicted from the valence electrons (i.e. they only form cations with one possible charge).
|
|
|
Name the trustworthy metals.
|

Group 1, 2, and 3 and aluminum, zinc, and silver.
|
|
|
_____ _____ form cations with different charges.
|
Untrustworthy Metals
|
|
|
What are the Untrustworthy Metals?
|
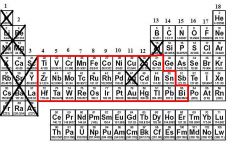
All of the metals except group 1, 2, 3, and aluminum, zinc, and silver.
|
|
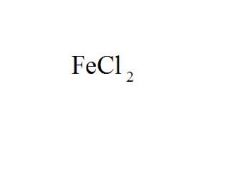
Give the EXACT name of the compound.
|

iron (ii) chloride
|
|
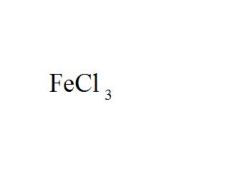
Give the EXACT name of the compound.
|
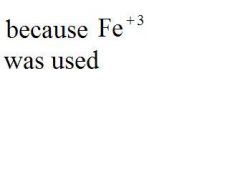
iron (iii) chloride
|
|
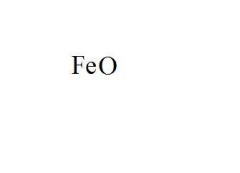
Give the EXACT name of the compound.
|

iron (ii) oxide
|
|
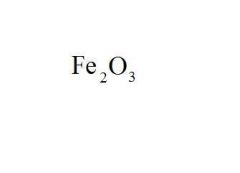
Give the EXACT name of the compound.
|
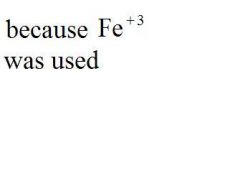
iron (iii) oxide
|
|
|
All nomenclature using untrustworthy metals requiring how many names?
|
three (a first, a middle, and a last)
e.g. copper (ii) sulfate |
|
|
In compounds made with untrustworthy metals, the first name is the name of the _____, the middle name is a _____ _____ describing which metal was used, and a last name describing the _____.
|
metal
roman numeral nonmetal/anion |
|
|
_____ are compounds that have hydrogen as the cation.
|
acids
|
|
|
Acids are compounds that have _____ as the cation.
|
hydrogen
|
|
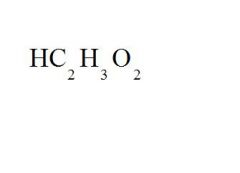
Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen acetate
acid name = acetic acid |
|

Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen sulfate
acid name = sulfuric acid |
|

Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen nitrate
acid name = nitric acid |
|

Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen carbonate
acid name = carbonic acid |
|

Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen sulfide
acid name = hydrosulfuric acid |
|

Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen fluoride
acid name = hydrofluoric acid |
|
|
Acids with ionic names that end in "ate" have acid names that end in _____.
|
"ic"
|
|
|
Acids with ionic names that end in "ite" have acid names that end in _____.
|
"ous"
|
|
|
Acids with ionic names that end in "ide" have acid names that start with _____ and end in _____.
|
"hydro"
"ic" |
|
|
What is the formula and the acid name for hydrogen acetate?
|

formula = see above
acid name = acetic acid |
|
|
What is the formula and the acid name for hydrogen sulfate?
|

formula = see picture
acid name = sulfuric acid |
|
|
What is the formula and the acid name for hydrogen nitrate?
|
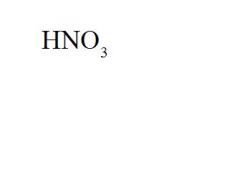
formula = see picture
acid name = nitric acid |
|
|
What is the formula and the acid name for hydrogen carbonate?
|

formula = see picture
acid name = carbonic acid |
|
|
What is the formula and the acid name for hydrogen sulfite?
|

formula = see picture
acid name = sulfurous acid |
|
|
What is the formula and the acid name for hydrogen nitrite?
|
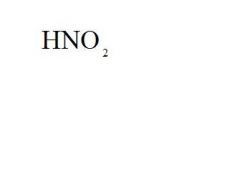
formula = see picture
acid name = nitrous acid |
|
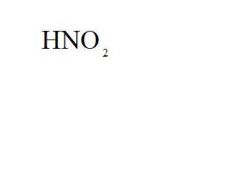
Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen nitrite
acid name = nitrous acid |
|
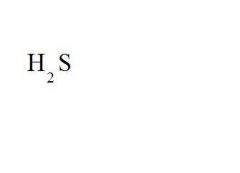
Give the ionic name and the acid name for this compound.
|
ionic name = hydrogen sulfide
acid name = hydrosulfuric acid |
|
|
What is the formula and the acid name for hydrogen sulfide
|

formula = see picture
acid name = hydrosulfuric acid |
|
|
What is the formula and the acid name for hydrogen fluoride?
|

formula = see picture
acid name = hydrofluoric acid |
|
|
Give the formula and the ionic name for acetic acid.
|

formula = see picture
ionic name = hydrogen acetate |
|
|
Give the formula and the ionic name for sulfuric acid.
|

formula = see picture
ionic name = hydrogen sulfate |
|
|
Give the formula and the ionic name for nitric acid.
|

formula = see picture
ionic name = hydrogen nitrate |
|
|
Give the formula and the ionic name for carbonic acid.
|

formula = see picture
ionic name = hydrogen carbonate |
|
|
Give the formula and the ionic name for sulfurous acid.
|

formula = see picture
ionic name = hydrogen sulfite |
|
|
Give the formula and the ionic name for nitrous acid.
|

formula = see picture
ionic name hydrogen nitrite |
|
|
Give the formula and the ionic name for hydrosulfuric acid.
|

formula = see picture
ionic name = hydrogen sulfide |
|
|
Give the formula and the ionic name for hydrofluoric acid.
|

formula = see picture
ionic name = hydrogen fluoride |
|
|
Name the four classes of ionic compounds.
|
Acids
Bases Oxides Salts |
|
|
What is an acid?
|

see the picture
|
|
|
What is a base?
|

see the picture
|
|
|
What is an oxide?
|

see the picture
|
|
|
What is a salt?
|

see the picture
|

