![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
36 Cards in this Set
- Front
- Back
|
Big Ideas: Life is work
|
- energy flows into an ecosystem as sunlight and leaves as heat - Photosynthesis generates O2 and organic molecules, which are used in cellular respiration - cells use chemical energy stored in organic molecules to regenerate ATP, which powers work |
|
|
4 steps of cellular Respiration
|
Catabolic pathways yield ATP by oxidizing organic fuels 1.Glycolysis: glucose is broken down to pyruvate 2. Pyruvate processing: pyruvate is oxidized to form acetyl CoA 3. Citric acid cycle: Acetyl CoA is oxidized to CO2 4. Electron transport and chemiosmosis: electron carriers used in 1-3 transfer electrons to proteins which create a force for ATP production |
|
|
Cellular respiration is the controlled release of energy from organic molecules (such as sugars) in cells to form....
|
ATP: adenosine Triphosphate C6H12O6 + 6O2 6CO2 + 6H2O + Release of Energy 40% 60% ATP Heat |
|
|
Where is energy stores?
|
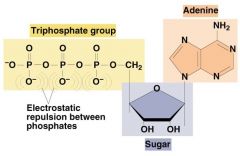
in the molecule ATP, the energy currency of the cell
|
|
|
Why does cellular respiration have to be a controlled release of energy? |
If energy is not stepwise, 1. a large release of energy occurs 2. as in the reaction of hydrogen and oxygen to form water 3. cell to fragile; self ignite |
|
|
How do cells achieve controlled release of energy? |
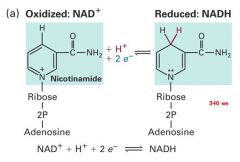
- a series of steps: Redox reactions- oxidation and reduction *Remember OILRIG! - Oxidation:(NAD+) is the loss of electrons and hydrogens - Reduction (NADH) is the gains of electrons and hydrogens - 686kcal/mol |
|
Glucose Catabolism |
the high energy electrons and hydrogens are stripped from glucose and picked up by NAP+ and FADH
|
|
|
What is chemical reaction for, where are electron carriers taking them?
|
To electron transport system: end point, lots of proteins TERMINAL ELECTRON ACCEPTOR IN CELLULAR RESPIRATION IS ENDPOINT OXYGEN- molecular oxygen |
|
|
Why is taking in oxygen so important?
|
hydrogen would make enviro very acidic
|
|
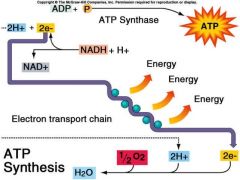
Electron Transport Chain
|
NADH and FADH2 passes electrons and hydrogens in a series of steps instead in one explosive reaction - uses the energy from the electron transfer to form ATP |
|
|
Stages of cellular respiration
|
Glycolysis (ATP "substrate level")> pyruvate> Citric acid cycle ("substate level, ATP electron carriers NADH and FADN2)> to... Oxidative phosphorylation (ATP32,38) : Electron transport and chemiosmosis (4 proteins) |
|
|
What is Phosphorylation? |
is the addition of a phosphate group to an organic molecule, in our case ADP - a small mount of ATP is formed in glycolysis and the citric acid cycle by substrate level phosphorylation - oxidative phosphorylation accounts for almost 90% of the ATP generated by cellular respiration |
|
|
Glycolysis |
glycolysis harvests energy by oxidizing glucose to pyruvate - means "splitting of sugar" - breaks down glucose into pyruvate -occurs in the cytoplasm of the cell |
|
|
Glucose is energized using ATP Steps 1-3
|

|
|
|
Step 4: glucose is broken into two 3 carbon intermediates |

|
|
|
Step 5 : a redox reaction generates NADH Step 6-9: ATP and Pyruvate are produced |

|
|
|
Glycoysis Summary |
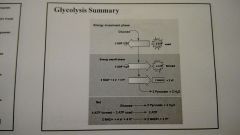
|
|
|
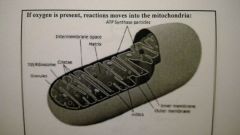
If oxygen is present, reactions moves into the mitochondria |
|
|
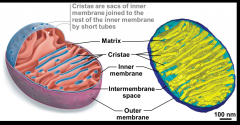
The mitochondrial matrix
|
- matrix is inside the inner membrane - in-folding of the inner memebrane, called CRISTAE, makes the intermembrane space |
|
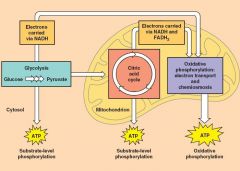
To begin the Citric Acid Cycle....
|
pyruvate is converted to acetyl CoA - occurs in the mitochondria (matrix) |
|
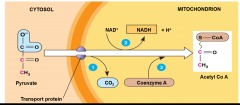
Oxidation of Pyruvate:
|
-more energy can be extracted if oxygen is present - within mitochondria, pyruvate is decarboxylated, yielding acetyl-CoA,NADH, and CO2 - before citric acid cycle can begin, pyruvate must be converted to acetyl CoA |
|
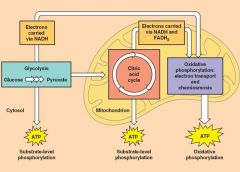
Citric Acid Cycle
|
the citric acid cycle completes the oxidation of organic molecules, generating many NADH and FADH2 molecules - occurs in the mitochaondia matrix - also known as the Krebs cycle - the cycle oxidizes organic fule derived from pyruvate, generating 3 NADH, 1 ATP, and 1 FADH2 per turn |
|
|
Break down of the Citric acid cycle (8 steps) and each step catalyzed by a specific enzyme
|
the acetyl joins the cycle by combining with oxaloacetate, forming citrate - the next 7 steps decompose the citrate back to oxaloacetate, making the process a cycle - The NADH and FADH2 produced by the cycle relay electrons extracted from food to the electron transport chain |
|
|
Oxidative Phosphorylation |
following glycolysis and the citric acid cycle, NADH and FADH2 account for most of the energy extracted from food - These two electron carriers donate electrons to electron transport chain, which creates a proton motive force by moving H+ across the inner mitochondrial memebrane - The Proton Motive Force (PMF) is used to make ATP by ATP synthase proteins |
|
|
Electron Pathway |
-Electrons are transferred from NADH or FADH2 to the electron transport chain. -The electron transport chain occurs in the folds of mitochondrion inner membrane. -The chain’s proteins relay electrons to each other while moving protons across the membrane -Electrons lose free energy as they go down the chain and are finally passed to O2, forming H2O. -The electron transport chain generates no ATP. The chain’s function is to pump protons into the inter membrane space, creating a proton gradient or PMF |
|
|
Proton-motive force |
the energy stored in the H+ gradient across a membrane is referred to as a proton-motive force, emphasizing its capacity to do work
|
|
|
where does the electron transport chain occur?
|
occurs in the inner membrane of the mitochondria (membranes of cristae) |
|
|
Chemiosmosis: The energy-coupling Mechanism
|
-Electron transfer in the electron transport chain causes proteins to pump H+ from the mitochondrial matrix to the intermembrane space -H+ then moves back across the membrane, passing through channels in ATP synthase -ATP synthase (an enzyme) uses the exergonic flow of H+ to drive phosphorylation of ATP -This is an example of chemiosmosis, the use of energy in a H+ gradient to drive cellular work |
|
|
Chemiosmosis: the use of H+ gradient to make ATP
|
ATP synthase uses the exergonic flow of H+ to drive phosphorylation of ATP. -ATP synthase acts as a molecular motor (Protons flowing through the F0 unit spin the rotor) -The rotor spins the F1 unit(As the F1 unit spins its subunits change shape) -Catalyze the phosphorylation of ADP to ATP |
|
|
Fermentation |
breakdown of organic molecules without net oxidation - many organisms can only use O2 as final e- acceptor, so under Anaerobic conditions, they need different way to produce ATP, e.g. glycolysis) - muscle cells solve probelm by reducing pryuvate into lactate - Yeast solve probelm my making ethanol - Fermentaion produces far less ATP than oxidative phosphorylation (34-38 ATP in aerobic; 2 ATP in anarobic) |
|
|
Obligate anaerobes |
carry out fermentaion or anaerobic respiration and cannot survive in the presence of O2
|
|
|
Faculatative |
(yeast and many bacteria) meaning that they can survive using either fermentation or cellular respiration - in faculative cells, pyruvate is a fork in the metabolic road that leads to two alternative digestive routes |
|
|
Glycolysis and the citric acid cycle connect to many other metabolic pathways
|
Gycolysis and the citric acid cycle are major intersections to various catabolic and anabolic pathways |
|
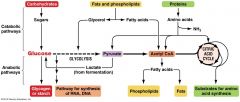
|
|
|
|
Fermentation and Aerobic Respiration Compared
|
-Both processes use glycolysis to oxidize glucose and other organic fuels to pyruvate -The processes have different final electron acceptors: an organic molecule in fermentation (such as pyruvate or acetaldehyde) and O2 in cellular respiration -Cellular respiration produces 29 ATP per glucose molecule; fermentation produces 2 ATP per glucose molecule |
|
|
Control of catabolism is based mainly on ...
|
regulating the activity of enzymes at strategic points in the catabolic pathway
|

