![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
527 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Vasculitis Definition, Histology, Etiology |
-inflammation of the blood vessel wall ---arterial wall is composed of: endothelial intima, SM media (endothelial cells on BM), and connective tissue adventitia -Etiology unknown |
|
|
|
Clinical Features of Vasculitis |
-nonspecific symptoms: inflammation = fever, fatigue, weight loss, and myalgias -symptoms of organ ischemia ---1. inflammation >> endothelial damage >> coagulation cascade >> thrombus ---2. inflammation >> healing & fibrosis >> luminal narrowing
|
|
|
|
Categorization of Vasculitis & Definition of Each |
-Large Vessel Vasculitis: involves aorta and its major branches -Medium Vessel Vasculitis: involves muscular arteries that supply organs -Small Vessel Vasculitis: involves arterioles, capillaries, and venules |
|
|
|
Types of Large Vessel Vasculitis |
-Temporal (Giant Cell) Arteritis -Takayasu Arteritis |
|
|
|
Temporal Arteritis
Definition/ Involvement? Seen in? |
-Large Vessel Vasculitis (of temporal artery & its branches) -Granulomatous vasculitis that classically involves branches of the carotid artery -most common form of vasculitis in older adults (>50 yrs old); usually affects women |
|
|
|
Temporal Arteritis Presentation & Associated Vascular Involvement |
-Headache from temporal artery involvement; unilateral headache -Visual disturbances from ophthalmic artery involvement/occlusion >> irreversible blindness -Jaw Claudication (muscles of jaw affected) -Polymyalgia rheumatica: Flu-like symptoms with joint and muscle pain (if no elevated CK, then it is this, if elevated CK, polymyositis) ---ESR elevated in these patients (>100) - if this isn't elevated, then it isn't a vasculitis >> TIA |
|
|
|
Temporal Arteritis Biopsy/Diagnosis |
-reveals inflamed vessel wall with giant cells and intimal fibrosis ---Lesions are segmental; diagnosis requires biopsy of a long segment of vessel and a negative vessel doesn't exclude the disease -inflammation >> increased ESR |
|
|
|
Temporal Arteritis Treatment & Complications |
-Treatment: corticosteroids prior to temporal artery biopsy to avoid vision loss -High Risk of blindness without treatment; therefore, treat immediately |
|
|
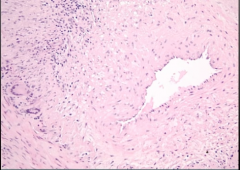
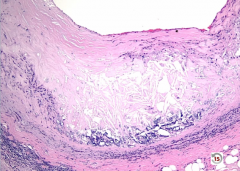
Biopsy of Vessel |
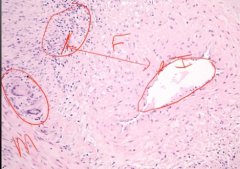
-broad area of fibrosis in between intima and media (separation should not be there) -fibrosis >> narrow lumen -inflammation in blood vessel wall -giant cells also observed |
|
|
|
Takyasu Arteritis
Definition? Seen In? Presentation? Treatment |
-Large Vessel Arteritis (elastic arteries) -Granulomatous vasculitis (thickening & narrowing) that classically involves the aortic arch & proximal great vessels -Presents in adults <40-50 yo (classically, young, Asian females) as visual and neurologic symptoms with a weak or absent pulse in the upper extremitiy ('pulseless disease'), fever, night sweats, arthritis, myalgias, skin nodules, ocular disturbances -Elevated ESR -Treatment: corticosteroids |
|
|
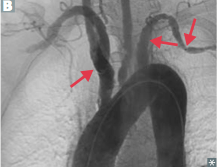
Aortic Arch |
Takayasu arteritis thickening and narrowing of aortic arch and its branches (proximal great vessels) |
|
|
|
Difference Between Temporal Arteritis & Takyasu Arteritis |
-Temporal Arteritis affects individuals > 50 old & distal (branches of carotid artery) -Takyasu affects Asian females < 50 yo & proximal (aortic arch branch points) |
|
|
|
Polyarteritis Nodosa
Definition? Seen In? |
-Medium Vessel Vasculitis - muscular arteries that supply organs -Necrotizing vasculitis involving multiple organs (renal and visceral) EXCEPT the lung -Seen In: young adults |
|
|
|
Polyarteritis Nodosa
Presentation & Pathogenesis/Involvement? |
-Hypertension: due to renal artery involvement >> renal damage -Abdominal Pain with melena: mesenteric artery involvement -Neurologic disturbances -Skin lesions - cutaneous eruptions -fever, weight loss, malaise, headache -Associated with HBsAg (30%) -pANCA (according to Goljan's) |
|
|
|
Polyarteritis Nodosa
Lesions/Stages? Pathogenesis of Imaging? |
-Lesions of varying stages are present -Early lesions consist of transmural inflammation with fibrinoid necrosis >> eventually heals (end stage) with fibrosis (feels bumpy) >> string of pearls appearance on imaging (long vessel w/ lesions of varying stage of dense fibrosis and inflammation >> weakened wall of vessel >> numerous microaneurysms & vasopasm on arteriogram) |
|
|
|
Polyarteritis Nodosa
Treatment? Prognosis? |
-Treatment: corticosteroids of cyclophosphamide -Fatal if not treated |
|
|
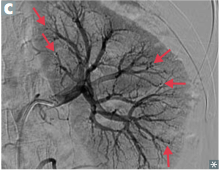
|
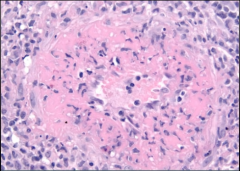
Polyarteritis Nodosa -numerous microaneurysms |
|
|
Biopsy of Vessel |
Polyarteritis Nodosa -fibrinoid necrosis w/ highlighter pink! |
|
|
|
Kawasaki Disease
definition? Seen in? |
-medium vessel vasculitis (muscular) -Seen In: Asian children < 4 yo
|
|
|
|
Kawasaki Disease
Presentation? |
-presents with nonspecific symptoms: fever, conjunctivitis, strawberry tongue (and lips), erythematous rash of palms and soles, enlarged cervical lymph nodes, desquamating rash -risk of thrombosis >> infarction >> can present with MI |
|
|
|
Kawasaki Disease
Pathogenesis/Involvement? Complications? |
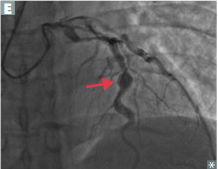
-coronary artery involvement common >> risk of thrombosis with MI and aneurysm with rupture -so could present with a child with an MI! |
|
|
|
Kawasaki Disease
Treatment? Prognosis? |
-Treat with aspirin to protect against thrombus (watch out!: this presents similar to a viral illness >> can't give a child w/ a viral illness aspirin or they will get Reye Syndrome) and IVIG -Disease is self-limited |
|
|
|
Reye Syndrome |
-when you give ASA to a child with a viral illness -results in encephalopathy & massive liver necrosis |
|
|
|
Buerger Disease
Pathogenesis/Involvement? Seen in? Treatment? |
-medium-vessel (muscular) segmental, thrombosing vasculitis involving the digits -Highly associated heavy smoking, males < 40 -Treat with smoking cessation |
|
|
|
Buerger Disease Presentation |
-Intermittent claudication >> ulceration, gangrene, superficial nodular phlebitis, and autoamputation of fingers & toes -Raynaud phenomenon often present (caused by vasospasm) |
|
|
|
Wegener Granulomatosis
Pathogenesis/Involvement? Seen In? |
-small-vessel vasculitis (muscular) =granulomatosis w/ polyangiitis -necrotizing granulomatous vasculitis involving the nasopharynx, lungs, and kidneys -middle aged male |
|
|
|
Wegener Granulomatosis Presentation, Findings, & Biopsy |
-middle aged male with chronic sinusitis or nasopharyngeal ulceration (perfration of nasal septum >> saddle nose deformity), otitis media, mastoiditis, hemoptysis, cough, dyspnea, with bilateral nodular lung infiltrates on CXR, and hematuria & red cell casts due to rapidly progressive glomerulonephritis -serum PR3-ANCA/c-ANCA (anti-neutrophil cytoplasmic antibody) levels correlate with disease activity -Biopsy: large necrotizing granulomas with adjacent necrotizing vasculitis |
|
|
|
Wegener Granulomatosis Treatment & Prognosis |
-cyclophosphamide and steroids -relapse common |
|
|
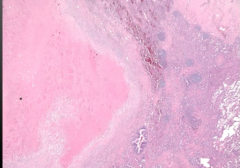
Vessel Biopsy |
Wegener Granulomatosis
-large area of necrosis (forms lung nodules) -wall has giant cells |
|
|
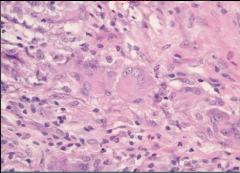
Vessel Biopsy |
Wegener Granulomatosis
-giant cells with epithelial histiocytes |
|
|
|
Microscopic Polyangiitis
Pathogenesis/Involvement?
|
-small vessel vasculitis -necrotizing vasculitis involving multiple organs, especially lung, kidney, and skin |
|
|
|
Microscopic Polyangiitis
Presentation? How is it different from Wegener Granulomatosis? |
-hemoptysis due to lung involvement -hematuria due to pauci-immune glomerulonephritis -palpable purpura -MPO-ANCA/p-ANCA levels correlate with disease activity -Difference: no nasopharyngeal involvement, no granulomas, & p-ANCA instead of c-ANCA |
|
|
|
Microscopic Polyangiitis
Treatment? |
-cyclophosphamide & corticosteroids -relapse common |
|
|
|
Churg-Strauss Syndrome
Pathogenesis/Involvement |
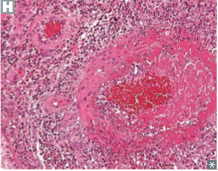
-small vessel vasculitis -necrotizing granulomatous inflammation with eosinophils involving multiple organs, especially lungs and heart |
|
|
|
Churg-Strauss Syndrome
Presentation? How to distinguish from Microscopic Polyangiitis? |
-Asthma, sinusitis, palpable purpura, wrist/foot dropp due to peripheral neuropathy, peripheral eosinophilia, increased IgE -Can also involve heart, GI, kidneys (pauci- immune glomerulonephritis) -serum MPO-ANCA/ p-ANCA levels correlate with disease activity -Distinguish from MP: MP has no granulomas, no hx of asthma, no eosinophilia |
|
|
|
Henoch-Schonlein Purpura
Pathogenesis/Involvement? Seen In? |
-Small vessel vasculitis -Vasculitis due to IgA immune complex deposition -most common vasculitis in children |
|
|
|
Henoch-Schonlein Purpura
Presentation & associated pathogenesis |
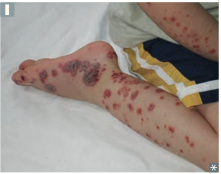
-Palpable purpura on buttocks and legs (bleeding into skin purpura are NOT palpable) -GI pain and bleeding (melana & multiple lesions of same age) -Arthrlagias -Hematuria (IgA nephropathy) - RBC casts -follow an URI (due to high levels of IgA >> deposits) |
|
|
|
Henoch-Schonlein Purpura
Treatment and Prognosis |
-Disease is self-limited, but may recur -Treat with steroids if severe |
|
|
|
Infectious vasculitis
Affects what? Seen in? Causes |
-small vessel vasculitis involving skin vessels -Seen in children and adults -Involves all microbial pathogens -Rickettsia rickettsii & Neisseria meningitidis |
|
|
|
Infectious vasculitis
Tick associated? Cause? Pathogenesis? Presentation? |
-RMSF most prevalent in SE then central states -Caused by dog tick by transferring Rickettsia reckettsii -Organisms invade endothelial cells >> vasculitits -Fever in 100% of cases -Petechiae begins on palms and spreads to trunk ---appear in irst days in 50% of cases, and by 5th day in 80%; no petechiae in 10% |
|
|
|
Infectious vascultiis
Neisseria meningitidis - Pathogenesis? Treatment? |
-Disseminated meningococcemia due to Neisseria meningitidis -Capillary thromboses, usually due to DIC, produce minute hemorrhages into the skin (petechiae) that become confluent ecchymoses as the disease progresses. Hemorrhagic infractions of both adrenal glands commonly occurs producing acute hypocortisolism and death (Waterhouse-Friderichsen syndrome) -Treat: IV penicillin G |
|
|
|
Hypertension definition |
-increased BP in pulmonary or systemic circuit -Systemic HTN = pressure ≥ 140/90 mmHg (20% of population); can be just systolic ≥140 or diastolic ≥ 90 -Normal BP ≤120/80 mmHg -usually systolic and diastolic increased together, but can be one or the other |
|
|
|
Primary Hypertension
% of cases? Risk Factors? Pathogenesis? |
-etiology unknown - related to increase CO and TPR -95% (90% in first AID) of cases -Risk factors: age, race (increased in African Americans, decreased in Asians), obesity, stress, lack of physical activity, high-salt diet -Pathogenesis: excess salt increased blood volume >> stroke volume >> effects systolic BP excess salt enters muscles, opens Ca channels >> contraction >> increased TPR >>increased diastolic pressure |
|
|
|
Is Renin high or low in primary HTN?
First-line treatment? Why? |
-low b/c of the increased BP due to Na retention -Hydrochlorathizide first line treatment for essential HTN in elderly and blacks because you get rid of salt and water |
|
|
|
Secondary Hypertension
% of cases? most common cause? |
-5% (10% in First Aid) of cases -due to identifiable cause -Most common cause: renal artery stenosis ----also related to fibromuscular dysplasia |
|
|
|
Hypertension Risk Factors [causes] Hypertension Predisposes you to...? |
-Risk Factors: age, obesity, diabetes, smoking, genetics, black > white > Asian -Causes: [sleep apnea, CKD is most common, drug-induced, primary aldosteronism, renovascular disease, chronic steroid therapy/Cushing's, Pheochromocytoma, coarctation of the aorta, thyroid/parathyroid disease -Predisposes to: atherosclerosis, LVH, stroke, CHF, renal failure, retinopathy, aortic dissection, Charcot Bouchard aneurysms (MCA) |
|
|
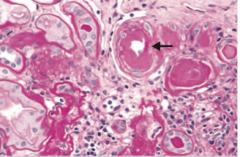
|
-Hyaline arteriosclerosis, specifically hypertensive nephropathy |
|
|
|
Renovascular Hypertension
Pathogenesis? |
-Due to renal artery stenosis -stenosis >> decreased blood flow to glomerulus >> JGA secretes renin >> angiotensinogen converted to angiotensin I >> angiotensin I converted to angiotensin II by ACE >> angiotensin II raises BP by contracting arteriolar SM increasing TPR and by promoting adrenal release of aldosterone, which increases resporption of Na in the distal convoluted tubule expanding plasma volume >> causes HTN with increased plasma renin and unilateral atropy (due to low blood flow) of the affected kidney |
|
|
|
Renovascular Hypertension
Causes? Seen In? |
-Renal artery stenosis caused by ---atherosclerosis (elderly males) ---fibromusclular dyspasia (thickening of wall) (young females) |
|
|
|
Fibromuscular Dysplasia |
development defect of the blood vessel wall, resulting in irregular thickening of large- and medium-sized arteries, especially the renal artery
cause of renovascular hyptertension/ secondary hypertension |
|
|
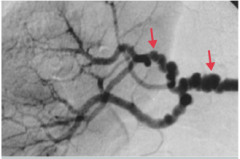
|
Fibromuscular dysplasia
-"string beads" appearance of renale artery in this picture -important cause of HTN in young patients |
|
|
|
Benign Hypertension |
-mild or moderate elevation in BP -most cases are benign -clinically silent: vessels and organs slowly damaged over time |
|
|
|
Malignant Hypetension
Definition? Presentation? |
-severe elevation in BP ≥180/120 mmHg -5% of cases -Can arise from preexisting HTN or de novo -Presents with acute, ongoing end-organ damage: acute renal failure, headache, mental status changes, and papilledema -MEDICAL EMERGENCY -another cause of fibrinoid necrosis of the blood vessel |
|
|
|
Arteriosclerosis Definition |
-thickening of blood vessel wall -hard arteries |
|
|
|
Atherosclerosis
Definition and Composition |
-disease of elastic and medium & large arteries -intimal plaque that obstructs blood flow ---constists of necrotic lipic core (mostly cholesterol) with a fibromusular cap that can undergo dystrophic calicification |
|
|
|
-Atherosclerosis -Necrotic plaque in intima with cholesterol deposition and fibromusclar cap |
|
|
|
Atherosclerosis
Involvement? Most common? |
-Involves large and medium sized arteries -Most common: abominal aorta > coronary artery > popliteal artery > internal carotid artery |
|
|
|
Atherosclerosis
Risk Factors/Seen In?
|
-Modifiable: HTN, hypercholesterolemia/hyperlipidemia (LDL increases risk, HDL decreases risk), smoking, diabetes -Nonmodifiable: age (number & severity increase with age), gender (increased in males and postmenopausal women b/c estrogen is protective), genetics (multifactorial - family history slightly predictive of risk) |
|
|
|
Atherosclerosis Pathogenesis |
-Damage to endothelium/endothelial cell dysfunction allows lipids to leak into the intima >> Lipids are oxidized and then consumed by macrophages via scavenger receptors >> foam cells (lipid laden macrophages) >> fatty streaks (begins in teens) -Inflammation and healing leads to deposition lipid in intima>> thickening of intima with necrotic lipid core >> healing >> deposition of ECM and proliferation/migration of SM (via PDGF and FGF) >> fibromuscular cap >> fibrous cap >> complex atheroma |
|
|
|
[Why is oxidized LDL so bad?] |
-activates inflammatory cytokines -attracts macrophages to vessel walls & up regulates chemotactic factors (IL-1, TNF, IFN-y & IL-6 >> acute phase reactants) -promotes endothelial and smooth muscle apoptosis -promotes platelet aggregation and thrombosis -activates endothelium >> up regulation of LAMs |
|
|
|
Atherosclerosis Complications/Symptoms |
-complications of atherosclerosis account for >50% of disease in Western countries -no symptoms until >70% stenosis >> angina, claudication 1. Stenosis of medium-sized vessels results in impaired blood flow (narrowing) and ischemia >> ---peripheral vascular disease (lower extremity arteries like popliteal) ---Angina (coronary arteries) ---Ischemic bowel disease (mesenteric arteries) 2. Plaque rupture w/ thrombosis >> MI (coronary arteries) or stroke (middle cerebral arteries) 3. Plaque rupture with emobilization >> atherosclerotic emboli, characterized by cholesterol clefts (crystals in embolus) 4. Weakening of vessel wall (intimal thickening >> diffusion barrier >> wall becomes atrophic) >> aneurysm (abdominal aorta) |
|
|
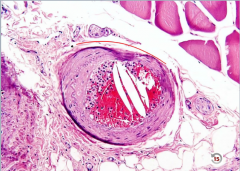
|
Atherosclerotic embolus with cholesterol clefts |
|
|
|
Hyaline Arteriolosclerosis Pathogenesis |
-caused by proteins leaking into the vessel wall >> vascular thickening ---proteins seen as pink hyaline on microscopy |
|
|
|
Hyaline Arteriolosclerosis Etiology/Causes & Complications
|
-Consequence of long-standing benign (essential) hypertension (forces protein into wall) or diabetes (nonenzymatic glycosylation of basement membrane >> vessel leaky)
-Results in reduced vessel caliber with end-organ ischemia ---classically produces glomerular scarring (arteriolonephrosclerosis) that slowly progresses to renal failure |
|
|
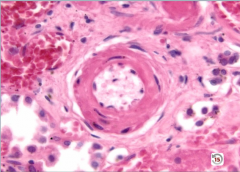
|
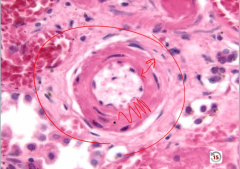
Hyaline Arteriolosclerosis
wall thickened by hyaline |
|
|
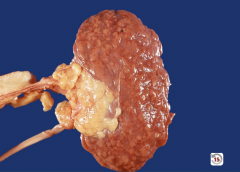
|
arteriolonephrosclerosis
-can see the scarring on the cortex of the kidney |
|
|
|
Hyperplastic Arteriolosclerosis Pathogenesis
Caused by? |
-involves thickening of vessel wall by hyperplasia of smooth muscle >> 'onion skin' appearance |
|
|
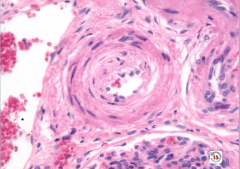
|
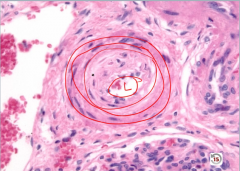
Hyperplastic Arteriolosclerosis
-onion skinning = too many layers of smooth muscle cells -narrow lumen >> reduced vessel caliber
|
|
|
|
Hyperplastic Arteriolosclerosis Causes & Complications |
-consequence of malignant hypertension -Results in reduced vessel caliber with end-organ ischemia -May lead to fibrinoid necrosis of the vessel wall with hemorrhage ---Classically causes acute renal failure with characteristic 'flea bitten' appearance (pinpoint hemorrhages on surface of kidney due to blowing out of blood vessel) |
|
|
|
Monckeberg Medial Calcific Sclerosis
What is it? Complications? |
-Calcification of the media of musclar (medium sized) arteries, esp. radial and ulnar arteries -Non-obstructive (doesn't alter lumen caliber/ intima not involved) -Not clinically significant: seen as an incidental findings on xray or mammography ("pipestem" arteries" |
|
|
|
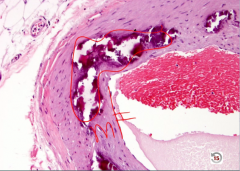
Monckeberg Medial Calcific Sclerosis
-media up against intima like it should be -media calcifed |
|
|
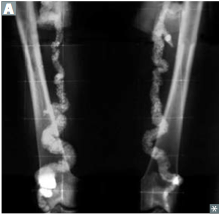
|
Monckenberg (medial calcific sclerosis) -showing "pipestem" arteries on xray -ulnar and radial arteries most common |
|
|
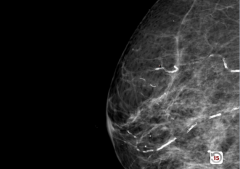
Mammogram |
Monckeberg Medial Calcification
-calcification running in vascular pattern |
|
|
|
Aortic aneurysms
What are they? Presentation? Indications of presentation? |
-localized pathologic dilation of the Aorta -May present with pain, which may indicate leaking, dissection, or imminent rupture |
|
|
|
Aortic Dissection
What is it? Where is it & requirements? |
-Intimal tear with dissection of blood through the media of the aortic wall >> longitudinal intraluminal tear >> false lumen -Occurs in the proximal 10 cm of the aorta (high stress region) with preexissting weakness of the media |
|
|
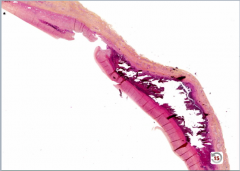
|
Aortic Dissection
-blood through intima, through media to wall |
|
|
|
Aortic Dissection
Causes & their associated pathogenesis |
-Most common causes is hypertension (older adults) -also associated with bicuspid aortic valve, and inherited defects of connective tissue (younger adults) 1. Hypertension >> hyaline arteriolosclerosis of the vasa vasorum >> decreased flow flow >> atrophy of media 2. Marfan Syndrome and Ehlers-Danlos >> weakness of connective tissue in media (cystic medial necrosis) 3. also seen in pregnant women due to increase in plasma volume |
|
|
|
Aortic Dissection Presentation & Complications |
-Presentation: sharp, tearing chest pain that radiates to the back +/- unequal BP in the arms -CXR shows mediastinal widening (wide aortic knob) -Complications: pericardial tamponade due to proximal dissection (most common cause of death), rupture with fatal hemorrhage (into mediastinum), and obstruction of arteries (coronary or renal) branching off the aorta with resultant end-organ ischemia |
|
|
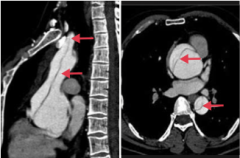
What is this? What kind is it? Where can they be located in general? |
-aortic dissection involving the ascending and descending aorta = Stanford type A -The false lumen can be limited to the ascending aorta, propagate from the ascending aorta, or propagate from the descending aorta |
|
|
|
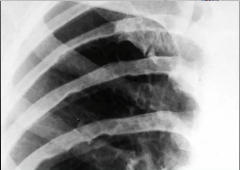
Thoracic Aortic Aneurysm
What is it? Due to? Seen In? Pathogenesis? |
-balloon like dilatation of the thoracic aorta -Due to weakness in the aortic wall -Seen in Tertiary syphilis: obliterative endarteritis of the vasa vasorum >> luminal narrowing, decreased flow, and atrophy of vessel wall >> 'tree bark appearance 'of aorta (due to scarring and fibrosis from endartaritis) -Seen in cystic medial degeneration due to hypertension (older patients) -Seen in Marfan's (younger patients) |
|
|
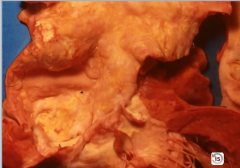
Thoracic Aorta |
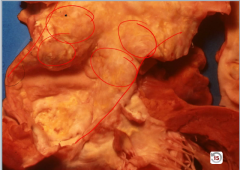
Thoracic Aortic Aneurysm
-caused classically by tertiary syphilis -tree bark appearance of aorta |
|
|
|
Thoracic Aneurysm Complications |
-Major Complication is dilation of the aortic valve root >> aortic insufficiency -Compression of mediastinal structures (airway, esophagus) -Thrombosis due to nonlaminar flow >> can lead to embolism |
|
|
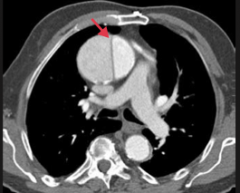
|
Thoracic aortic aneurysm - large aneurysm of ascending aorta with dissection |
|
|
|
Abdominal Aortic Aneurysm
What and where? Commonality |
-balloon-like dilatation of the abdominal aorta that arises below the renal arteries & above the aortic bifurcation -most common site of aortic aneurysm b/c no vaso vasorum |
|
|
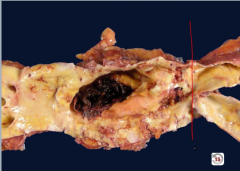
Abdominal Aorta |
Abdominal aortic aneurysm below renal arteries, above bifurcation -notice ballooning |
|
|
|
Abdominal Aortic Aneurysm
Causes/Pathogenesis? Seen In? |
-Primarily due to atherosclerosis -Classically seen in male smokers > 60 yo (> 50 yo in First Aid) with hypertension ---atherosclerosis increases the diffusion barrier to the media >> atrophy and weakness of the vessel wall |
|
|
|
Abdominal Aortic Aneurysm Presentation |
-60 yo male smoker w/ hx of hypertension -presents with pulsatile abdominal mass that grows with time |
|
|
|
Abdominal Aortic Aneurysm Complications |
-Major Complication is rupture, especially when > 5 cm -compression of local structures (ureter) and thrombosis/embolism (disruption of laminar flow) |
|
|
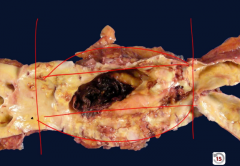
|
Abdominal aortic aneurysm - suprarenal anuerysm with eccentric mural thrombus |
|
|
|
Abdominal Aortic Aneurysm Rupture Presentation |
Triad: hypotension, pulsatile abdominal mass, flank pain (severe left) |
|
|
|
Syphilitic Heart Disease
What does it cause? Findings? Complications |
3° syphilis disrupts the vasa vasorum of the aorta (aortic arch has largest vaso vasorum) with consequent atrophy of the vessel wall (edarteritis obliterans) and dilation of the aorta and valve ring. May see calcification of the aortic root and ascending aortic arch. Leads to “tree bark” appearance of the aorta. Also see head bobbing, water-hammer pulse, pulsating uvula (any thoracic anuerysm >> aortic insufficiency) Can result in aneurysm of the ascending aorta or aortic arch and aortic insufficiency (AV regurg) |
|
|
|
Hemangioma
What is it? Affects who? Involvement? |
-Benign tumor comprised of blood vessels -Commonly present at birth -Often regresses during childhood (so don't surgically remove) -Most often involves skin and liver |
|
|
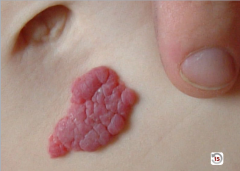
What is it?
DDx? |
Hemanigioma
-could be purpura which is a bleed into the skin: hemangioma blanches with pressure, purpura doesn't |
|
|
|
Strawberry hemangioma |
Benign capillary hemangioma of infancy. Appears in first few weeks of life (1/200 births); grows rapidly and regresses spontaneously at 5–8 years old. |
|
|
|
Cherry hemangioma |
Benign capillary hemangioma of the elderly. Does not regress. Frequency increases with age. |
|
|
|
Angiomyolipoma |
-Kidney hamartoma: composed of blood vessels, muscle, and mature adipose tissue -Associated with tuberous scerlosis |
|
|
|
Cavernous hemangioma |
-Most common benign tumor of the liver and spleen -May rupture if large and produce hemopertineum |
|
|
|
Angiosarcoma
What is it? Prognosis? Sites? Associations? |
-Malignant proliferation of endothelial cells -Common sites: skin (head, neck), breast, liver ---Liver angiocarcinoma associated with polyvinyl chloride (plastics), arsenic, radiation and Thorotrast (thorium dioxide) -Highly aggressive and difficult to resect due to delay in diagnosis
|
|
|
|
Kaposi Sarcoma
What is it? Associated with? |
-Low-grade malignant proliferation of endothelial cells, mainly of the skin, but also seen in mouth, GI tract, and respiratory tract -Associated with HHV-8 |
|
|
|
Kaposi Sarcoma Presentation |
-Presents with purple patches plaques, and nodues(proliferation of endothelial cells NOT blood vessels so doesn't blanch) -may also involved visceral organs |
|
|
|
Kaposi Sarcoma
Seen in & treatment for those populations |
-Older Eastern European males: tumor reminas localized to skin; treatment involves surgical removal -AIDS- tumor spreads early; treatment is antiretroviral agents (boost immune system to kill virus) -Transplant recipients: skin, tumor spreads early; treatment involves decreasing immunosuppression |
|
|
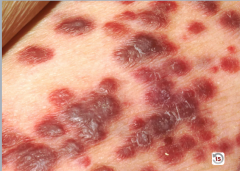
|
Kaposi Sarcoma
Purplish nodules that do NOT blanch (blood isn't in a channel) |
|
|
|
Pyogenic granuloma |
Polypoid capillary hemangioma that can ulcerate and bleed. Associated with trauma and pregnancy. |
|
|
|
Cystic hygroma |
Cavernous lymphangioma of the neck. Associated with Turner syndrome. |
|
|
|
Glomus tumor |
Benign, painful, red-blue tumor under fingernails. Arises from modified smooth muscle cells of glomus body. |
|
|
|
Bacillary angiomatosis |
Benign capillary skin papules found in AIDS patients. Caused by Bartonella henselae infections. Frequently mistaken for Kaposi sarcoma. -use silver stain -has satellite lesions at periphery of main lesion |
|
|
|
Hereditary Telangiectasia |
-AD -Dilated vessels on the skin and mucous membranes in the mouth and throughout the GI tract -Chronic iron deficiency anemia may occur because of bleeding from telangiectasias in the GI tract |
|
|
|
Von Hippel-Lindau Syndrome |
-AD -Cavernous hemangiomas in the cerebellum and retina -Increased incidence of bilateral pheochromocytoma and bilateral renal cell carcinomas |
|
|
|
Spider Telangiectasia |
-Arteriovenous fistula (disappears when the body is compressed) -Associated with hyperestrinism (cirrhosis - can't metabolize 17ketosteroids or estrogen, normal pregnancy) |
|
|
|
Lymphangiosarcoma |
Lymphatic malignancy associated with persistent lymphedema (e.g., post–radical mastectomy). |
|
|
|
Sturge-Weber Syndrome |
-nevus flemmeus (birthmark) on the face in the distribution of the opthalmic branch and/or maxillary branch of CN V -some cases show ipsilateral AV malformation of pia mater vessels overlying the occipital and parietal lobes >> can bleed >> subarachnoid hemorrhage -some pts can show signs of MR |
|
|
|
Raynaud phenomenon
affects what? causes what? types? |
-affects small vessels - decreased blood flow to skin due to arteriolar vasospasm in response to cold temperature or emotional stress. Most often in fingers and toes. Called Raynaud disease when 1° (idiopathic), Raynaud syndrome when 2° to a disease process like mixed connective tissue disease, SLE, or CREST (limited form of systemic sclerosis) syndrome. |
|
|
|
Ischemic Heart Disease
General Epidemiology & Cause/Risk Factors |
-syndromes related to myocarcial ischemia -IHD is the leading cause of death in the US -Usually due to atherosclerosis of coronary arteries, which decreases blood flow to the myocardium -Risk factors similar to those of atherosclerosis: incidence increases with age (biggest risk factor for CAD = age) |
|
|
|
Stable Angina
What is it? Cause? |
-chest pain that arises with exertion or emotional stress that resolves with rest -Due secondarily to atherosclerosis of the coronary arteries with > 70% stenosis >> decreased blood flow can't meet metabolic demands of the myocardium -Represents reversible injury to myocytes (no necrosis, will see cellular swelling) |
|
|
|
Stable Angina
Presentation & Treatment |
-Presents as chest pain -Levine's sign (lasting < 20 min; > 20 min = irreversible injury & cell death) that radiates to the left arm or jaw, diaphoresis, and shortness of breath -EKG shows ST-segment depression due to subendocardial ischemia -Relieved by rest or nitroglycerin |
|
|
|
Unstable Angina
What is it? Cause? |
-chest pain that occurs at rest; increase in frequency and/or intensity of chest pain (stable >> unstable angina) -Usually due to rupture of an atherosclerotic plaque with thrombosis and incomplete occlusion of the coronary artery -Represents reversible injury to myocytes (blood still getting through) |
|
|
|
Unstable Angina
Presentation? Treatment? Complications? |
-EKG shows ST-segment depression due to subendocarcial ischemia -Relieved by nitroglycerin -High risk of progression to MI |
|
|
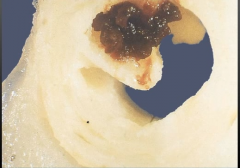
|
-Thrombosis of coronary artery -partial occlusion = unstable angina |
|
|
|
Prinzmetal angina
What is it? Cause? Triggers? |
-episodic chest pain unrelated to exertion -Due secondarily to coronary artery vasospasm (completely clamps down >> ischemia) -Represents reversible injury to myocytes (no necrosis) -Known triggers: cocaine, tobacco, triptans -occurs in the morning more |
|
|
|
Prizmetal angina
Presentation? Treatment? |
-Non-exertional chest pain -EKG shows transient ST-segment elevation due to transmural ischemia -Relieved by nitroglycerin or Ca-channel blockers (or smoking cessation if applicable) |
|
|
|
Coronary Steal Syndrome |
Distal to coronary stenosis, vessels are maximally dilated at baseline. Administration of vasodilators (e.g., dipyridamole, regadenoson) dilates normal vessels and shunts blood toward well-perfused areas >> decreased flow and ischemia in the post-stenotic region. Principle behind pharmacologic stress tests. |
|
|
|
Myocardial infarction
What is it? Cause? |
-Necrosis of cardiac myocytes (>20 min occlusion) -Usually due to rupture of an atherosclerotic plaque with thrombosis and complete occlusion of a coronary artery -Other causes: coronary artery vasospasm (due to Prizmetal angina or cocaine use), emboli, and vasculitis (Kawasaki disease preferentially affects coronary artery) |
|
|
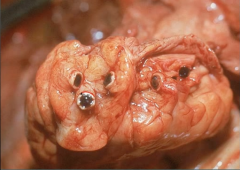
Myocardium |
Myocardium w/ complete occulusion of coronary arteries = MI |
|
|
|
Myocardial Infarction
Presentation |
-Generally: severe crushing chest pain (last > 20 minutes) that radiates to the left arm or jaw, diaphoresis, and dyspnea (due to decreased CO and pulmonary edema), nausea, vomiting, fatigue -LAD occlusion: crushing chest pain, that radiates to jaw & left arm -RCA: bradycardia & atypical chest pain & epigastric pain, simulating GERD -Symptoms NOT relieved by nitroglycerin |
|
|
|
Myocardial Infarction
Locations/Commonality & associated complications? |
-Usually involves the LV; RV & atria generally spared ---Occlusion of LAD >> infarction of anterior wall and anterior septum; most commonly involved artery (45% of cases); complication: complete heart block ---Occlusion of RCA >> infarction of posterior wall, posterior septum, and posterior papillary muscle of the LV, RV; 2nd most commonly involved artery; Complication: rupture of papillary muscle >> insufficiency; AV block ---Occlusion of the left circumflex >> infarction of the lateral wall of the LV |
|
|
|
Myocardial Infarction
Initial involvement & EKG findings during this time? Progession? |
-ECG is gold standard for diagnosis of MI in the first 6 hours -Initial phase of infarction >> subendocardial necrosis (most distal of blocked artery) involving <50% of the myocardial thickness (=subendocardial infarction) ---EKG shows ST-segment depression -Continued/Severe ischemia >> transmural necrosis involving most of the myocardial wall (transmural infarction) ---EKG shows ST-segment elevation ---pathological Q waves indicate an evolving or old transmural infarct |
|
|
|
Myocardial Infarction
Lab values & their timelines? |
-To see if there is irreversible damage to myocytes >> membrane damage >> enzymes in myocytes leak -Troponin I is the most sensitive and specific marker (gold standard) for MI ---Level rises 2-4 hours after infarction, peaks at 24 hours, and returns to normal by 7-10 days -CK-MB useful for detecting reinfarction that occurs days after initial MI ---CK-MB rises 4-6 hours after infarction, peaks at 24 hours, and returns to normal after 72 hours -LDH2 higher than LDH1 usually - see LDH flip in MI to LDH 1 being higher - showes up at 18 hours, peaks at 3 days, lasts for 7 days |
|
|
|
Myocardial Infarction Treatment |
-ASA and/or heparin - limits additional thrombosis [effective in decreasing mortality rate - 20% releative risk reduction if ASA + Clopidogrl] -Supplemental O2 - minimize ischemia -Nitrates - vasodilate coronary arteries and veins -B-blocker - slows HR >> decreasing O2 demand and risk for arrhythmia -ACE inhibitor - decrease LV dilation -Fibrinolysis or angioplasty - opens blocked vessel [STEMI only] -ALWAYS check EF after MI b/c that is biggest prognostic factor |
|
|
|
What occurs after fibrinolysis/angioplasty to treat MI? |
-Contraction band necrosis: Reperfusion of irreversible-damage cells>> Ca influx >> hypercontraction of myofibrils -Reperfusion injury: return of oxygen and inflammatory cells may lead to free radical generation further damaging myocytes ---cardiac enzymes continue to rise after you unblock the vessel |
|
|
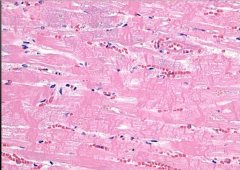
|
-area of dead heart, myocytes w/ no nucleus -Contraction band necrosis |
|
|
|
What do you see with reperfusion injury? |
-Continued rise in cardiac enzymes due to additional myocardial damage/necrosis |
|
|
|
<4 hrs from MI
Gross changes? Microscopic changes ? Complications? |
-Gross changes: none -Microscopic changes: none -Complications: Cardiogenic shock (massive infarction), CHF (seen as decreased EF), arrhythmia, sudden death |
|
|
|
4-12 hours from MI
Gross changes? Microscopic changes ? Complications? |
-Gross changes: dark mottling; pale with tetrazolium stain -Microscopic: early coagulative necrosis (pyknosis, karyohexis, karyolysis), release of necrotic cell contents into blood; edema hemorrhage, wavy fibers -Complications: arrhythmia, HF, cardiogenic shock, death |
|
|
|
12-24 hours from MI
Gross changes? Microscopic changes? Complications? |
-Gross changes: dark discoloration; pale with tetrazolium stain -Microscopic changes: coagulative necrosis (pyknosis, karyohexis, karyolysis) >> neutrophil migration begins; repurfusion injury may cause contraction bands due to free radical damage -Complications: arrhythmia (caused by damage to conducting system), cardiogenic shock, death; less likely to occur > 24 hours |
|
|
|
1-3 days from MI
Gross changes? Microscopic changes? Complications? |
-Gross changes: yellow pallor, hyperemia -Microscopic changes: extensive coagulative necrosis; neutrophils (acute inflammation always follows necrosis) -Complications: Fibrinous pericarditis (inflammatory exudate) - ONLY SEEN WITH TRANSMURAL INFARCTION; presents as chest pain with friciton rub |
|
|
|
4-7 days from MI
Gross changes? Microscopic changes? Complications? |
-Gross change: yellow pallor (hyperemic border) -Microscopic changes: Macrophages -Complications: ---Rupture of ventricular wall (due to macrophage mediate structural degradation) >> cardiac tamponade (most occur with LAD thrombosis) ---Rupture of interventricular septum >> L to R shunt (step up O2 in RV) ---Rupture of posterior medial papillary muscle (w/ occlusion of RCA) >> mitral insufficiency (mitral regurgitation murmur - pansystolic w/ S3, S4) --- LV pseudoaneurysm (mural thrombus “plugs” hole in myocardium>> “time bomb”). |
|
|
|
1-3 weeks from MI
Gross changes? Microscopic changes? Complications? |
-Gross changes: Red border (capillaries) emerges as granulation tissue enters from edge of infarct; hyperemic border w/ central yellow- brown softening - maximally yellow at 10 days -Microscopic changes: Granulation tissue with plump fibroblasts, collagen, and blood vessels (healing) -Complications: none - may still be susceptible to ruptures seen at 4-7 days; |
|
|
|
Months from MI
Gross changes? Microscopic changes? Complications? |
-Gross changes: contracted white scar -Microscopic changes: Fibrosis -Complications: aneurysm (scar = weak wall >> true ventricular aneurysm: outward bulging during contraction, dyskinesia), mural thrombus (wall not moving as well near scar), Dressler syndrome, HF, arrhythmias |
|
|
|
Dressler Syndrome
Cause? Pathogenesis? |
rare: transmural infarction >> inflammation to pericardium >> exposed antigens >> formation of antibodies to pericardium >> autoimmune fibrinous pericarditis 6-8 weeks after infarction |
|
|
|
Mural Thrombus
Caused by? Treatment? |
-almost always from LAD thrombi >> transmural infarction >> injury to endothelium >> platelets stick + muscle not contracting b/c injurred >> stasis >> platelets + venous clot so factors V, VII, and RBCs >> so give aspirin (platelets) and heparin and warfarin for the factors -with anterior infarct, give aspirin + warfarin + heparain - |
|
|
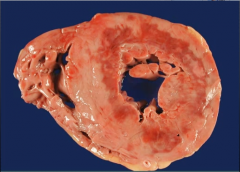
What is it? Time of? |
-subendocardial infarction -dark discoloration of part of myocardium = 4-24 hours after MI |
|
|
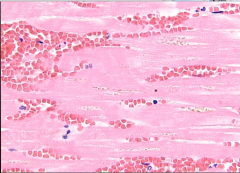
What is it? Timing? |
-Myocytes w/ no nuclei = coagulative necrosis; w/o neutrophils = 4-24 hours |
|
|
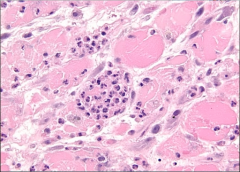
What is it? Timing? |
-MI w/ neutrophils = day 1-3 |
|
|
What is it? Timing? |
-Fibrinous pericarditis = transmural infarct -1-3 days post MI |
|
|
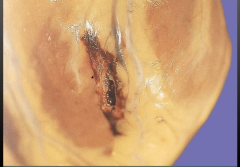
Heart - What is it? Timing? |
-Rupture of ventricular free wall -Happens when macrophages come in day 4-7 |
|
|
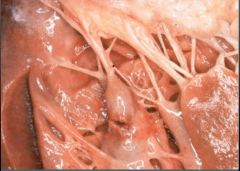
What is it? Timing? |
-Ruptured papillary muscle = occurs with RCA blockage -4-7 days post MI |
|
|
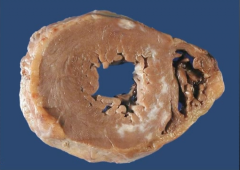
What is it? Timing? |
-Myocardial scar -months after MI |
|
|
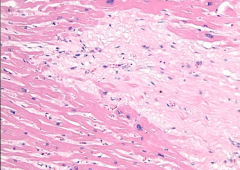
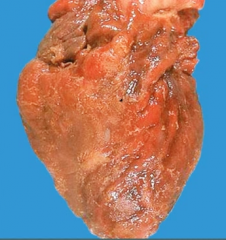
What is it? Timing? Complications? |
-myocardial scar showing dense collagen (type I) -months after MI -Complications: causes weak wall >> aneurysm |
|
|
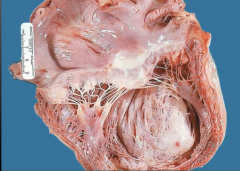
What is it? Timing? Complications? |
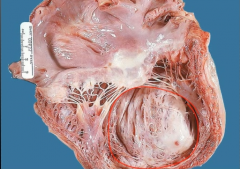
-dilated ventricular aneurysm -months after MI -Could form mural thrombosis along scar -patients die to HF - does NOT rupture |
|
|
|
Sudden cardiac death
What is it? Timing? Death due to?
|
-unexpected death due to cardiac disease -occurs without symptoms or <1 hr after symptoms arise -usually due to fatal ventricular arrhythmia |
|
|
|
Sudden cardiac death
Causes/Etiology |
-most common etiology = acute ischemia -90% of patients have preexisting atherosclerosis; associated with CAD (up to 70% of cases) -less common causes: mitral valve prolapse, cardiomyopathy, cocaine abuse (vasospasm) hereditary ion channelopathies (e.g., long QT syndrome). |
|
|
|
ECG Diagnosis of MI
Anterior Wall - vessel? ECG leads? |
-LAD -Q waves in V1-V4 |
|
|
|
ECG Diagnosis of MI
Anteroseptal Wall - vessel? ECG leads? |
-LAD -Q waves in V1-V2 |
|
|
|
ECG Diagnosis of MI
Anterolateral wall - vessel? ECG leads? |
-LAD or LCX -V4-V6 |
|
|
|
ECG Diagnosis of MI
Lateral wall- vessel? ECG leads? |
-LCX - I, avL |
|
|
|
ECG Diagnosis of MI
Inferior Wall - vessel? ECG leads |
-RCA -II, III, aVF |
|
|
|
MI Complications & timelines |
-Cardiac arrhythmia—important cause of death before reaching hospital; common in first few days. LV failure and pulmonary edema. -Ventricular pseudoaneurysm formation---CO, risk of arrhythmia, embolus from mural thrombus; greatest risk approximately 1 week post-MI. -Ventricular Aneurysm - 3 wks out, pt has systolic bulge of pericardium >> HF |
|
|
|
Chronic Ischemic Heart Disease |
-poor myocardial function due to chronic ischemic damage w/ or w/o infarction -small subendocardial infarctions >> muscle replaced by fibrous tissue >> low ejection fraction >> progresses to CHF -2nd most common indication for heart transplant |
|
|
|
CHF - Symptoms? Signs? |
Clinical syndrome of cardiac pump dysfunction. Symptoms include dyspnea, orthopnea, and fatigue; signs include rales, JVD, and pitting edema |
|
|
|
CHF - systolic vs. diastolic dysfunction |
Systolic dysfunction—low EF, poor contractility, often 2° to ischemic heart disease or DCM.
Diastolic dysfunction—normal EF and contractility, impaired relaxation, decreased compliance. |
|
|
|
CHF Treatment |
ACE inhibitors, β-blockers (except in acute decompensated HF), angiotensin II receptor blockers, and spironolactone decrease mortality. Thiazide or loop diuretics are used mainly for symptomatic relief. Hydralazine with nitrate therapy improves both symptoms and mortality in select patients. |
|
|
|
Left-Side Heart Failure
Causes & Associated Pathogenesis? |
-ischemia: damage >> can't pump properly -hypertension: concentric hypertrophy >> can't oxygenate wall >> ischemia -Dilated cardiomyopathy: 4 chamber dilation >> stretches muscle >> can't contract -Myocardial infarction: myocardium can't function -Restricted cardiomyopathy: can't fill heart appropriately >> therefore, can't pump what's not there |
|
|
|
Left-Sided Heart Failure
Clinical Features & their pathogenesis |
-due to decreased forward progression and pulmonary congestion 1.Pulmonary congestion >> increased pulmonary venous presure >> pulmonary venous distention & transudation of fluid >>pulmonary edema ---Results in dyspnea, paroxysmal nocturnal dyspnea (pillow orthopnea) (due to increased venous return when lying flat), orthopnea, & crackles (fluid in interstium) ---Small, congested capillaries may burst >> intraalveolar hemorrhage >> hemosiderin-laden macrophages (iron in macs) ('heart-failure' cells) >> rusty sputum 2. Decreased flow to kidneys (decreased forward perfusion) >> activation of RAAS >> fluid retention & exacerbation of CHF |
|
|
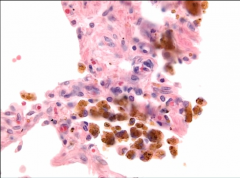
Lung biopsy |
-Hemosiderin-laden macrophages in alveolar air sac seen in left-sided CHF |
|
|
|
Treatment of Left-Sided Heart Failure
Why this? |
-ACE inhibitor b/c it decreases preload and afterload at the same time -ACE inhibitors increase longevity by (1) decreased aldosterone, therefore decreased salt and water reabsorption which decreases preload and (2) by blocking Angiotensin II, will lead to a decrease in vasoconstrictor effect on peripheral resistance arterioles, which will decrease afterload. -add spironolactone w/ aldosterone escape |
|
|
|
Right-Sided Heart Failure
Causes? Most common cause? |
-Most commonly due to left-sided HF -Other causes: left-to-right shunt, chronic lung disease (cor pulmonale) (hypoxia >> vasoconstriction of hypoxic area which is the whole lung) |
|
|
|
Right-Sided Heart Failure
Clinical Features |
Backward failure -JVD due to increased venous pressure -increased central venous pressure >> increased resistance to portal flow >> Painful hepatosplenomegaly with 'nutmeg' liver >> cardiac cirrhosis -Dependent pitting edema due to increased hydrostatic pressure (increased venous presure >> fluid transudation) |
|
|
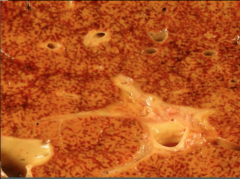
Liver |
Nutmeg liver / cardiac cirrhosis |
|
|
|
Congenital Defects
Arise when & how? Seen in how many live births? & cause what? |
-Arise during embryogenesis (weeks 3-8) -Seen in 1% of live births -Most are sporadic -Results in shunting between left (systemic) and right (pulmonary) circulations |
|
|
|
Left-to-right shunting
Features? Causes & their frequency? Pathogenesis/Complications? |
-can be relatively asymptomatic at birth, but the shunt can eventually reverse >> late cyanosis "blue kids" -Causes: VSD > ASD > PDA 1. increased flow through pulmonary circulation >> hypertrophy of pulmonary vessels and pulmonary hypertension 2. Increased pulmonary resistance results in reversal of shunt >> late cyanosis (Eisenmenger syndrome) with RVH, polycythemia, and clubbing |
|
|
|
Eisenmenger Syndrome |
-Uncorrected left-to-right shunt (VSD, -RVH occurs to compensate >> shunt becomes right to left. -Causes late cyanosis, clubbing, and polycythemia -Age of onset varies |
|
|
|
Right-to-left shunting
Features? Causes? Common Treatment? |
Present with cyanosis shortly after birth - "blue babies" -often diagnosed prenatally, or become evident right after birth. -causes polycythemia Causes: 5 Ts 1. Trucus arteriosus 2. Transposition 3. Tricuspid Atresia 4. Tetralogy of Fallow 5. TAPVR -Treatment: maintenance of PDA and early surgical intervention |
5T's 1 - 5 involvement of vessels all beginning with T |
|
|
Ventricular Septal Defect
What is it? Association? [Mild, Moderate, Severe?] |
-Defect in the septum that divides the R & L ventricles -Most common heart defect -Associated with Fetal Alcohol Syndrome [-mild: Qp/Qs = 1.25-1.5; moderate: Qp/Qs - these 2 are considered restrictive 1.5-2.5; severe: Qp/Qs > 2.5 - nonrestrictive >> can lead to aortic regurgitation] |
|
|
|
Ventricular Septal Defect
Describe the murmur. |
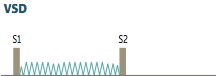
-holosystolic, harsh-sounding murmur -loudest at tricuspid area (left-sternal border) -accentuated with hand-grip due to increased afterload |
|
|
|
Ventricular Septal Defect
Complications? Treatment? |
-Results in left-to-right shunt; may manifest weeks later -Size of defect determines extent of shunting and age at presentation ---small defects asymptomatic, often self-resolve ---large defects >> Eisenmenger syndrome w/ RVH, polycythemia, clubbing (reversal of shunt due to pulmonary hypertension caused by increased volume in pulmonary volume) [RV heave, loud P2] -Treatment: surgical closure; small may close spontaneously [moderately restructive and nonrestrictive should be closed immediately and/or Qp/Qs >2 + evidence of LV volume overload] [if pulm HTN present, close if shunt 1.5/1, pulm reactivity when challenged w/ vasodilation, PA pressure <2/3 systemic BP] |
|
|
|
Atrial Septal Defect
What is it? Types? Most common? Associated with who? |
-Defect in septum that divides the right and left atria; [1 of the 2 most common defects in adults & more common in females 2:1] -Most common type is ostium/septum secundum (90% of cases) [60% according to lecture] -Osteium primum type associated with Down Syndrome and other anomalies [also causes mitral regurge >> pansystolic murmur] -Also associated with Fetal Alcohol Syndrome |
|
|
|
Atrial Septal Defect
Results in? Complications? |
-Results in left-to-right shunt >> loud S1 and a wide fixed split S2 on auscultation (increased blood in right heart delays closure of pulmonary valve) -Complication: paradoxical emboli (DVT normally goes to lung, but b/c ASD, it could cross to left side >> systemic embolus); can also cause HF/ Eisenmenger Syndrome (b/c volume overloading the right side heart >> RV dilation >> presents with SOB [usually happens around 4th decade]) |
|
|
|
ASD
Presentation heart sound wise & progression |
-ASD commonly presents with a pulmonary flow murmur (increased flow through the pulmonary valve) and a diastolic rumble (increased flow across tricuspid valve) -blood flow across the ASD itself doesn't cause a murmur b/c there is not pressure gradient -Murmur later progress to a louder diastolic murmur of pulmonic regurgitation from dilation of the pulmonary artery -loud S1, wide fixed split S2 [can also have palpitations due atrial arrhythmias] |
|
|
|
[How to/ who to treat for ASD] How is an ASD different from a patent foramen ovale? |
-indications for treatment: dilated RV pulmonary artery or mean pressure 50% or < than the corresponding aortic pressures even if asymptomatic; close regardless of age -survival benefit is if the ASD is surgically closed by <25 yo >> reduces risk for RHF, pulmonary HTN, and arrhythmias (pts > 40 yo remain at risk for arrhythmias) -in ASD, septa are missing rather than unfused |
|
|
|
Patent Ductus Arteriosis
Caused by? Associated with? |
-Failure of ductus arteriosus to close -Associated with congenital rubella or prematurity |
|
|
|
Patent Ductus Arteriosis
What does it cause/Pathogenesis? |
-Results in left-to-right shunt between aorta and pulmonary artery >> step up in O2
---During development, ductus arteriosus normally shunts blood form pulmonary artery to aorta, bypassing the lungs >> first breath decreases lung resistance >> closure of PDA -Baby gets differential cyanosis b/c PDA is distal to subclavian - baby is pink up top and blue/cyanotic on the bottom [associated lesions: VSD & coarctation of the aorta] |
|
|
|
Patent Ductus Arteriosis
Clinical Features and why?
Describe the associated murmur. |
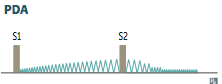
-Asymptomatic at birth with continuous/holosystolic 'machine like' murmur loudest at S2 best heard at left infraclavicular area >> can lead to Eisenmenger syndrome, RVH and/or LVH >> HF - if PAH severe >> PA dilation and pulmonic valve regurge - also get lower extremity cyanosis (b/c PDA arises after major branches of aortic arch) |
|
|
|
Patent Ductus Arteriosis
Treatment? |
-Indomethacin: decreases PGE >> PDA closure ---PGE kEEps PDA open |
|
|
|
Tetralogy of Fallot
Caused by? What 4 things? |
Caused by anteriorsuperior displacement of the infundibular septum Characterized by: 1. Pulmonary infundibular stenosis = stenosis of RV outflow tract (most important determinant of prognosis/cyanosis) 2. RVH 3. Aorta that overrides VSD 4. VSD |
|
|
|
Tetralogy of Fallot
Leads to what? What determines prognosis? |
-right-to-left shunt across VSD >> early cyanosis >> step down >> "tet" spells (HR & CO increase >> venous return increase >> increased R to L shunt >> perpeturation of cycle) -degree of pulmonic stenosis determines extent of shunting and cyanosis -can cause polycythemia |
|
|
|
Tetralogy of Fallot
Clinical Features & Treatment
What congenital heart defects would be good to have with Tetralogy of Fallow |
-most common cause of early childhood cyanosis -Patients learn to squat in response to cyanotic spell to increase arterial resistance and decrease shunting >> more blood can reach lungs >> improves cyanosis -'Boot-shaped' heart on xray -Treatment: surgical correction [long term, can see pulmonary regurge] |
|
|
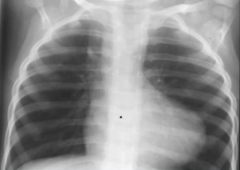
|
-Tetralogy of Fallot -boot shaped heart |
|
|
|
Transposition of the Great Vessels
What is it? Caused by? Associated with? |
-Characterized by pulmonary artery arising from left ventricle and aorta arising from right ventricle >> separation of systemic and pulmonary circulations -Caused by failure of the aorticopulmonary septum to spiral -Associated with maternal diabetes |
|
|
|
Transposition of the Great Vessels
Presentation? Treatment? Complications? |
-Presents with early cyanosis: pulmonary & systemic circuits don't mix = do independent circuits - not compatible w/ life unless shunts present to allow mixing of blood (VSD, PDA, patent foramen ovale)
-Treatment: 1. creation of shunt (allowing blood to mix) after birth is required for survival 2. PGE can be used to maintain PDA until surgical repair >> w/o surgical intervention, infants die within months -Results in RVH and atrophy of left ventricle |
|
|
|
Total anomalous pulmonary venous return (TAPVR) |
Pulmonary veins drain into right heart circulation (SVC, coronary sinus, etc.); associated with ASD and sometimes PDA to allow for right-to-left shunting to maintain CO. |
|
|
|
Truncus Arteriosus
What is it? Caused by? What is also seen in this condition? |
-Characterized by a single large vessel arising from both ventricles ---Truncus fails to divide into the pulmonary trunk and the aorta -most patients have accompanying VSD |
|
|
|
Truncus Arteriosus
Presentation? |
-Presents with early cyanosis: deoxygenated blood from the RV mixes with oxygenated blood from LV before pulmonary and aortic circulations seperate |
|
|
|
Tricuspid Atresia
What is it? Associated with? Presentation? |
-Tricuspid valve orifice fails to develop >> hypoplastic RV -requires ASD and VSD for viability -Often associated with ASD >> R to L shunt -Presents with early cyanosis |
|
|
|
Coarctation of the Aorta
What is it? Generally associated with what? |
-Narrowing of the aorta -associated with bicuspid aortic valve and other heart defects |
|
|
|
Infantile coarctation of the aorta
Associated with what? Where is it? Presentation? Associated with who? |
-Associated with a PDA -Coarctation lies distal to aortic arch but proximal to PDA >> R to L shunt due to low pressure caused by block -Presents as lower extremity cyanosis in infants, often at birth (can present with closure of PDA)(also see different pressures in upper vs. lower extremities) -Associated with Turner Syndrome |
|
|
|
Adult coarctation of the aorta
-Where is it? Presentation? Seen in? Associated with? |
-Coarctation lies distal to aortic arch (distal to ligamentum arteriosum) -Presents as HTN in upper extremities (blood being shunted into early branches of aorta) and hypotension w/ weak pulses in lower extremities (radiofemoral delay) >> claudication of lower extremities, decreased perfusion to kidneys >> activation of RAAS >> HTN -can develop Berry aneurysm at jxn of comm. branches to main cerebral arteries (no elastic lamina there) -Discovered in adulthood -Collateral circulation develops across intercostal arteries >> engorged arteries >> 'notching' of ribs on xray -Associated with bicuspid aortic valve |
|
|
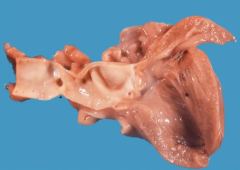
|
-Coarctation of the aorta -notice the narrowing |
|
|
|
-Adult coarctation of the aorta -notching of ribs due to collateral circulations running across ribs |
|
|
|
Acute Rheumatic Fever
What is it? Culprit? Seen in who? When? How does it cause this disease? |
-Systemic complication of pharyngitis due to group A Beta-hemolytic strepotococci |
|
|
|
Acute Rheumatic Fever
Who is it seen in? When does it present? |
Affects children 2-3 weeks after an episode of streptococcal pharyngitis |
|
|
|
Acute Rheumatic Fever
How does the culprit cause systemic disease? |
Caused by molecular mimicry: bacterial M protein resembles human tissue |
|
|
|
Acute Rheumatic Fever
Diagnosis based on? |
Jones Criteria: Evidence of prior group A Beta-hemolytic streptococcal infection (elevated ASO or anti-DNAase B titers) w/ presence of major and minor criteria |
|
|
|
Jones criteria: minor |
nonspecific: include fever and elevated ESR |
|
|
|
Jone criteria: major (describe them) |
1. Migratory polyarthritis - swelling and pain in a large joint (wrist, knees, ankles) that resolves withing days and migrates to involved another large joint 2. Pancarditis - each layer of the heart will be inflamed ---Endocarditis - Mitral valve more commonly involved than aortic valve (mitral > aortic >> tricuspid). Characterized by small vegetations along lines of closure >> regurgitation ---Myocarditis - w/ Aschoff bodies characterized by foci of chronic inflammation, reactive histiocytes w/ slender wavy nuclei (Anitschkow cells), giant cells, and fibrinoid material; MOST COMMON CAUSE OF DEATH DURING ACUTE PHASE ---Pericarditis - >> friction rub and chest pain 3. Subcutaneous nodules 4. Erythema marginatum - annular, nonpruritic rash with erythematous border, commonly involving trunk & limbs |
J - joints O - <3 N - nodules E - erythema marginatum S - Sandringham's? chorea |
|
|
Acute Rheumatic Fever
Prognosis/Progression/Complications |
-Acute attack often resolves, but may progress to chronic rheumatic heart disease (Acute RF >> mitral regurgitation) -Repeat exposure to group A Beta-hemolytic streptococci >> relapse of acute phase and increases risk for chronic disease ---Usually mitral valve, can involve aortic valve: leads to thickening of chordae tendinae & cusps, fusion of commisures >> stenosis w/ "fish mouth" or "button hole" appearance of valve (chronic RF =mitral stenosis) -Complication of damaged valves = future endocarditis |
|
|
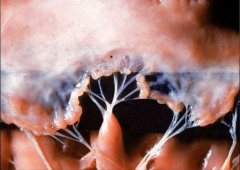
Mitral Valve |
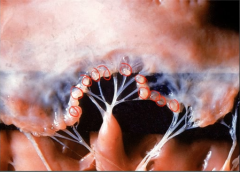
Acute Rheumatic Fever
-Mitral Valve vegetations - endocarditis
|
|
|
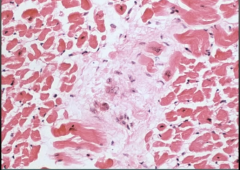
|
Acute Rheumatic Fever
-Aschoff Body: chronic inflammation with giant cells and fibrinoid materal w/ Anitschkow cells (histiocytes with slender wavy nuclei running down middle) -showing myocarditis |
|
|
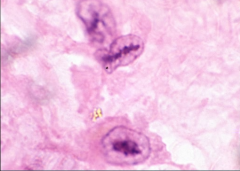
|
Acute Rheumatic Fever
|
|
|
|
Chronic Rheumatic Fever showing fusion of aortic valve commissures = fish mouth appearance |
|
|
|
Aortic Stenosis
Definition? Caused by? See in? |
-Narrowing of aortic valve orifice -Usually due to fibrosis and calcification from wear and tear 1. presents in late adulthood (>60 yo) 2. Bicuspid aortic valve increases risk and hastens disease onset; normal valve has 3 cusps >> fewer cusps >> increased wear and tear on each cusp -Can also arise as a consequence of chronic rheumatic valve disease ---coexisting mitral stenosis and fusion of the aoritc valve comissures distinguis rheumatic disease from wear and tear |
|
|
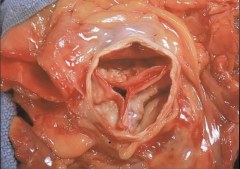
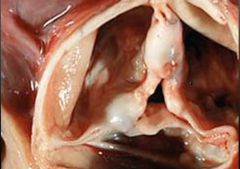
Aortic Valve |
-Aortic Valve Stenosis |
|
|
|
Aortic Stenosis
Initial Presentation? Describe the murmur. |
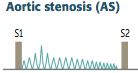
-Cardiac compensation >> prolonged asymptomatic stage during which a systolic ejection click followed by a crescendo-descresendo murmur is heard -LV > aortic pressure during systole -loudest at heart base - 2nd ICS -radiates to carotids - soft S2; would probably hear S4 |
|
|
|
Aortic Stenosis
What changes the intensity of the murmur? |
-intensity increases with expiration, and increased preload of the ventricle -Decreased blood in the ventricle decreases the intensity of the murmur b/c less blood has to move across the valve -differentiates from hypertrophic cardiomyopathy |
|
|
|
Aortic Stenosis
Complications & associated presentations? Treatment? |
-concentric LVH >> can progress to cardiac failure -Angina, dyspnea and syncope w/ exercise - limited ability to increase blood flow across stenotic valve >> decreased perfusion of the myocardium & brain -Microangiopathic hemolytic anemia - RBCs damaged (producing schistocytes) while cross calcified valve -Pulsus parvus et tardus: pulses are weak with a delayed peak (delayed rate of rise in carotid pulse) -Treatment: complications >> valve replacement |
|
|
|
Aortic Regurgitation
Definition? Causes? most common cause? |
-Backflow of blood from the aorta into the LV during diastole -Arises due to: ---Aortic root dilation: most common cause is isolated ARD; pulls on valves, increases space between them >> regurgitation ------can be caused by syphilitic aneurysm and aortic dissection ---bicuspid valve ---Valve damage: caused by infectious endocarditis/rheumatic fever |
|
|
|
Aortic Regurgitation
Clinical Features? Describe the Murmur |
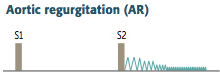
1. High-pitched, early, blowing diastolic decrescendo murmer 2. Hyperdynamic circulation due to increased pulse pressure ---pulse pressure = difference between systolic & diastolic pressures ---Diastolic pressure decreases due to regurgitation, while systolic pressure increases due to increased stroke volume - seen in chronic cases ---presents with bounding pulse (water-hammer pulse), pulsating nail bed (Quincke pulse), head bobbing 3. Results in LV dilation and eccentric (one aspect) hypertrophy (due to volume overload) - so S3 & S4 also |
|
|
|
Aortic Regurgitation
How can the intensity of the murmur be altered? Treatment and how do you know when its absolutely time for treatment? |
-Hand grip increases the intensity of the murmur -expiration -Vasodilators decrease the intensity -Treatment: LV dysfunction >> valve replacement ---Austin flint murmur - anterior leaflet of the mitral valve is dripping blood (on side of outflow tract of the aorta) >> immediate valve replacement |
|
|
|
Mitral Valve Prolapse
Definition? Prevalence? |
-Ballooning of mitral valve into left atrium during systole -seen in 2-3% of US adults - most frequent valvular lesion |
|
|
|
Mitral Valve Prolapse
Causes by/ Etiology? Seen In? |
-Due to myxoid degeneration (accumulation of ground substance - excess of dermatan sulfate) of the valve making it floppy, rheumatic fever, or chordae rupture -Etiology mostly unknown -Seen in Marfan Syndrome and Ehlers-Danlos syndrome |
|
|
|
Mitral Valve Prolapse
Describe the Murmur. What affects its intensity? |
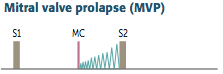
-Presents with incidental mid-systolic click (sudden tensing of chordae tendinae) followed by regurgitation murmur (late systolic crescendo) -best heard over the apex -loudest just before S2 -Usually asymptomatic/benign -Click and murmur become softer with squatting due to increased systemic resistance decreases LV emptying (closer to S2) -murmur/click occurs earlier w/ standing/ valsava maneuver/anxiety due to increase in venous return (S1) |
|
|
|
Mitral Valve Prolapse
Complications? Treatment? |
-Complications are rare but include: infectious endocarditis, arrhythmia, severe mitral regurgitation -Treatment: valve replacement |
|
|
|
Mitral Regurgitation
Definition? |
-Reflux of blood from the LV into the L atrium during systole |
|
|
|
Mitral Regurgitation
Caused by? Seen in? |
-Usually arises as a complication of mitral valve prolapse -Other causes: LV dilatation (left-sided cardiac failure), infective endocarditis (can damage valve leaflets), acute rheumatic heart disease (vegetations on valve; most common one involved), papillary muscle rupture after MI |
|
|
|
Mitral Regurgitation
Clinical Features and Complications? |
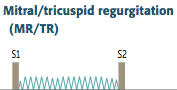
-Holosystolic, high-pitched "blowing" murmur -loudest at apex ---can get louder with squatting (increased systemic resistance decreases LV emptying), hand grip (increased TPR) and expiration (increased return to L atrium) -Results in volume overload and left-sided failure |
|
|
|
Tricuspid Regurgitation
Describe the Murmur. What changes its intensity? Causes? |
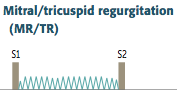
-holosystolic, high-pitched blowing murmur -loudest at tricuspid area and radiates to right sternal border -enhanced by maneuvers that increase RA return (inspiration) -Commonly caused by RV dilation -Also caused by rheumatic fever and infective endocarditis |
|
|
|
Mitral Stenosis
Definition? Caused by? |
-Narrowing of the mitral valve orifice -Usually due to chronic rheumatic valve disease -chronic MS can result in LA dilation |
|
|
|
Mitral Stenosis
Describe the murmur. |
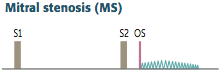
-Opening snap (due to abrupt halt in leaflet motion in diastole, after rapid opening due to fusion at leaflet tips) followed by a late diastolic rumble -decreased interval between S2 and OS correlates w/ an increase in severity -LA > LV during diastole -Enhanced by maneuvers that increase LA return (expiration) |
|
|
|
Difference in Mitral Valve involvement in Acute vs. Chronic Rheumatic Fever |
-Acute: causes mitral regurgitation -Chronic: causes mitral stenosis |
|
|
|
Mitral Stenosis
Clinical Features/Complications? |
-Volume overload >> dilatation of left atrium >>: ---pulmonary congestion w/ edema and alveolar hemorrhage ---pulmonary HTN >> R sided HF ---atrial fibrillation (caused by stretching of atrium >> damage to conducting system) w/ associated risk of mural thrombi -NO ventricular hyerptrophy... it had decreased volume actually |
|
|
|
-Mural Thrombus -Complication of mitral stenosis (from a fib) |
|
|
|
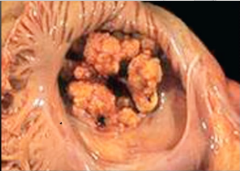
Endocarditis
Definition? Most Common Cause? |
-Inflammation of the endocardium that lines the surface of the cardiac valves -Usually due to bacterial infection -Most common cause: Steptococcus viridans - low virulence organism that infects previously damaged valves (chronic rheumatic heart disease & mitral valve prolapse) - mitral valve most commonly involved |
|
|
|
Streptococcus viridans & Endocarditis Pathogenesis |
-most common cause -low-virulence - infects previously damage valves -Results in small vegetations that do not destroy the valve (subacute endocarditis) 1. Damaged endocardial surface develops thrombotic vegetations (platelets and fibrin) 2. Transient bacteremia >> trapping of bacteria in vegetations -Prophylactic antibiotics decrease risk |
|
|
|
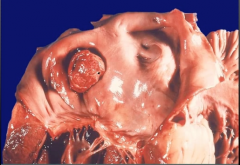
Staphylococcus aureus & Endocarditis
Seen In? What other organisms can you see as the cause of endocarditis in this population? Pathogenesis? |
-most common cause in IV drug abusers - can also see Pseudomonas and Candida -High-virulence organism that infects normal valves, most commonly tricuspid valve -Results in large vegetations on previously normal valves that destroy the valve (acute endocarditis) - rapid onset |
|
|
|
Staphylococcus epidermidis & Endocarditis |
-endocarditis of prosthetic valves |
|
|
|
S. aureus endocarditis -large vegetations sitting on valve >> acute endocarditis |
|
|
|
Streptococcus bovis & Endocarditis |
associated with endocarditis in patients with underlying colorectal carcinoma |
|
|
|
HACEK organisms |
Haemophilus Actinobacillus Cardiobacterium Eikenella Kingella
-associated w/ endocarditis w/ negative blood cultures |
|
|
|
Endocarditis
Clinical Features |
1. Fever - due to bacteremia 2. Murmur (new or worsening)- due to vegetations on valve 3. Septic Embolization of Vegetations (Type III HS -immune complex) ---Janeway lesions - erythematous nontender lesions on palms and soles ---Osler nodes - tender lesions on fingers or toes ---Splinter hemorrhages in nail bed ---Roth spots - white spots on retina surrounded by hemorrhage ---glomerulonephritis can be seen 4. Anemia of chronic disease - due to chronic inflammation |
FROM JANE |
|
|
Endocarditis
Lab Findings |
-Positive blood cultures -Anemia of chronic disease (decrease Hb, decreased MCV, increased ferritin (b/c of hepcidin), decreased TIBC, decreased %saturation) -TEE detects lesions on valves |
|
|
|
Nonbacterial thrombotic endocarditis |
-sterile vegetations that arise in association with hypercoagulable state or underlying adenocarcinoma -vegetations arise on mitral valve along lines of closure >> mitral regurgitaiton |
|
|
|
Libman-Sacks Endocarditis |
-due to sterile vegetations that arise in association with SLE -vegetations present on the surface & undersurface of the mitral valve >> mitral regurgitation |
|
|
|
Complications of Endocarditis |
chordae rupture, glomerulonephritis, suppurative pericarditis, emboli |
|
|
|
Dilated Cardiomyopathy
Definition? Commonality? |
-Dilation of all 4 chambers of the heart -most common form (90% of cases) |
|
|
|
Dilated Cardiomyopathy Complications & Presentation |
-Results in systolic dysfunction (ventricles can't pump) >> biventricular CHF -Complications: Mitral & Tricuspid regurgitation, arrhythmia, eccentric hypertrophy (sarcomeres added in series) -Presentation: heart failure, S3, dilated heart on echocardiogram, balloon appearance of heart on CXR |
|
|
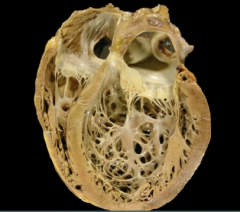
|
-Dilate cardiomyopathy - all 4 chambers massively dilated |
|
|
|
Dilated Cardiomyopathy Causes |
Most commonly idiopathic; others: -Genetic mutations (AD) -Myocarditis - usually due to coxsackie A or B >> characterized by lymphocytic infiltrate of the myocardium >> chest pain, arrhythmia w/ sudden death, or HF (dilated cardiomyopathy late complication if they survive the acute phase) - other virses: influenza, adeno, echo, CMV, HIV -Alcohol Abuse -Drugs - doxorubicin, cocaine -Pregnancy - late pregnancy or weeks to months after childbirth >> develop HF due to dilated cardiomyopathy -Hemochromatosis -Chaga's Disease |
|
|
|
Dilated Cardiomyopathy Treatment |
Na+ restriction, ACE inhibitors, β-blockers, diuretics, digoxin, implantable cardioverter defibrillator (ICD), heart transplant. |
|
|
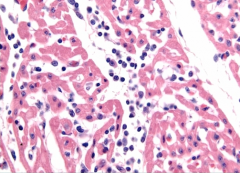
Heart Biopsy |
Myocarditis >> dilated cardiomyopathy -notice the lymphocytic infiltrate |
|
|
|
Hypertrophic Cardiomyopathy
Definition? Cause? |
-Massive hypertorphy of LV, often septa predominance -Usually (60-70% of cases) due to genetic mutations in sarcomere proteins (B-myosin heavy chain mutation); most common AD -rarely associated with Friedrich ataxia |
|
|
|
Hypertrophic Cardiomyopathy
Clinical Features? Biopsy |
1. Decreased cardiac output - LVH >> diastolic dysfunction & S4 2. Sudden death due to ventricular arrhythmias ---hypertrophic cardiomyopathy is a common cause of sudden death in young athletes 3. Syncope with exercise - subaortic hyerptrophy of the ventricular septum >> functional aortic stenosis 4. Biopsy: myofiber hypertrophy with disarray |
|
|
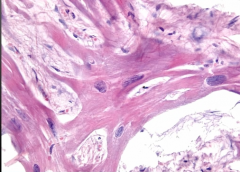
Heart Biopsy |
Hypertorphyic Cardiomyopathy - Disorganization with myocytes in every direction instead of in line -myofibrillar disarray and fibrosis |
|
|
|
Hypertrophic Cardiomyopathy Treatment |
Cessation of high-intensity athletics, use of β-blocker or non-dihydropyridine calcium channel blockers (e.g., verapamil). ICD if patient is high risk. |
|
|
|
Obstructive Hypertrophic Cardiomyopathy |
-hypertrophied septum is too close to the anterior mitral valve leaflet >>outflow obstruction >> dyspnea >> syncope |
|
|
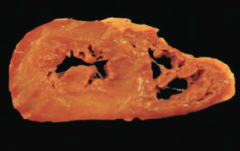
|
Hypertrophic Cardimyopathy -concentric ventricular hypertrophy |
|
|
|
Restrictive Cardiomyopathy
Defintion? |
Decreased compliance of ventricular endomyocardium that restricts filling during diastole |
|
|
|
Restrictive Cardiomyopathy
Causes? |
-Amyloidosis, Sarcoidosis, Hemochromatosis, Endocardial Fibroelastosis (children - have a dense layer of fibrosis and elastic tissue in the endocardium >> can't stretch when heart expands) -Loeffler Syndrome - endomyocardial fibrosis with an eosinophilic infiltrate and eosinophillia >> fibrosis of the endo- and myo- cardium |
|
|
|
Restrictive Cardiomyopathy
Presentation |
-dyastolic dysfunction >> Presents as CHF -Classic finding = EKG w/ low QRS amplitude despite thick myocardium (especially with amyloid) |
|
|
|
Myxoma
Definition? Seen in? Where? Complication? |
-Benign mesenchymal tumor w/ gelatinous appearance and abundant ground substance on histology -most common primary cardiac tumor in adults -Usually forms a pedunculated mass in the left atrium (ball valve obstruction of the atrium) that causes multiple syncopal episodes due to obstruction of mitral valve -Can also embolize b/c soft >> fever |
|
|
|
Rhabdomyoma |
-Benign hamartoma of cardiac muscle -Most common primary cardiac tumor in children ---associated with tuberous sclerosis -Usually arises in the ventricle |
|
|
|
Metastasis to Heart
From where? Involves what part of the heart? Causes what? |
-Metastatic tumors are more common in the heart than primary tumors ----common metastases to the heart: breast & lung carcinoma, melanoma, and lymphoma -Most commonly involves pericardium >> pericardial effusion |
|
|
|
Heart embryology
Truncus arteriosus |
Ascending Aorta & pulmonary trunk |
|
|
|
Heart embryology
Bulbous cordis |
Smooth parts (outflow tract) of the left and right ventricles |
|
|
|
Heart embryology
Primitive Atria |
Trabeculated part of the left and right atria |
|
|
|
Heart embryology
Primitive Ventricles |
Trabeculated part of the left and right ventricles |
|
|
|
Heart embryology
Primitive pulmonary vein |
smooth part of the left atrium |
|
|
|
Heart Embryology
Left horn of the sinus venosus |
Coronary Artery |
|
|
|
Heart Embryology
Right horn of the sinus venosus |
smooth part of the right atrium |
|
|
|
Heart Embryology
right common cardinal vein and right anterior cardinal vein |
SVC |
|
|
|
What is the first organ to form in embryo? |
The heart is the first functional organ in vertebrate embryos |
|
|
|
When does the heart start beating? |
week 4 |
|
|
|
Heart Embryology
Cardiac Looping
What is it? When does it start? |
-primary heart tube loops to establish left-right polarity -begins in week 4 of gestation |
|
|
|
Kartagener Syndrome
What is it? Complications? |
-It is a defect in dynein >> primary ciliary dyskinesia -In terms of cardiac embryology, it is a defect in L-R dynein which affects L-R symmetry >> dextrocardia -It can also affect the lungs mimicing CF, and cause infertility |
|
|
|
Heart Embryology
Septation of the Atria |
1. Septum primum grows towards the endocardial cushions, narrowing the foraman primum 2. Foramen secundum forms in septum primum; foraman primum disappears 3. Septum secundum develops foraman secundum maintaining the L-to-R shunt 4. Septum secundum expands to cover most for foraemen secundum forming residual foramen ovale 5. Remaining septum primum forms valve of foramen ovale 6. Septum primum & septum secundum fuse to form the atrial wall 7. Foramen ovale usually closes soon after birth due to increase in LA pressure |
|
|
|
Patent Foramen Ovale
What is it? Treatment? Complications? |
-caused by failure of septum secundum and septum primum to fuse after birth -most are left untreated -can cause paradoxical emboli |
|
|
|
Heart Embryology
Vetrincular Formation |
1. Muscular ventricular septum forms; Opening is the interventricular foramen 2. Aorticopulmonary septum rotates and fuses with muscular ventricular septum to form the muscular interventricular septum, closing the interventricular foramen 3. Growth of the endocardial cushions separetes atria from the ventricles and contributes to atrial septation and membranous portion of interventricular septum |
|
|
|
Ventricular Septal Defect |
-Most commonly occurs in the membranous septum -acyanotic at birth due to left-to-right shunt |
|
|
|
Heart embryology
Outflow tract formation |
-truncus arteriosus rotates ---neural crest & endocardial cell migrations >> truncal and bulbar ridges that spiral and fuse to form the aorticopulmonary septum >> ascending aorta and pulmonary trunk |
|
|
|
Heart embryology
Conotruncal abnormalities |
-Transposition of the great vessels -Tetralogy of Fallot -Persistent truncus arteriosus |
|
|
|
Heart Embryology
Valve development |
-Aortic/pulmonary: derived from the endocardial cushions of the outflow tract -Mitral/tricuspid: derived form the fused endocardiacl cushions of the AV canal |
|
|
|
Heart Embryology
Valve Abnormalities |
stenotic, regurgitant, atretic (tricuspid atresia), displaced (Ebstein anomaly) |
|
|
|
Fetal erythropoiesis |
-Yolk sac (3-8 weeks) -Liver (6 weeks - birth) -Spleen (10-28 weeks) -Bone marrow (18 weeks to adult) |
Young Liver Synthesizes Blood |
|
|
Fetal hemoglobin development
What is special about HbF |
-Fetal Hemoglobin (HbF) = alpha2gamma2 ---HbF has higher affinity for O2 b/c 2,3-BPG can't bind >> right curve shift, facilitating unloading of O2 form HbA across the placenta -Adult Hemoglobin (HbA) = alpha2beta2 |
Alpha always; gamma goes, becomes beta |
|
|
Fetal circulation
vein vs artery O2 saturation |
-Blood in umbilical vein has PO2 of 30 mmHg and is 80% saturated with O2 -Umbilical arteries have low O2 saturation |
|
|
|
Fetal circulation
Shunts |
1. Blood entering the fetus through the umbilical vein is conducted via the ductus venus into the IVC to bypass hepatic circulation 2. Most highly oxygenated blood reaching the heart via the IVC is diverted through the foramen ovale and pumped out the aorta to the head and body 3. Deoxygenated blood entering the RA from the SVC goes: RA >> RV >> main PA >> patent ductus arteriosus >> descending aorta due to high fetal pulmonary artery resistance and low O2 tension |
|
|
|
What happens in fetal circulation when the first breath is taken? |
Breath >>: 1. decreased resistance in pulmonary vasculature >> increased LA pressure vs. RA pressure >> closure of foramen ovale (now called fossa ovalis) 2. increased O2 (from respiration) and decreased protaglandins (from placental separation - PGE2 comes from placenta) >> closes ductus arteriosus >> DA remant = ligamentum arteriosum |
|
|
|
What keeps the PDA open?
What closes it? |
-PGE1 and 2 keep it open -Indomethacin can help close |
|
|
|
Follow fetal circulation from placenta through fetus |
1. oxygenated blood from placenta >> umbilical vein >> ductus venosus >> IVC >> RA >> foramen ovale >> LA >> aorta to brain & down descending also (most oxygenated blood to brain) >> descending aorta >> umbilca arteries >> placenta 2. deoxygenated blood from SVC >> RA >> SV >> pulmonary trunk >> through PDA >> descending aorta |
|
|
|
Fetal-postnatal derivatives
Umbilical Vein |
Ligamentum teres hepatis - contained in falciform ligament |
|
|
|
Fetal-postnatal derivatives
Umbilical artery |
Medial umbilical ligaments |
|
|
|
Fetal-postnatal derivatives
Ductus arteriosus |
Ligamentum arteriosum |
|
|
|
Fetal-postnatal derivatives
Ductus venosus |
Ligamentum venosum |
|
|
|
Fetal-postnatal derivatives
Foramen ovale |
Fossa ovalis |
|
|
|
Fetal-postnatal derivatives
Allantois |
Urachus - median umbilical ligament ---urachus is part of the allantoic duct between the bladder and the umbilicus ---Urachal cyst/sinus is a remnant |
|
|
|
Fetal-postnatal derivatives
Notochord |
Nucleus pulpous of intervertebral disc |
|
|
|
Right coronary artery
supplies what? infarction leads to? |
-supplies SA & AV nodes -infarct can cause nodal dysfunction (bradycardia or heart block) -infarct affects posterior wall, posterior septum, and posterior papillary muscles of the LV (via PDA) (can lead to rupture and valvular insufficiency) |
|
|
|
Acute marginal artery
Comes off what? Supplies what? |
-from RCA -supplies right ventricle |
|
|
|
What does right-dominant circulation mean? What is its prevalence? |
-PDA arises from RCA -85% of the population |
|
|
|
What does left-dominant circulation mean? What is its prevalence? |
-PDA arises from LCX -8% of the population |
|
|
|
What does co-dominant circulation mean? Prevalence? |
-PDA arises from RCA & LCX -7% of the population |
|
|
|
Most common sight of coronary artery occlusion |
LAD |
|
|
|
Coronary blood flow peaks when? |
in early diastole |
|
|
|
Left Circumflex artery supplies what? |
lateral and posterior walls of the left ventricle |
|
|
|
Left Anterior Descending Artery supplies what? |
supplies anterior 2/3 of the interventricular septum, anterior papillary muscle, and anterior surface of the left ventricle |
|
|
|
Left marginal artery arises from? |
LDA |
|
|
|
Posterior descending artery supplies what? |
posterior 1/3 of the interventricular septum and posterior walls of the ventricles |
|
|
|
What is the most posterior part of the heart? What can enlargement cause? |
-left atrium is the most posterior portion of the heart -enlargement can cause dysphagia (compression of esophagus) or hoarseness (compression of recurrent laryngeal, a branch of the vagus) |
|
|
|
Cardiac Output (equation) |
=stroke volume x heart rate |
|
|
|
Stroke volume equation |
= End-diastolic volume - end-systolic volume
proportional to pulse pressure |
|
|
|
Ejection Fraction equation |
= stroke volume/ end-diastolic volume |
|
|
|
Fick Principle |
CO = rate of O2 consumption / (arterial O2 content - venous O2 content) |
|
|
|
Mean arterial pressure equation |
MAP = CO x TPR
MAP = 2/3 diastolic pressure + 1/3 systolic pressure |
|
|
|
Pulse pressure |
= systolic pressure - diastolic pressure
proportional to stroke volume & inversely proportional to arterial compliance (C=v/p) |
|
|
|
Describe exercise in terms of CO, HR, and SV |
-Early stages of exercise: CO maintained by increased HR and increased SV -Late stages: CO maintained by increased HR only; SV plateaus |
|
|
|
What happens to diastole with increased HR? Consequences? |
-increased HR >> shortened diastole >> less filling time >> decreased cardiac output (ventricular tachycardia) |
|
|
|
What causes an increased pulse pressure? |
hyperthyroidism, aortic regurgitation, arteriosclerosis, obstructive sleep apnea (increased sympathetic tone), exercise (transient) |
|
|
|
What causes a decreased pulse pressure? |
aortic stenosis, cardiogenic shock, cardiac tamponade, advance heart failure |
|
|
|
What affects stroke volume? |
-SV affected by contractility, afterload, and preload -SV increases with an increase in contractility, an increase in preload, and/or a decrease in afterload |
SV CAP |
|
|
What causes an increase in contractility? |
-also causes an increase in SV -Catecholamines (increase activity of Ca2+ pump in SR) -increased intracellular Ca2+ -decreased extracellular Na+ (decreases activity of Na+/Ca2+ exchanger) -Digitalis - blocks Na+/Ca2+ pump |
|
|
|
What causes an changes in stroke volume? |
-Increase in SV: anxiety, pregnancy -Decrease in SV: heart failure |
|
|
|
Myocardial O2 Demand |
-increase in afterload -increase in contractility -increase in HR -increase in ventricular diameter (increases wall tension) |
|
|
|
What causes a decrease in contractility? |
-results in decreased SV -B1-blockade (decreases cAMP) -heart failure w/ systolic dysfunction -acidosis -hypoxia/hypercapnia (decreases PO2/PCO2) -non-dihydropyradine Ca2+ channel blockers |
|
|
|
How is preload approximated? What affects it? |
-Preload is approximated by ventricular EDV -Depends on venous tone and circulating blood volume ---thus venodilators (nitroglycerine) decrease preload |
|
|
|
How is afterload approximated? Equation associated? What affects it? |
-afterload approximated by MAP -Relation of LV size and afterload: ---Laplace's Law: Wall tension = (pressure x radius) / (2 x wall thickness) ---LV hypertrophies in response to increased afterload -vasodilators (hydralazine) decrease afterload (arterial) -ACE inhibitors & ARBs decrease preload and afterload -Chronic HTN (increased MAP) >> LV Hypertrophy |
|
|
|
How does the ventricle respond to increased afterload? Equation? |
-LV compensates for increased afterload by thickening (hypertrophy) to decrease wall tension -Laplace's Law: Wall tension = (pressure x radius) / (2 x wall thickness) |
|
|
|
Ejection Fraction
Equation? Normal? What happens to it in heart failure? |
EF = SV/EDV = (EDV - ESV) / EDV
-left ventricular EF is an index of ventricular contractility -normal ≥ 55% -Systolic HF - EF decreases -Diastolic HF - EF normal |
|
|
|
Force of contraction is proportional to... |
-end-diastolic length of cardiac muscle fiber (pre-load) |
|
|
|
What causes increase in contractility? |
catechoalmines (exercise), digoxin |
|
|
|
What causes a decrease in contractility? |
loss of myocardium (MI), B-blockers, Ca-channel blockers, dilated cardiomyopathy |
|
|
|
Starling curve
What does it describe? Relationships? |
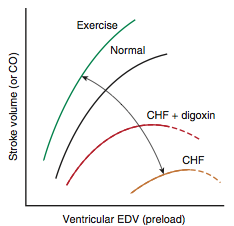
-describes increases in CO or SV in response to an increase in venous return or end-diastolic volume -increases in contractility increase CO for any level of end-diastolic volume |
|
|
|
Blood flow can be expressed by which equation? Describe the components of this equation. |
Q = change in pressure/ resistance CP = MAP - RA pressure / TPR -analagous to Ohm's law -pressure gradient drives flow from high to low pressure -Resistance is directly proportional to viscosity and vessel length and inversely proportional to radius to the 4th power -Arterioles account for most of TPR as they are the site for highest resistance >> they regulate capillary flow |
|
|
|
Equation for Resistance |
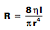
|
|
|
|
Systemic Circulation exhibits what type of resistance? |
-Parallel resistance - exhibited by each organ system 1/Rtotal = 1/R1 + 1/R2 -total resistance less than any individual artery -each time you add an artery to the system, you decrease total resistance |
|
|
|
Circulation within an organ exhibits what type of resistance? |
-Series resistance - exhibited by arrangement of vessels withing a given organ Rtotal = Rarteries+ Rarterioles + R3caps -arterioles contribute largest proportion of resistance -pressure decreases as blood flows through a series |
|
|
|
What causes changes in viscosity? |
-depends on mostly on Hct -increased by: polycythemia, hyperproteinemic state (multiple myeloma), aHereditary spherocytosis -decreases in anemia |
|
|
|
Inotropy & Cardiac/Vascular Function Curve
What affects inotropy? |
-inotropy = change in contractility >> altered CO for a given RA pressure (preload)
1.Increased by: catechoalmines, digoxin 2.Decreased by: uncompensated HF, narcotics overdose |
|
|
|
Inotropy & Cardiac/Vascular Function Curve
What affects inotropy? |
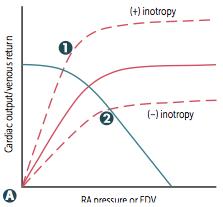
-inotropy = change in contractility >> altered CO for a given RA pressure (preload) 1.Increased by: catechoalmines, digoxin 2.Decreased by: uncompensated HF, narcotics overdose |
|
|
|
Venous Return & Cardiac/Vascular Function Curve
What affects venous return? |
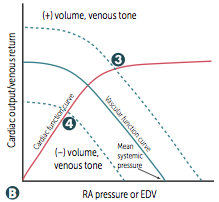
-Changes in circulating volume or venous tone >> altered RA pressure for a given CO -MSP (x-intercept) changes w/ volume & venous tone 3. venous return increased by: fluid infusion, sympathetic activity 4. decreased by: acute hemorrhage, spinal anesthesia |
|
|
|
Total Peripheral Resistance & Cardiac/Vascular Function Cruve
What affects TPR? |
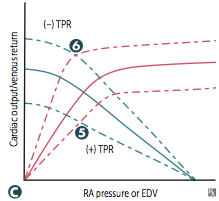
-changes in TPR >> altered CO for a given RA pressure, but MSP is unchanged 5. TPR increased by: vasopressors 6. TPR decreased by: exercise, AV shunt |
|
|
|
Exercise effects on inotropy, TPR, CO |
Exercise >> increased inotropy and decreased TPR to maximize CO |
|
|
|
HF effects on inotropy, TPR, CO |
HF >> decreased inotropy >> fluid retention to increase preload and maintain CO |
|
|
|
Pressure volume loops and cardiac cycle
Normal cycle/loop? When do you hear heart sounds? |
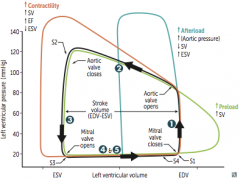
1. isovolmetric contraction - period between mitral valve closing and aortic valve opening; highest O2 consumption - S1 at beginning 2. Systolic ejection - period between aortic valve opening and closing - S2 at end 3. Isovolumetric relaxation - period between aortic valve closing and mitral valve opening 4. Rapid filling - mitral valve opens - S3 at beginning 5. Reduced filling - just before mitral valve closes - S4 at end |
|
|
|
Pressure volume loops and cardiac cycle
What does increased contractility do to the loop? |
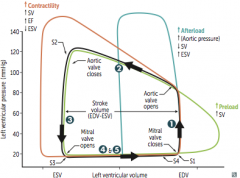
- increased SV >> larger steeper part 2 >> increased EF & decreased ESV |
|
|
|
Pressure volume loops and cardiac cycle
What does increased afterload due to the loop? |
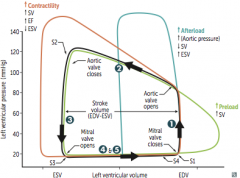
= increased aortic pressure >> decreased SV and increased ESV |
|
|
|
Pressure volume loops and cardiac cycle
What does increased preload due to the loop? |
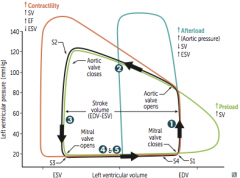
= increased venous return >> increased SV |
|
|
|
S1 heart sound
What is it? Loudest where? What else is happening at this time?
|
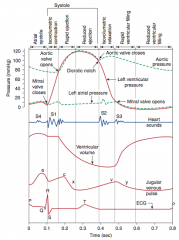
-mitral & tricuspid valve closure -loudest in mitral area -Concurrent events: isovolumetric contraction (end of QRS)
|
|
|
|
S2 heart sound
What is it? Loudest where? What happened between S1 & S2? What else is happening at this time? |
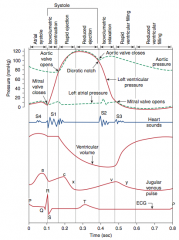
-aortic and pulmonary valve closure >> dicrotic notch -loudest at left sternal border -Between S1 & S2: LV pressure rose & peaked, ventricles repolarized (T wave) -Concurrent events: LV pressure & volume decreased, isovolumetric relaxation |
|
|
|
S3 heart sound
What is it? Normal? What else is happening at this time? |
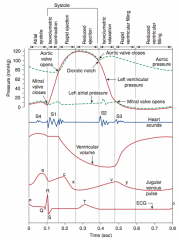
-in early diastole during rapid ventricular filling -associated with increased filling pressures (CHF, mitral regurge) -more common in dilated ventricles (volume overload) -normal in children & pregnant women |
|
|
|
S4 heart sound
What is it? Normal? What else is happening at this time? Associated with? |
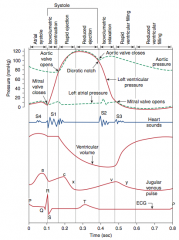
-S4 = atrial kick in late diastole -high atrial pressure -associated with ventricular hypertrophy >> LA must push against stiff LV wall (decreased compliance) -associated with HTN -can also occur with volume overload |
|
|
|
Jugular venous pulse (describe points on the wave) |
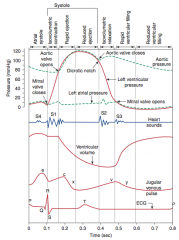
-a wave = atrial contraction -c wave = RV contraction (closed tricuspid valve bulging into atrium) -x descent = atrial relaxation and downward displacement of tricuspid valve during ventricular contraction; absent in tricuspid regurgitation -v wave = increased atrial pressure due to filling against closed tricuspid valve -y descent = blood flow from RA to RV
|
|
|
|
Normal splitting of heart sounds |
-inspiration >> decreased thoracic pressure >> increased venous return to RV >> increased SV >> increased ejection time >> delayed closure of pulmonic valve -decreased pulmonary impedance (increased capacity of pulmonary circulation) also occurs during inspiration, also contributing to delayed closure |
|
|
|
Wide splitting of heart sounds |
-seen in conditions that delay RV emptying (pulmonic stenosis, RBBB) -delayed RV emptying >> delay in pulmonic sound regardless of breath =exaggeration of normal splitting |
|
|
|
Fixed splitting of heart sounds |
-ASD >> L to R shunt >> increases RA volumes >> increases RV volumes >> increased flow through pulmonic valve so that regardless of breath, closure of pulmonic valve delayed |
|
|
|
Paradoxical splitting |
-seen in conditions that delay LV emptying (aortic stenosis, LBBB) -P2 occurs before A2 >> inspiration >> P2 moves closer to A2 paradoxically eliminating the split |
|
|
|
Auscultation of Heart
Aortic Area: what will you hear here? |
Systolic murmur: aortic stenosis, flow murmur, aortic valve sclerosis |
|
|
|
Auscultation of Heart
Pulmonic Area: what will you hear here? |
Systolic Ejection murmur: pulmonic stenosis, flow murmur (physiologic murmur - ASD from increased flow) |
|
|
|
Auscultation of Heart
Left Sternal border: What will you hear here? |
-Diastolic murmur: aortic regurgitation, pulmonic regurgitation -Systolic murmur: hypertrophic cardiomyopathy |
|
|
|
Auscultation of Heart
Tricuspid Area: What will you hear here? |
-Pansystolic murmur: tricuspid regurgitation, VSD -Diastolic murmur: tricuspid stenosis, ASD (rumble due to increased flow) |
|
|
|
Auscultation of Heart
Mitral Area: What will you hear here? |
-Systolic murmur: mitral regurgitation -Diastolic murmu: mitral stenosis |
|
|
|
Systolic murmurs |
-aortic/pulmonic stenosis -mitral/tricuspid regurgitation -VSD |
|
|
|
Diastolic murmurs |
-aortic/pulmonic regurgitation -mitral/tricuspid stenosis |
|
|
|
Bedside Maneuver: Heart Sounds
Inspiration |
-increases intensity of right heart sounds |
|
|
|
Bedside Maneuver: Heart Sounds
Hand grip |
-causes increased systemic vascular resistance -increased intensity of MR, AR, and VSD murmurs -decreased intensity of AS, hypertrophic cardiomyopathy murmurs -MVP: increased murmur intensity, later onset of click/murmur |
|
|
|
Bedside Maneuver: Heart Sounds
Valsalva (phase II), standing |
-decreases venous return -decreased intensity of most murmurs (including AS) -increased intensity of hypertrophic cardiomyopathy murmurs -MVP: decreased murmur intensity, earlier onset of click/murmur
|
|
|
|
Bedside Maneuver: Heart Sounds
Rapid Squatting |
-increased venous return, increase preload, increased afterload with prolonged squating -decreased intensity of hypertrophic cardiomyopathy murmurs -increased intensity of AS murmur -MVP: increased intensity of murmur, later onset of click/murmur |
|
|
|
Ventricular (Bundle of His, Purkinje Fibers) Action Potential Phase 0 |
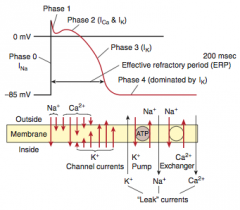
Phase 0 = rapid upstroke and depolarization; voltage gated Na+ channels open (increase in conductance) |
|
|
|
Ventricular (Bundle of His, Purkinje Fibers) Action Potential Phase 1 |
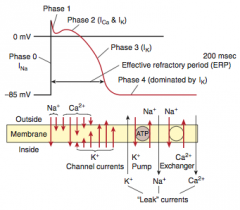
Phase 1 = initial repolarization - decreased conductance/ inactivation of VG Na+ channels & VG K+ channels begin to open (increase in conductance) |
|
|
|
Ventricular (Bundle of His, Purkinje Fibers) Action Potential Phase 2 |
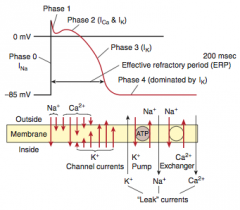
Phase 2 = plateau - Ca2+ influx through VG Ca2+ channels balance K+ efflux; Ca2+ influx triggers release of Ca2+ from SR >> myocyte contraction |
|
|
|
Ventricular (Bundle of His, Purkinje Fibers) Action Potential Phase 3 |
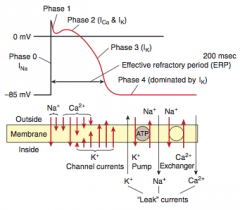
Phase 3 = rapid repolarization - massive K+ efflux due to opening of VG slow K+ channels and closing of VG Ca2+ channels |
|
|
|
Ventricular (Bundle of His, Purkinje Fibers) Action Potential Phase 4 |
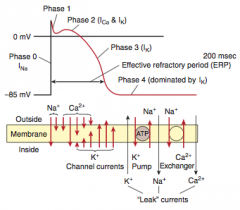
Phase 4 = resting membrane potential - high K+ permeability through K+ channels; about -85mV (close to K+ equilibrium potential) |
|
|
|
Contrast cardiac action potentials to skeletal muscle action potentials |
In contrast to skeletal muscle: -cardiac muscle AP has a plateau due to Ca2+ influx and K+ efflux; myocyte contraction is due to Ca-induced-Ca release from the SR -cardiac nodal cells spontaneously depolarize during diastole resulting in automaticity of the If channels (slow mixed Na+/K+ inward current) -cardiac myocytes are electrically coupled to each other by gap junctions |
|
|
|
Pacemaker Action Potential Phase 0
|
Phase 0 = upstroke of AP - opening of VG Ca2+ channels; Fast VG Na+ channels are permanently inactivated b/c of the less negative resting voltage of these cells
-results in slow conduction velocity that is used by the AV node to prolong transmission from the atria to the ventricles |
|
|
|
Pacemaker Action Potential Phase 0 |
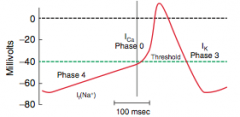
Phase 0 = upstroke of AP - opening of VG Ca2+ channels; Fast VG Na+ channels are permanently inactivated b/c of the less negative resting voltage of these cells -results in slow conduction velocity that is used by the AV node to prolong transmission from the atria to the ventricles |
|
|
|
Pacemaker Action Potential Phase 3 |
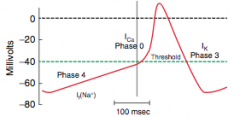
Phase 3 = inactivation of Ca2+ channels and increased activation of K+ channels >> efflux of K+ |
|
|
|
Pacemaker Action Potential Phase 4
What affects this phase? |
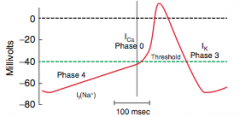
Phase 4 = slow diastolic depolarization; membrane potential spontaneously depolarizes as Na+ conductance increases (If different from INa in phase 0 of ventricular AP) -accounts for automaticity of SA and AV nodes -slope of phase 4 determines HR -Ach/adenosine decrease rate of depolarization and thus decrease HR; Catecholamines increase rate of depolarization increasing HR -Sympathetic stimulation increases chance that If is open >> increased HR |
|
|
|
ECG
P wave |
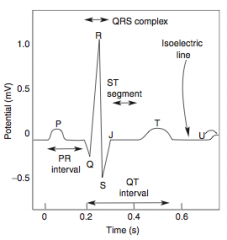
-atrial depolarization; repolarization is hidden by QRS |
|
|
|
ECG
PR interval |
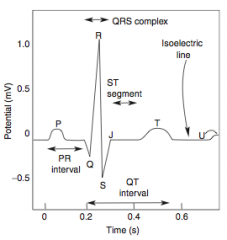
-conduction delay through the AV node >> allows ventricular filling (100 msec delay) -normally <200 msec |
|
|
|
ECG
QRS complex |
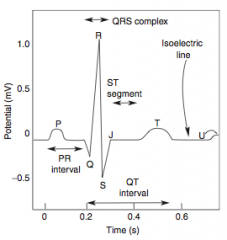
-ventricular depolarization -normally <120 msec |
|
|
|
ECG
QT interval |
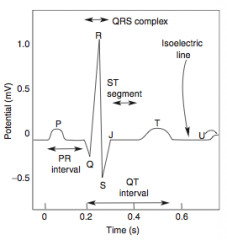
-mechanical contraction of the ventricles |
|
|
|
ECG
T wave |
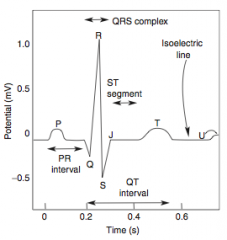
-ventricular repolarization -inversion may indicate MI |
|
|
|
ECG
ST segment |
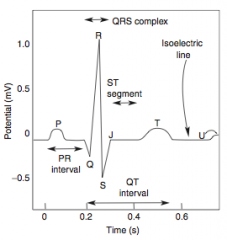
-isoelectric, ventricles depolarized |
|
|
|
ECG
U wave |
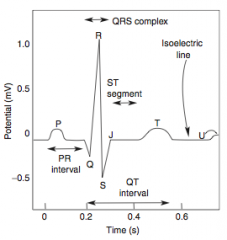
-caused by hypokalemia and bradycardia |
|
|
|
Speed of conduction |
Purkinje > atria > ventricles > AV node |
|
|
|
Pacemakers of the heart
Which is dominant? |
SA > AV > bundle of His / Purkinje fibers / ventricles
SA node is the pacemaker of the heart has inherent dominance with slow phase of upstroke |
|
|
|
Conduction through the heart |
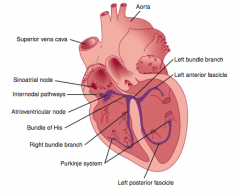
SA node >> atria >> AV node >> common bundle >> bundle branches >> Purkinje fibers >> ventricles |
|
|
|
Torsades de pointes
What is it? Complication? |
-polumorphic ventricular tachycardia characterized by sinusoidal wave forms on ECG -can progress to ventricular fibrillation |
|
|
|
What Causes Torsades de pointes? |
-long QT predisposes to it -Caused by drugs, decreased K+, decreased Mg2+, others -Drugs: Sotalol, Risperidone (antipsychotics), Macrolides, Chloroquine, Protease Inhibitors (-navir), Quinidine (class Ia, class III), Thiazides |
Some Risky Meds Can Prolong QT |
|
|
Congenital long QT syndrome |
-Inherited disorder of myocardial repolarization, typically due to ion channel defects -Increased risk of sudden cardiac death due to torsades de pointes -Includes; Romano-Ward Syndrome & Jervell and Lang-Neilsen Syndrome |
|
|
|
Romano-Ward Syndrome |
-autosomal dominant, pure cardiac phenotype >> congenital long QT syndrome |
|
|
|
Jervell and Lange-Nielsen Syndrome |
-autosomal recessive congenital long QT syndrome that also causes sensorineural deafness |
|
|
|
Wolff-Parkinson-White syndrome
What is it? Pathogenesis & Presentation? Complications? |
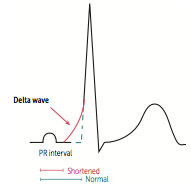
-most common type of ventricular pre-excitation syndrome -abnormal fast accessory conduction pathway form atria to ventricle (bundle of Kent) bypasses the rate-slowing AV node >> ventricles begin to partially depolarize earlier >> characteristic delta wave w/ shortened PR interval on ECG -May lead to reentry circuit >> supraventricular tachycardia |
|
|
|
Atrial Fibrillation |
|
|
|
Atrial fibrillation
What is it? Complications |
-chaotic and erratic baseline (irregular irregular) with no discrete P waves in between iregularly spaced QRS complexes -can lead to atria stasis >> thromboembolic stroke |
|
|
|
Atrial fibrillation
Treatment? |
-rate control -anticoagulation -pharmacologic or electrical cardioversion ---glycosides (digoxin) ---Na+ channel blockers (Quinidine, Procainamide, Disopyramide) ---B-blockers ---K+ channel blockers (amiodarone...) ---Ca2+ channel blockers |
|
|
|
Atrial Flutter -a rapid succession of back to back identical atrial depolarization waves -identical appearance gives risk to "sawtooth" appearance on EKG |
|
|
|
Atrial Flutter
Treatment? |
-Pharmacological cardioversion: class IA, IC, or III antiarrhythmics -Rate control: B blockers, or Ca channel blocker -Definitive treatment = ablation |
|
|
What is it? Complications? |
Ventricular Fibrillation -a completely erratic rhythm with no identifiable waves -Fatal arrhythmia without immediate CPR and defibrillation |
|
|
|
AV block
Definition? What does it look like on ECG? Treatment? |
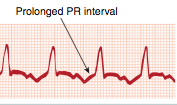
-1st degree block w/ PR interval > 200ms -benign and asymptomatic >> no treatment necessary |
|
|
|
Mobitz type 1 block
Other names? Definition? What does it look like on ECG? Treatment? |
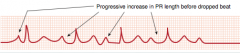
-2nd degree block/ Weckenbach block -progressive lengthening of the PR interval until a beat is dropped (P wave not followed by QRS) -Usually asymptomatic |
|
|
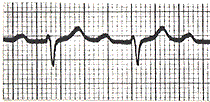
What is this? |
AV / 1st degree block |
|
|
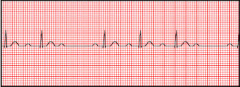
What is this? |
2nd degree block (Mobitz type I) (Weckenbach) |
|
|
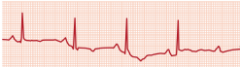
What is this? Definition? Complications? Treatment? |
Mobitz type II -dropped beats that are not preceded by a change in the PR interval -often found as a 2:1 where there are 2 or more P waves to 1 QRS response -may progress to a 3rd degree block -treat with a pacemaker |
|
|
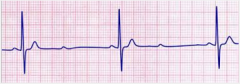
What is it? |
Mobitz type II |
|
|
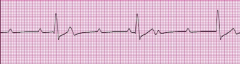
What is this? Definition? |
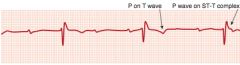
3rd degree (complete) heart block -the atria and ventricles beat independently of 1 another -Both P waves and QRS complexes are present, but the P waves bear no relation to the QRS complexes -the atrial rate is faster than the ventricular rate |
|
|
|
Treatment of 3rd degree heart block
What can cause it? |
-Treatment: pacemaker -Lyme Disease can result in a 3rd degree heart block |
|
|
|
Atrial natriuretic peptide
When/where is it released? What does it do? |
-released from atrial myocytes in response to increased blood volume and atrial pressure -Causes vasodilation and decreased Na+ reabsorption at the renal collecting tubule -Constricts efferent renal arterioles and vasodilates afferent renal arterioles via cGMP, promoting diuresis and contributing to the aldosterone escape mechanism |
|
|
|
B-type (brain) netriuretic peptide
What is it? What does it do? What is it used for clinically?
[NT-proBNP?, normal levels BNP and associated disease levls] |
-released from ventricular myocytes in response to an increase in tension -similar physiological actions to ANP, w/ an increased half life -BNP blood tests used for diagnosing HF (good NPV) - esp. used for assessing acute decompensation and monitoring response to rx - better than using Framingham guidelines as a predictive factor -recombinant form (nesiritide) for treatment of HF -normal NT-proBNP rises with age and declining renal function -no HF: BNP = 100 pg/dL; non-cardiac dyspnea = 346 pg/dL; Acute HF = 675 pg/dL |
|
|
|
Aortic arch receptors transmit via? to? in response to? |
Aortic arch transmits via the vagus nerve to the solitary nucleus of the medulla in response ONLY to increases in BP |
|
|
|
Carotid sinus receptors transmit via? to? in response to? |
Carotid sinus (dilated region at carotid bifurcation) transmits via the glossopharyngeal nerve to the solitary nucleus of the medulla in response to BOTH increases and decreases in BP |
|
|
|
Response of Baroreceptors to Hypotension |
Hypotension >> decrease arterial pressure >> decrease in stretch >> decrease in afferent baroreceptor firing >> increased efferent sympathetic firing and decrease efferent parasympathetic firing >> vasocontriction, increased HR, increased contractility, and increased BP -important response for severe hemorrhage |
|
|
|
Carotid massage |
increased pressure on carotid sinus >> increase stretch >> increase afferent baroreceptor firing >> increased AV node refractory period >> decreased HR |
|
|
|
Cushing reaction |
-triad: hypertension, bradycardia, respiratory depression -increased intracranial pressure constricts arterials >> cerebral ischemia and reflex sympathetic increased in perfusion pressure (HTN) >> increased stretch >> reflex baroreceptor-induced bradycardia |
|
|
|
Chemoreceptors
Where are they & what stimulates them? |
-Peripheral: carotid and aortic bodies - response to decreases in PO2 (< 60 mmHg), increases in PCO2, and decreased pH of blood -Central: stimulated by changes in pH and PCO2 of brain interstitial fluid, which are influenced by arterial PCO2 ---do NOT directly respond to PO2 |
|
|
|
What organ has the largest blood flow? largest share of CO? highest blood flow per gram of tissue? largest arteriovenous difference in O2? |
-Largest blood flow: Lung - 100% of cardiac output -Largest share of CO: liver -Highest blood flow: kidney -The heart has the largest difference in arteriovenous O2 b/c extraction is about 80%.
|
|
|
|
How is increased O2 demand met by the heart |
-the heart has the largest difference in arteriovenous O2 b/c extraction is 80% -increases in O2 demand must be met by an increase in coronary artery blood flow, NOT by an increase in extraction of O2 |
|
|
|
Normal heart pressures |
-PCWP is a good approximation of LA pressure -in mitral stenosis PCWP > LV diastolic pressure -pressures are measured with a pulmonary artery catheter (Swanz-Ganz catheter) |
|
|
|
What is autoregulation? |
How blood flow to an organ remains constant over a wide range of perfusion pressures |
|
|
|
Factors Determining Heart Autoregulation |
Local metabolites are vasodilatory - CO2, adenosine, NO |
|
|
|
Factors Determining Brain Autoregulation |
Local metabolies are vasodilatory - CO2 (pH) |
|
|
|
Factors Determining Kidney Autoregulation |
Myogenic and tubuloglomerular feedback |
|
|
|
Factors Determining Lung Autoregulation |
pulmonary vasculature is unique in that hypoxia causes vasoconstriction so that only well-ventialted areas are perfused -in all other organs, hypoxia causes vasodilation |
|
|
|
Factors Determining Skeletal Muscle Autoregulation |
Local metabolites - lactate, adenosine, K+, H+, CO2 |
|
|
|
Factors Determining Skin Autoregulation |
Sympathetic stimulation is the most important - for temperature control |
|
|
|
Starling forces that determine fluid movement across capillary membranes |
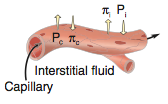
- Pc = capillary hydrostatic pressure - pushes fluid out of capillary - Pi = interstitial fluid pressure - pushes fluid into capillary - pi c = plasma colloid pressure - pulls fluid into capillary - pi i = interstitial colloid pressure - pulls fluid out of capillary |
|
|
|
Net filtration pressure across a capillary |
P[net] = [(Pc -Pi) - (pi c - pi i)] |
|
|
|
Net fluid flow in a capillary |
Kf = filtration constant
Jv = net fluid flow = (Kf)(Pnet) |
|
|
|
Edema definition & causes |
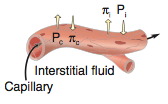
Edema is excess fluid outflow into the interstitium. Commonly caused by: - increased capillary pressure due to HR - decreased plasma protein due to nephrotic syndrome and/or liver failure - increase capillary permeablity = increase Kf caused by infection, toxins, burns - increased interstial fluid colloid osomotic pressure cause by lymph blockage |
|
|
|
Congenital Cardiac Defect(s) associated with
22q11 syndrome |
-Truncus arteriosus -Tetralogy of Fallot |
|
|
|
Congenital Cardiac Defect(s) associated with
Down Syndrome |
-ASD -VSD -AV septal defect (endocardial cushion defect) [Tetralogy of Fallot] |
|
|
|
Congenital Cardiac Defect(s) associated with
Congenital Rubella |
-septal defects -PDA -pulmonary artery stenosis |
|
|
|
Congenital Cardiac Defect(s) associated with
Turner Syndrome |
-infantile coarctation of the aorta -bicuspid aortic valve |
|
|
|
Congenital Cardiac Defect(s) associated with
Marfan Syndrome |
-MVP -thoracic aortic aneurysm and dissection -aortic regurgitation |
|
|
|
Congenital Cardiac Defect(s) associated with
Infant of Diabetic Mother |
Transposition of the great vessels |
|
|
|
Signs of Hyperlipidemia |
Xanthomas, Tendinous xanthoma, corneal arcus |
|
|
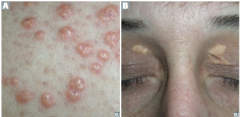
|
Due to hyperlipidemia
A. Xanthomas -plaques or nodules composed of lipid-laden histiocytes in the skin
B. Xanthelasma - xanthomas on the eyes - very common |
|
|
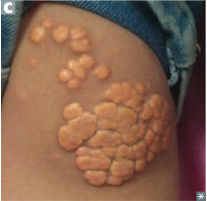
Tendinous xanthoma |
-lipid deposit in tendon, especially Achilles -due to hyperlipidemia |
|
|
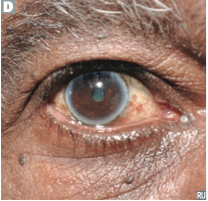
|
Due to hyperlipidemia
Corneal arcus -lipid deposits in the corney, appear early in life with hypercholesteremia -common in the elderly - arcus senilis |
|
|
|
Cardiac Dilation is Caused by? |
-greater ventricular end-diastolic volume |
|
|
|
Dyspnea on exertion is caused by? |
Failure for CO to increase during exercise |
|
|
|
Acute peridcariditis
Presentation? Types & their causes? |
Commonly presents w/ sharp pain, aggravated by inspiration, & relieved by sitting up and leaning forward. Presents with friction rub. ECG changes include widespread ST-segment elevation and/or PR depression. |
|
|
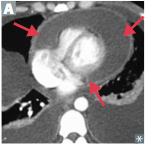
Causes what? Findings? |
Compression of heart by fluid (e.g., blood, effusions) in pericardium, leading to decreased CO. Equilibration of diastolic pressures in all 4 chambers. |
|
|
|
Pulsus paradoxus
What is it? Caused by? |
-Decrease in amplitude of systolic blood pressure by ≥ 10 mmHg during inspiration. -Seen in cardiac tamponade, asthma, obstructive sleep apnea, pericarditis, and croup.
|
|
|
|
Treatment for Primary (essential) hypertension |
Diuretics, ACE inhibitors, angiotensin II receptor blockers (ARBs), calcium channel blockers |
|
|
|
Treatment of Hypertension with CHF |
Diuretics, ACE inhibitors/ARBs, β-blockers (compensated CHF), aldosterone antagonists.
β-blockers must be used cautiously in decompensated CHF and are contraindicated in cardiogenic shock. |
|
|
|
Treatment of Hypertension with diabetes mellitus |
ACE inhibitors/ARBs. Calcium channel blockers, diuretics, β-blockers, α-blockers.
ACE inhibitors/ARBs are protective against diabetic nephropathy. |
|
|
|
Ca channel blockers
Names? Use of each? |
Amlodipine, nimodipine, nifedipine (dihydropyridine); diltiazem, verapamil (non-dihydropyridine). Use: -Dihydropyridine (except nimodipine): hypertension, angina (including Prinzmetal), Raynaud phenomenon. -Non-dihydropyridine: hypertension, angina, atrial fibrillation/flutter. -Nimodipine: subarachnoid hemorrhage (prevents cerebral vasospasm). |
|
|
|
MOA of Ca channel blockers |
-Block voltage-dependent L-type calcium channels of cardiac and smooth muscle, thereby reduce muscle contractility. -Vascular smooth muscle—amlodipine = nifedipine > diltiazem > verapamil. -Heart—verapamil > diltiazem > amlodipine = nifedipine (verapamil = ventricle). ` |
|
|
|
Ca channel blockers toxicity |
Cardiac depression, AV block, peripheral edema, flushing, dizziness, hyperprolactinemia, and constipation. |
|
|
|
Hydralazine
Use? |
Use: Severe hypertension, CHF. First-line therapy for hypertension in pregnancy, with methyldopa. Frequently coadministered with a β-blocker to prevent reflex tachycardia. |
|
|
|
Hydralazine MOA |
increased cGMP >> smooth muscle relaxation. Vasodilates arterioles > veins; afterload reduction. |
|
|
|
How do you treat a hypertensive emergency? |
-Commonly used drugs include nitroprusside, nicardipine, clevidipine, labetalol, and fenoldopam. |
|
|
|
Nitroprusside
MOA? toxicity? |
Short acting; increases cGMP via direct release of NO. Can cause cyanide toxicity (releases cyanide) |
|
|
|
Fenoldopam
MOA? |
Dopamine D1 receptor agonist—coronary, peripheral, renal, and splanchnic vasodilation. decreases BP and increases natriuresis. |
|
|
|
Nitroglycerin, isosorbide dinitrate
Use? MOA? |
-Use: Angina, acute coronary syndrome, pulmonary edema. -MOA: Vasodilate by increasing NO in vascular smooth muscle >> increase in cGMP and smooth muscle relaxation. Dilate veins >> arteries. Decrease preload. |
|
|
|
Nitroglycerin, isosorbide dinitrate
Toxicity? |
Reflex tachycardia (treat with β-blockers), hypotension, flushing, headache, “Monday disease” in industrial exposure: development of tolerance for the vasodilating action during the work week and loss of tolerance over the weekend results in tachycardia, dizziness, and headache upon reexposure. |
|
|
|
Antianginal therapy goals and determinants
Drugs used? |
Goal—reduction of myocardial O2 consumption (MVO2) by decreasing 1 or more of the determinants of MVO2: end-diastolic volume, blood pressure, heart rate, contractility.
Use nitrate, B blockers, or a combination of the 2 |
|
|
|
Nitrates in antianginal therapy
Effects? |
-Overall, they affect the preload -decrease end-diastolic volume -decrease blood pressure -increase contractility (reflex response) -increase HR (reflex response) -decrease ejection time -decrease MVO2 |
|
|
|
B blockers in antianginal therapy
Effects? |
-Overall, they affect afterload -increase end-diastolic volume -decrease blood pressure -decrease contractility -decrease HR -increase ejection time -decrease MVO2 |
|
|
|
Nitrate + B blockers in antianginal therapy
Effects? |
-no effect or decrease in end-diastolic volume -decrease blood pressure -little/no effect on contractility -decreased HR -little/no effect on ejection time -very decreased MVO2 |
|
|
|
Aternates to nitrats and/or B blockers in antianginal therapy? |
Calcium channel blockers—nifedipine is similar to nitrates in effect; verapamil is similar to β-blockers in effect.
|
|
|
|
What is contraindicated for antianginal therapy? |
Pindolol and acebutolol—partial β-agonists contraindicated in angina. |
|
|
|
lovastatin, pravastatin, simvastatin, atorvastatin, rosuvastatin
Class? MOA? Effects? Toxicity? |
-Class: HMG-CoA reductase inhibitors -MOA: Inhibit conversion of HMG-CoA to mevalonate, a cholesterol precursor [>> decreased hepatic cholesterol pool >> increased expression of hepatic LDL receptors >> increased uptake of LDL & IDL >> decreased availability of LDL and IDL for uptake by arterial macrophages[ -Effects: greatly decreases bad cholesterol, low increase on good cholesterol, low decrease on triglycerides -Toxicity: Hepatotoxicity (increased LFTs), rhabdomyolysis (esp. when used with fibrates and niacin) |
|
|
|
Niacin (vitamin B3)
Class? MOA? Effects? Toxicity? |
-Class: vitamin - lipid-lowering agent -MOA: Inhibits lipolysis in adipose tissue; reduces hepatic VLDL synthesis -Effects: medium decrease in bad cholesterol, medium increase in good cholesterol, low decrease in triglycerides -Toxicity: Red, flushed face, which is decreasedby aspirin or long- term use; Hyperglycemia (acanthosis nigricans); Hyperuricemia (exacerbates gout) |
|
|
|
cholestyramine, colestipol, colesevelam
Class? MOA? Effects? Toxicity? |
-Class: bile acid resins >> lipid-lowering agent -MOA: Prevent intestinal reabsorption -Effects: medium decrease in bad cholesterol, slight increase in good cholesterol, slight increase in triglycerides -Toxicity: Patients hate it—tastes bad and causes GI discomfort, decreases absorption of fat-soluble vitamins, Cholesterol gallstones |
|
|
|
ezetimibe
Class? MOA? Effects? Toxicity? |
-Class: cholesterol absorption blocker -MOA: Prevent cholesterol absorption at small intestine brush border -Effects: medium decrease of bad cholesterol, no effect on good cholesterol or triglycerides -Toxicity: Rare increased LFTs, diarrhea |
|
|
|
gemfibrozil, clofibrate, bezafibrate, fenofibrate
Class? MOA? Effects? Toxicity? |
-Class: Fibrates -MOA: Upregulate LPL >> increased TG clearance, Activates PPAR-α to induce HDL synthesis -Effects: low decrease in bad cholesterol, low increase in good cholesterol, great decrease in triglycerides -Toxicity: Myositis (increased risk with concurrent statins), hepatotoxicity (increasedLFTs), cholesterol gallstones (esp. with concurrent bile acid resins) |
|
|
|
Digoxin
Class? bioavailability/metabolism? |
-Class: cardiac glycoside -75% bioavailability, 20–40% protein bound, t1/2 = 40 hours, urinary excretion. |
|
|
|
Digoxin
MOA? Clinical Use? |
-MOA: Direct inhibition of Na+/K+ ATPase leads to indirect inhibition of Na+/Ca2+ exchanger/antiport. increased [Ca2+]i >> positive inotropy. Stimulates vagus nerve >> decreases HR. |
|
|
|
Digoxin
Toxicity? |
-Cholinergic—nausea, vomiting, diarrhea, blurry yellow vision (think Van Gogh). ECG—increased PR, decreased QT, ST scooping, T-wave inversion, arrhythmia, AV block. |
|
|
|
Treatment of Digoxin Toxicity |
Antidote: Slowly normalize K+, cardiac pacer, anti-digoxin Fab fragments, Mg2+. |
|
|
|
Class I antiarrhythmics
What are they (class) and what is their general mechanism? |
Na+ channel blockers - Slow or block/decrease conduction (especially in depolarized cells). Decrease slope of phase 0 depolarization and increase threshold for firing in abnormal pacemaker cells. Are state dependent (selectively depress tissue that is frequently depolarized [e.g., tachycardia]). Hyperkalemia causes increased toxicity for all class I drugs |
|
|
|
Class Ia antiarrhythmics
Names? MOA? |
Quinidine, Procainamide, Disopyramide -MOA: Na+ channel blockers - Increase AP duration, increase effective refractory period (ERP), increase QT interval.
|
|
|
|
Class Ia antiarrhythmics
Use? Toxicity? |
-Use: Both atrial and ventricular arrhythmias, especially re-entrant and ectopic SVT and VT. -Toxicity: Cinchonism (headache, tinnitus, psychosis with quinidine), reversible SLE-like syndrome (procainamide), heart failure (disopyramide), thrombocytopenia, torsades de pointes due to increased QT interval. |
|
|
|
Class Ib antiarrhythmics
Names? MOA? |
Lidocaine, Mexiletine -MOA: Na+ channel blockers - Decrease AP duration. Preferentially affect ischemic or depolarized Purkinje and ventricular tissue. Phenytoin can also fall into the IB category.
|
|
|
|
Class Ib antiarrhythmics
Use? Toxicity? |
-Use: Acute ventricular arrhythmias (especially post- MI), digitalis-induced arrhythmias. IB is Best post-MI. -Toxicity: CNS stimulation/depression, cardiovascular depression. |
|
|
|
Class Ic antiarrythmics
Names? MOA? |
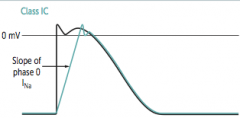
Flecainide, Propafenone. “Can I have Fries, Please.” -MOA: Na+ channel blocker - Significantly prolongs refractory period in AV node. Minimal effect on AP duration. |
|
|
|
Class Ic antiarrythmics
Use? Toxicity? |
-Use: SVTs, including atrial fibrillation. Only as a last resort in refractory VT. -Toxicity: Proarrhythmic, especially post-MI (contraindicated). IC is Contraindicated in structural and ischemic heart disease. |
|
|
|
Class II antiarrhythmics
Names? their Class? |
Metoprolol, propranolol, esmolol, atenolol, timolol, carvedilol -B blockers |
|
|
|
Class II antiarrhythmics
MOA? |
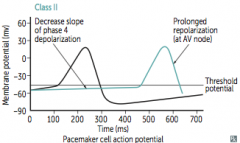
MOA: Decrease SA and AV nodal activity by decreasing cAMP & Ca2+ currents. Suppress abnormal pacemakers by decreasing slope of phase 4. AV node particularly sensitive— increasing the PR interval. Esmolol very short acting. |
|
|
|
Class II antiarrhythmics
Toxicity? Treatment of toxicity? |
Impotence, exacerbation of COPD and asthma, cardiovascular effects (bradycardia, AV block, CHF), CNS effects (sedation, sleep alterations). May mask the signs of hypoglycemia. Metoprolol can cause dyslipidemia. Propranolol can exacerbate vasospasm in Prinzmetal angina. Contraindicated in cocaine users (risk of unopposed α-adrenergic receptor agonist activity). Treat overdose with glucagon. |
|
|
|
Class III antiarrhythmics
Names? Their Class? MOA? Use? |
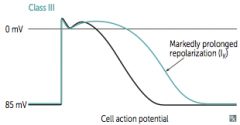
Amiodarone, Ibutilide, Dofetilide, Sotalol -Class: K+ channel blockers -MOA: increases AP duration, increases ERP. Used when other antiarrhythmics fail. increases QT interval. -Use: Atrial fibrillation, atrial flutter; ventricular tachycardia (amiodarone, sotalol) |
|
|
|
Class III antiarrhythmics
Toxicity? |
-Sotalol—torsades de pointes, excessive β blockade. -Ibutilide—torsades de pointes. -Amiodarone—pulmonary fibrosis, hepatotoxicity, hypothyroidism/ hyperthyroidism (amiodarone is 40% iodine by weight), corneal deposits, skin deposits (blue/gray) resulting in photodermatitis, neurologic effects, constipation, cardiovascular effects (bradycardia, heart block, CHF). -Remember to check PFTs, LFTs, and TFTs when using amiodarone. Amiodarone has class I, II, III, and IV effects and alters the lipid membrane. |
|
|
|
Class IV antiarrhythmics
Names? Their Class? MOA? Use? |
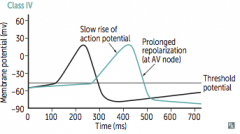
Verapamil, diltiazem -Class: voltaged gated Ca2+ channel blockers -MOA: decreases conduction velocity, increases ERP, increases PR interval -Use: Prevention of nodal arrhythmias (e.g., SVT), rate control in atrial fibrillation |
|
|
|
Class IV antiarrhythmics
Toxicity? |
Constipation, flushing, edema, CV effects (CHF, AV block, sinus node depression). |
|
|
|
Adenosine
Class? MOA? Use? Toxicity? |
-Class: antiarrhythmics -MOA: increases K+ out of cells >> hyperpolarizing the cell and decreasing ICa. -Use: Drug of choice in diagnosing/abolishing supraventricular tachycardia. -Very short acting (~ 15 sec). -Adverse effects include flushing, hypotension, chest pain. Effects blocked by theophylline and caffeine. |
|
|
|
Mg2+ |
-Class: antiarrhythmic -Use: Effective in torsades de pointes and digoxin toxicity. |
|
|
|
How can you tell which side of the heart the abnormal heart sound or murmur is on? |
all right sided heart sounds and murmur increase in intensity with inspiration |
|
|
|
What risk do all congenital heart defects have in common? |
infective endocarditis |
|
|
|
CABG
What do they use? |
Saphenous vein - will be come fibrosed over time (usually about 10 yrs) due to it become arteriolized
Can also use internal mammary artery - lasts longer |
|
|
|
Blood Pressure = |
BP = CO x TPR |
|
|
|
Hypertension is an important modifiable risk factor for.. [lecture] |
Stroke, MI, renal failure, CHF, progressive atherosclerosis, Dementia* |
|
|
|
J curve
[lecture] |
The threshold for which there is evidence that lowering the blood pressure outweighs the potential risks of treatment
-lower the BP too low is just as bad as having BP too high (think in terms of diabetes glucose levels) |
|
|
|
[How low should BP be lowered?] |
-chronic kidney disease + proteinuria = 130/80 -Diabetes = 140/90 -General population = 150 -Uncomplicated HTN = 140/90 (AHA) -HTN + Angina = 130/80 (AHA) -HTN + High risk CAD = 130/90 (AHA) |
|
|
|
[Proper BP Reading techniques] |
-In office: 2 readings, 5 min. apart, sitting in chair (arm @ heart); confirm in contralateral arm -Ambulatory: possible white coat HTN; Dipping Pressure: BP drops over night 10-20% >> those that don't dip = poor cardiovascular diagnosis -Self measurement: important for monitoring response to therapy and can evaluate white coat HTN; can also see masked HTN when it is high at high home and low in the clinic;135/85 @ home = HTN |
|
|
|
[Potential Indications for ambulatory BP monitoring] |
-unusualy variability of BP -possible white coat HTN (10-15% of pts) - may be precursor to sustained HTN -nocturnal HTN -drug-resistant HTN -determining efficacy of drug treatment over 24 hrs -HTN in pregnancy -symptomatic hypotension on meds, suggesting that patient may be normotensive -episodic HTN or autonomic dysfunction it helps to identify abnormalities, as well as normal circadian rhythm/failure of BP to decrease the 10-20% at night |
|
|
|
[Factors that may increase probability of white coat HTN] |
-office BP of 150-159/90-99 -female sex -not smoking cigarettes -HTN of recent onset -limited # of office BP readings -normal left ventricular mass |
|
|
|
[Goals of Hypertensive therapy] |
-reduce CVD mortality and morbidity -treat to BP <140/90 or BP 130/80 in patients with diabetes or CKD -achieve ABP in persons ≥ 50 yo |
|
|
|
[Treatment of HTN] |
-start with thiazide diruetics for most, but comorbidities are good reason to use other drug classes (also look at age, race, hx, efficacy, adverse rxns) -most pts will need 2 or more drugs to control it - monotherapy effective <40% of patients -If BP is ≥20/10 mmHg over target goal, start with 2 drugs, one of which should be thiazide diuretics (stage 2 HTN or higher) -reduce BP by 10-15%, minimize side effects, favor longer acting meds, be willing to change -combing drugs to lower BP w/ less side effects improve compliance, more rapidly achieves BP goal, and shows improved CV outcomes -after BP stable, follow up every 3-6 months |
|
|
|
[General Classes of antihypertensive drugs, indications for their use depending on comorbidities, etc., contraindications, and side effects] |

|
|
|
|
[Most common factor contributing to resistant HTN & most appropriate treatment] |
-non-adherence to therapy or inadequate dosing may cause patients to appear resistant to therapy -the most common factor contributing to resistant HTN in compliant patients is excess Na/volume -longer acting diruetics like hydrochlorathiazide are more appropriate for resistant HTN than shorter acting agents like furosemide -other contributors to resistant HTN: obesity, sleep apnea, cigarette smoking, panic attacks |
|
|
|
[Prognosis of HF] |
-5 yr mortality rate = 50% |
|
|
|
[High Output Cardiac Failure Causes & their Presentation] |
-wide pulse pressure, bounding pulse, flow murmur -Anemia: pallor -Hyperthyroidism: A Fib w/ RVR -Systemic AV Fistula: continuous murmur -Berberi: wasting, malnutrition -Paget's disease |
|
|
|
[DDx of of PHTN: WHO Classifications] |
-Group I: PAH - Idiopathic, Familial, associated w/ other diseases -Group II: PHTN associated w/ L heart disease -Group III: PHTN associated w/ lung disease (COPD, OSA, etc.) -Group IV: PHTN associated with thromboembolitic disease -Group V: miscellaneous (sarcoidosis, scleroderma, CREST, etc.) |
|
|
|
[R sided failure signs due to pericardial disease] |
-Elevated JVP, + Kussmaul's sign -pericardial knock -pulsus alternans -pulsus paradoxus -unexplaned ascites |
|
|
|
[R sided failure signs due to PHTN] |
-Increased P2 -Murmurs: PI, TR, diastolic rumble -Respiratory variation of S3 or S4 -Elevated JVP, +HJR -Hepatomegaly -Pedal edema |
|
|
|
[Framingham criteria for HF] |
-2 major or 1 major +2 minor criteria -Major: PND, orthopnea, elevated JVP, rale, S3, cardiomegaly, +HJR -Minor: ankle edema, night cough, DOE, hepatomegaly, pleural effusion, HR > 120 |
|
|
|
[NYHA Functional Class for HF Assessment] |
-Class I: symptoms with greater than normal exertion (no limitation of normal physical activity); Stage B & C -Class II: symptoms with normal exertion; Stage C -Class III: symptoms with restricted activity, comfortable at rest but less than ordinary activity >> symptoms; Stage C -Class IV: symptoms at rest; some Stage C, mostly D |
|
|
|
[NYHA Stages & Phenotypes of HF] |
-Stage A: at risk for HR but w/o structural disease or symptoms of HF (pts w/ HTN, atherosclerosis, DM, obesity, metabolic syndroms, using cardiotoxins, family hx) -Stage B: Structural heart disease, but without symptoms - Class I (pts w/ previous MI, remodeling including LVH & low EF, asymptomatic valvular disease) -Stage C: Structural heart disease, with prior or current symptoms - Class I, II, III (pts w/ known structural heart disease w/ signs & symptoms) -Stage D: Refractory HF requiring specialized interventions - Class IV (pts with marked HF symptoms, recurrent hospitalizations despite GDMT) |
|
|
|
[Cardiac MRI] |
- need for a biopsy is obviated by cardiac MRI; routine biopsy is not recommended - it provides unique info related to cardiac pathology - delayed enhancement >> ischemia vs. scar - complex congenital heart disease - Concern - impaired renal function (CrCl < 30) |
|
|
|
[Summary of Evidence of Statin Trials] |
-every trial showed decreased DV event rate -any statin is better than non -more potent/higher doses are better than lesser -NNT much larger for primary prevention than in secondary prevention trials -statin plus another lipid-lowering drug, is not better than statin alone |
|
|
|
[Why are statins so good?] |
-dramatic decreased in LDL -decreased in small, dense LDL -Pleiotropic: anti-inflammatory, increased eNOS activity, plaque stabilization |
|
|
|
[Statins recommended for:] |
-known CVD -Age >40-75 if: DM or LDLc 70-189 mg/dL, LDL > 190 mg/dL, 10 yr estimated CVD risk >7.5%
|
|
|
|
[Heart Pressures] |
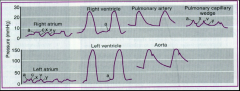
-RA: 0-5 -RV: 30/5 -PA: 30/10 -PCWP = LA: about 8 (normally about equal to PA pressure; if going into HR, >25 mmHg) -LV: 120/8 -Aorta: 120/80 |
|
|
|
[SVR & PVR equations] |
SVR= [(MAP - RAP)/CO] x 80
PVR = [(MPAP - PCWP)/CO] x 90 |
|
|
|
[Cardiac Indices and Associated PA saturations] |
-70% saturation (normal) = 3 L/min cardiac index (normal) -50% saturation = 2 L/min cardiac index -30% saturation = 1 L/min cardiac index |
|
|
|
[Hemodynamic chart from lecture w/ associated pressures and pathology] |
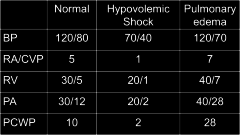
Pathologic States: -loss of volume >> low pressures & high resistance -Septic Shock >> low pressure & low resistance -Cardiogenic shock >> low pressures, high filling pressures (PCWP, LVEDP) |
|
|
|
[Hemodynamics in acute MI] |
-Normal Hemodynamics: PCWP <18 mmHg, CI > 2.2 -Pulmonary Congestion: Warm & Wet >> PCWP > 25 mmHg, CI > 2.2 -Peripheral Hypoperfusion: Cold & dry >> PCWP < 10 mmHg, CI < 2.2 -Pulmonary Congestion & Peripheral Hypoperfusion: Cold & Wet >> PCWP > 25 mmHg, CI < 2.2 |
|
|
|
[What defines a shunt as significant?] |
-Qp/Qs ≥ 1.5 (normal = 1) >> L to R shunt -Qp = blood flow through pulmonary circuit -Qs = blood flow through systemic circuit |
|
|
|
CHA2DS2 VASC Score |
-CHF/LV dysfunction - 1 pt -Hypertenion - 1 pt -Age >75 - 2 pts -Diabetes Mellitus - 1 pt -Stroke, TIA, Thromboembolism - 2 pts -Vascular disease - 1 pt - age 65=75 - 1 pt -Sex (female) - 1 pt |
|
|
|
[Grading of Angina Pectoris -CCS] |
-Class I: ordinary physical activity doesn't cause angina; agina occurs with strenuous/rapid/prolonged exertion -Class II: slight limitation of ordinary activity - angina w/ climbing flight of stairs or w/ walking > 2 blocks, walking or climbing stairs rapidly, uphill or after meals, strong emotional rxn, cold, wind, or during first few hours of waking -Class III: Marked limitations of normal activity; angina occurs w/ walking 1-2 blocks or climbing one flight of stairs normally -Class IV: Inability to carry out any physical activity without symptoms; angina may occurs at rest |
|
|
|
[JNC 8 summary for treatment/ prevention of ischemic heart disease] |
-≥ 60 yo, treat when BP ≥ 150 systolic or 90 diastolic -< 60 yo, treatment initiation and goals are 140/90, use in pts with ≥ 18 yo w/ CKD or diatbets -nonblack pts w/ HTN, initial treatment should be thiazide diuretic, CCV, ARB, or ACE inhibitor; general black popularion: use thiazide diuretic or CCB -pts ≥ 18 yo w/ CKD, initial or add on therapy should include ACE inhibitor or ARB |
|
|
|
[TIMI Score of UA/STEMI] |
predicts 30 day mortality rate after MI -Aged ≥ 65 yrs -≥ 3 CAD risk factors -prior CAD (>50% stenosis) -aspirin in last 7 days -≥ 2 anginal events in ≤ 24 hrs -ST deviation -elevated cardiac markers (CK-MB or troponin 1) |
|
|
|
[Summary of inpatient meds for MI patients] |
-aspirin + 2nd antiplatelet (clopidogrel) + GpIIb/IIIa inhibitors -unfractionated heparing or enoxaparin -high dose stain -Beta blocker -ACE I (if BP tolerates) -send them home on aspirin, clopidogrel, beta blockers, ACE I, and statin |
|
|
|
Treatment of HF
Stage A |
Goal: Prevention -ACE I or if untolerant of ACE I >> ARB for patients with vascular disease or DM |
|
|
|
Treatment HF
Stage B |
Goal: prevent HF symptoms, prevent further remodelling -ACE I or ARB if intolerant to ACE I (should be used in all esp. w/ reduced EF) -Beta blockers if hx of MI or ACS and reduced EF OR just reduced EF regardless of hx -statins should also be used in those patients with hx of MI or ACS - prevent symptomatic HF and cardiovascular events -ICD placement in those at ≥ 40 days post MI, EF ≤30%, on appropriate medical therapy, and have a good survival outlook -Nondihydropyridine CCB may be harmful in asymptomatic pts w/ low LVEF and no symptoms after MI |
|
|
|
Treatment of HF
Stage C |
Systolic HF - Goals: control symptoms, educate, prevent hospitalization & mortality; exercise training, Na restriction, CPAP for those with sleep apnea, cardiac rehab -diuretics for fluid retention -ACE I or ARB + Beta Blockers for all -Aldosterone Antagonists if GFR > 30mL/min if EF <35%, or post MI w/ EF <40% -Add loop diuretics if volume overloaded -Add Hydral-Nitrates if African American -ICD therapy same as for Stage B; CRT also considered Diastolic HF - Goals: control symptoms, improve HRQOL, prevent hospitalization & mortality; Na restriction, exercise therapy -diuresis to relieve congestive symptoms, repurfusion? |
|
|
|
Treatment of HF
Stage D |
Goals: control symptoms, improve HRQOL, reduce hospital readmissions, establish pts' end of life goals -fluid restriction -IV Inotropic support if cardiogenic shock (systolic dysfunction) -heart transplant -chronic inotropes -temporary or permanent MCS - VADS used as bridge to transplant -palliative care and hospice -ICD deactivation |
|
|
|
Treatment of Hospitalized HF Patient (decompensated HF) |
-Loop diuretics for volume control (high dose then taper >> followed by Beta blocker at low does after stabilized volume >> thrombosis prophylaxis -monitor electrolytes, BUN, Cr during titration of meds -if diuresis inadequate, increase dose or add 2nd line diuretic like thiazide; low dose dopamine can be attempted, and nitro (if patient stable) can be attempted to help with diuresis |
|

