![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
53 Cards in this Set
- Front
- Back
|
Mole
|
A number of particles the same as in 12g of carbon.
|
|
|
Relative Formula Mass
|
The formula mass of a molecule is the sum of the atomic weights of the atoms in the empirical formula of the compound.
|
|
|
Relative Atomic Mass
|
The mass of an atom compared to 1/12 of a carbon atom.
|
|
|
Molar Mass
|
The relative formula mass in grams-unit g/mol.
|
|
|
Conservation of Mass
|
The total mass of reactants equals the total mass of the products formed.
|
|
|
Thermal Decomposition
|
The breaking down of a compound into two or more products on heating.
|
|
|
Molecualar Formula
|
The formula of a chemical using symbols in the periodic table. E.G. methane has a molecular formula of CH4.
|
|
|
Displayed Formula
|
When the formula of a chemical is written showing all the atoms and all the bonds.
|
|
|
Empirical Formula
|
The simplest way of writing a whole number ratio of the atoms in a compound.
|
|
|
Concentration
|
The amount od chemical dissolved in a certain volume of solution.
|
|
|
Solution
|
When a solute dissolves in a solvent, a solution forms.
|
|
|
Solvent
|
Liquids in which the solutes dissolve to form a solution.
|
|
|
Solute
|
Solids which dissolves in a solvent to form a solution.
|
|
|
Guideline Daily Amount (GDA)
|
Recommended values for safe amounts of fats, saturated fats, sugar and salt.
|
|
|
Neutralisation Reaction
|
Reaction between H+ ions and OH- ions (acid and base react to make a salt and and water).
|
|
|
Acid
|
Solution with a pH less than 7.
|
|
|
pH Scale
|
Scale in which acids have a pH of less than 7, alkalis a pH of above 7 and neutral have a pH of 7.
|
|
|
Alkali
|
Substances which produce OH- ions in water.
|
|
|
Neutral
|
A neutral substance has a pH of 7.
|
|
|
Burette
|
A graduated tube with a tap for accurately adding a liquid, showing the amount added.
|
|
|
Pipette
|
A narrow, usually calibrated glass tube into which small amounts of liquid are suctioned for transfer or measurement.
|
|
|
Pipette Filler
|

Item used to fill pipettes.
|
|
|
Curved Line
|
A line with changing gradient.
|
|
|
End Point
|
The point at which an indicator changes colour and a titration is stopped.
|
|
|
Indicator
|
Chemicals which change colour according to the pH (indicators show how acid or alkali a substance is).
|
|
|
Titration
|
An accurate method for neutrilisation.
|
|
|
Titre
|
The concentration in a solution of a dissolved substance as shown by titration.
|
|
|
Anomalous
|
Abnormal result.
|
|
|
Gas Syringe
|
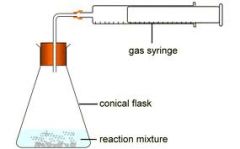
-
|
|
|
Displacement Reaction
|
Chemical reaction where one element displaces another element from a compound.
|
|
|
Limiting Reactant
|
Chemical used up in a reaction that limits the amount of product formed.
|
|
|
Excess Reactant
|
The excess reactant is the reactant in a chemical reaction with greater amount than necessary to react completely with the limiting reactant.
|
|
|
RTP
|
Rapid thermal processing (RTP) is a process in which a wafer is heated to a specified temperature for short periods of time.
|
|
|
Molar Volume
|
The volume occupied by one mole of a substance in the form of a solid, liquid, or gas.
|
|
|
Reversible Reaction
|
A reaction where products are made, which break back down into the reactants.
|
|
|
Equilibrium
|
When the forward and backwards reactions are happening at the same rate.
|
|
|
Position of Equilibrium
|
Position refers to the physical change in the equilibrium if it is disturbed.
|
|
|
Closed System
|
A closed system is a type of thermodynamic system where mass is conserved within the boundaries of the system, but energy is allowed to freely enter or exit the system.
|
|
|
Contact Process
|
The major industrial process used to make sulfuric acid, by oxidizing sulfur dioxide in the presence of a solid catalyst and absorbing the resulting sulfur trioxide in water.
|
|
|
Weak Acid
|
Acid that has a pH of between 4 and 6, and only partly ionises.
|
|
|
Strong Acid
|
Acid that has a pH of between1 and 3, and completly ionises.
|
|
|
Hydrogen Ions
|
The positively charged ion of hydrogen, H+, formed by removal of the electron from atomic hydrogen and found in all aqueous solutions of acids.
|
|
|
Ionisation
|
Ionisation is the formation of ions. An ion is a charged ion - positive or negative.
|
|
|
Electrode
|
Terminal that conducts electricity, put into a cell to perform electrolysis.
|
|
|
Electrolysis
|
When an electrical current is passed through a solution which conducts electricity.
|
|
|
Electrical Conductivity
|
The measurment of the ability to conduct electricity.
|
|
|
Limescale
|
Hard white substance found inside 'kettles' (mostly calcium carbonate).
|
|
|
Precipitation Reaction
|
Chemical test in which a solid precipitate is formed- tests for metal ions.
|
|
|
Precipitate
|
Solid formed in a solution during a chemical reaction.
|
|
|
State Symbols
|
Symbols used in equations to show whether something is solid, liquid,gas or in solution in water.
|
|
|
Ionic Equation
|
An equation representing the formation of ions, by the transfer of electrons.
|
|
|
Spectator Ions
|
Ions that do not take part directly in reactions.
|
|
|
Ionises
|
Adds or removes electrons from an atom leaving it charged.
|

