![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
53 Cards in this Set
- Front
- Back
|
What do geologists study rocks? |
To see how the Earth's surface has changed |
|
|
How can tectonic plates move? |
By sliding past each other, colliding or pulling apart. |
|
|
What do plate collisions make? |
Mountain ranges |
|
|
What is a supercontinent? |
Different continents that have drifted and crashed into each other. |
|
|
How are igneous rocks formed? |
As volcanic lava solidifies |
|
|
Who proposed the theory of continental drift at the beginning of the 20th century? |
Alfred Wegener |
|
|
What was Wegener's theory? |
His idea was that the Earth's continents were once joined together, but gradually moved apart over millions of years. |
|
|
Why does Wegener's theory provide an explanation of similar fossils and rocks on continents that are far apart from each other? |
If we apply the continental drift theory, then it would suggest that all the continents used to be in the same area in order to have the same fossils and rocks on far away continents. This means that continents must have moved away from each other in order for their to be varieties on every continent. |
|
|
What are rocks? |
Raw materials found buried in the Earth's crust |
|
|
Name three important raw materials. |
Coal, salt and limestone |
|
|
How was limestone formed? |
-Limestone formed while Britain was covered by sea. -Shellfish died forming sediments on the sea bed. -Sediments compacted and hardened to form limestone, a sedimentary rock. -Tectonic plate movements pushed the rock to the surface. -Gradually the rocks above were eroded away until the limestone was exposed. |
|
|
How was coal formed? |
Coal formed in wet swampy conditions when plants like trees and ferns died and became buried. This excluded oxygen, slowing down decay. |
|
|
How was salt formed? |
-Rivers brought dissolved salts into the sea. -Climate warming evaporated the water, leaving salt that mixed with sand blown in by the wind. -Rock salt formed and was buried by other sediments. |
|
|
What do ripple marks in rocks indicate? |
Water flow from rivers or waves in the sea |
|
|
Why is rock salt spread on icy roads? |
The salt in solution lowers the freezing point, preventing ice forming easily. |
|
|
How can pure salt be obtained? |
Solution mining |
|
|
What can mining rock salt and solution mining cause? |
Subsidence |
|
|
What is an environmental impact of extracting salt? |
Evaporating salt from sea water takes up large areas and spreads salt into the local environment, damaging habitats. |
|
|
Name a use of salt in the food industry. |
-Flavouring -Preserving |
|
|
A higher salt level prevents bacteria growth. True of false? |
True |
|
|
What can a large salt intake cause in the body? |
-High blood pressure -Heart failure -Strokes |
|
|
Alkalis make indicators change colour. Litmus paper turns what colour in acids? |
Blue |
|
|
What do alkalis neutralise acids to make? |
Salts |
|
|
What is neutralisation? |
Acids react with alkalis to form salts |
|
|
Acid + Alkali = ? + ? |
salt and water |
|
|
List three uses of alkalis. |
-Dyeing cloth -Neutralising acid soil -Making soap -Making glass |
|
|
What were used in the past as sources of alkalis? |
stale urine and ash from burnt wood
|
|
|
What was the first alkali to be manufactured? |
Lime (calcium oxide) |
|
|
In 1787 what did Nicholas Leblanc discover? |
How to manufacture an alkali |
|
|
How did the Leblanc process make Sodium Carbonate? |
By reacting salt and limestone, heated with coal. |
|
|
The Leblanc process gave off which harmful gases? |
-Hydrogen Chloride -Hydrogen Sulfide |
|
|
Later, a process was invented to change the harmful hydrogen chloride into useful substances. What were they? |
Chlorine and hydrochloric acid |
|
|
Which chemical reaction converts hydrogen chloride to chlorine? |
Oxidation |
|
|
What is an alkali? |
A solution with a pH greater than 7 and contains Hydroxide ions (OH-). |
|
|
Hydroxide + Acid = ? + ? |
salt + water |
|
|
Carbonate + Acid = ? + ? + ? |
salt + water + carbon dioxide gas |
|
|
Sodium Hydroxide + Sulfuric Acid = ? + ? |
sodium sulfate + water |
|
|
Why is Chlorine added to water supplies? |
To kill microorganisms |
|
|
Why did Chlorination make a big contribution to public health? |
Chlorination killed water-borne microorganisms that cause diseases like cholera and typhoid. |
|
|
Hydrochloric acid + sodium hydroxide = ? + ? |
sodium chloride + water |
|
|
What is Electrolysis? |
Breaking up compounds using electric current. |
|
|
The electrolysis of brine (sodium chloride solution) makes which 3 products? |
-Chlorine gas -Hydrogen gas -Sodium Hydroxide solution |
|
|
What are the two electrodes called and what are their charge? |
-Anode and Cathode -PANIC -> Positive Anode Negative Is Cathode |
|
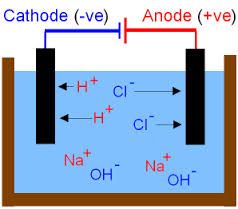
Describe the process of Electrolysis of Brine using Sodium Chloride solution. |
1. NaCl is split into Cl- ions, Na+, H+ and OH-. 2. H+ (cation) is attracted to the cathode (which is negative) and forms Hydrogen (H2). 3. Cl- is attracted to the anode and forms Chlorine (Cl2). 4. NaOH is left in the solution. |
|
|
H2O = ? + ? |
H+ + OH- |
|
|
What is the difference between bases and alkalis? |
Bases can't dissolve in water, but alkalis can. |
|
|
Why is Electrolysis expensive? |
Large amounts of electricity are needed for electrolysis. |
|
|
During Brine Electrolysis, where does the Chlorine and Hydrogen form? |
Chlorine is formed at the anode and Hydrogen at the cathode. |
|
|
Why will chemicals stay in the environment forever? |
Chemicals contain elements and elements can't be destroyed, so they remain in the environment forever. |
|
|
To decide the level of risk of a particular chemical, what are the 4 main things we need to know? |
-how much of it is needed to cause harm -how much will be used -the chance of it escaping into the environment -who or what it may effect |
|
|
What is the impact if PVC is burnt? |
Burning PVC gives off toxic gases including dioxins. If eaten, these chemicals build up in fat and are thought to cause cancer. |
|
|
What does a LCA measure? |
LCAs measure the energy used to make, use and dispose of a substance, and its environmental impact. |
|
|
List the things that we need to consider at each stage of an LCA. |
-How much natural resources are required? -How much energy is needed or produced? -How much water and air is used? -How is the environment affected? |

