![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
14 Cards in this Set
- Front
- Back
|
Rate of reaction depends upon |
Amount of energy transfered in each collision The frequency of collisions Increasing the temperature increases the chance of a sucessful collision as particles have more energy |
|
|
What is relative atomic mass |
Mass compared to carbon Shown in periodic table top number in each box |
|
|
What is relative formula mass |
Mr mass of a formula compared to a single carbon atom |
|
|
Conservation of mass |
Mass of products must equal the mass of reactants |
|
|
Percentage yield |
Actual yield / predicted yield x 100 |
|
|
Atom economy |
Mr of desired product / total Mr of all products x 100 |
|
|
Endothermic vs exothermic reactions |
Endothermic energy is absorbed to break bonds Exothermic energy is released to create bonds |
|
|
Function of a calorimeter |
Filled with a known mass of water with measure temperature Wick of a spirit burner is lit After experiment temperature of water is measured again |
|
|
Specific latent heat |
How much energy needed to melt or boil it |
|
|
Specific heat capacity |
Amount of energy needed to change the temperature of a 1kg substance by 1 Water 4181 Oxygen 918 Led 128 |
|
|
Batch vs continously |
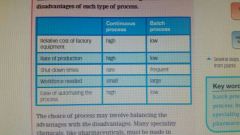
|
|
|
Tests for purity |
Chromotography paper Substances are separated and appear when heated or under UV light |
|
|
Structure of diamond vs graphite |
Diamond one carbon connected to four others Graphite layers can slide apart and have free electrons |
|
|
Bucky balls |
Discovered in 1985 each molecule has 60 carbon atoms Joined together to make nano tubes: Strongest material can conduct electricity can act as catalysts |

