![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
225 Cards in this Set
- Front
- Back
|
what is the first site of hematopoiesis in an embryo?
When does hematopoiesis stop at this site? |
the Yolk Sac
Stops before 3 months gestation |
|
|
after the yolk sac, where does the site of hematopoiesis move to?
|
Liver and Spleen
|
|
|
at about the 4th month of gestation, hematopoiesis begins at this site and continues to be the site of hematopoiesis throughout life.
|
bone marrow
|
|
|
at term, where is the main site of hematopoiesis?
|
bone marrow
|
|
|
compare the bone marrow specific sites of hematopoiesis between a child and adult
|
infancy, early childhood -> all marrow cavities are active
adolescence onward -> axial skeleton only |
|
|
in which bone is the cellularity of bone marrow in adults the highest?
|
vertebrae
- vertebrae > sternum > rib |
|
|
explain the difference between a pluripotent and a multipotent stem cell
|
1. pluripotent - capable of ALL hematopoietic cell lines
2. multipotent - capable of EITHER lymphoid or non-lymphoid cell lines |
|
|
define a progenitor cell
are they capable of renewal? |
cell capable of unipotential maturation to hematopoietic cells: NO RENEWAL
|
|
|
define a precursor cell
|
first morphologically recognized cell in each respective hematopoietic line. "Blasts"
|
|
|
four major controls of differentiation in hematopoiesis?
|
1. growth factors
2. colony stimulating factors (CSF) 3. erythropoietin 4. thrombopoietin |
|
|
what type of growth factors are used in control of differentiation in hematopoiesis? (2)
|
interleukins
stem cell factor |
|
|
what are colony stimulating factors used for in differentiation of hematopoiesis?
|
CSFs control maturation of specific cell lineages
|
|
|
where is erythropoietin found? function?
|
EPO is found in kidney interstitial tubule cells
controls erythrocyte maturation |
|
|
function of thrombopoietin?
|
controls and facilitates maturation of megakaryocytes to platelets
|
|
|
two stem cells involved in erythropoiesis?
|
1. puripotent
2. myeloid multipotent |
|
|
two types of progenitor cells in erythroid hematopoiesis?
|
1. burst forming unit, erythroid (BFU-E)
2. colony forming unit, erythroid (CFU-E) |
|
|
three molecules that control progenitor cell (BFU-E, CFU-E) formation?
|
1. thrombopoietin (initial)
2. IL-11 3. erythropoietin (last) |
|
|
metabolism differences when in marrow vs. blood stream?
|
marrow - aerobic metabolism and hemoglobin synthesis
bloodstream - anaerobic |
|
|
four erythrocyte precursur cells in order of differentiation?
|
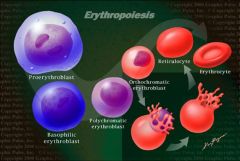
1. proerythroblast
2. polychromatic erythroblast 3. orthochromatic erythroblast 4. reticulocyte |
|
|
describe a proerythroblast
|
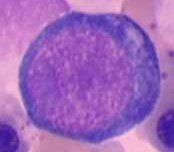
- first recognizable erythroid cell
- large with small intense blue cytoplasm - round, homogenous nucleus |
|
|
describe a polychromatic erythroblast
|
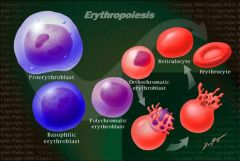
- somewhat smaller
- more cytoplasm - greyish from initiation ribosome for hemoglobin synthesis - nucleus has clumped chromatin |
|
|
describe an orthochromatic erythroblast
|
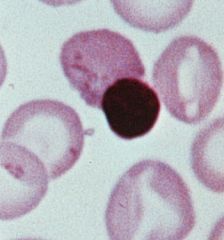
- reddish cytoplasm from start of hemoglobin synthesis
- dense pyknotic nucleus is expelled to form a reticulocyte |
|
|
describe a reticulocyte
|
- anucleated cell
- circular without biconcave shape - small #s may appear in peripheral blood |
|
|
describe structure of an erythrocyte
|
- biconcave with central pallor
|
|
|
lifespan in blood of a mature erythrocyte?
|
~120 days
|
|
|
three major metabolic pathways used by a RBC?
|
ALL ANAEROBIC PATHWAYS
1. glycolysis 2. pentose shunt (thru glucose-6-phosphate) 3. shunt through 2,3-DPG pathway |
|
|
how many globin chains in one hemoglobin molecule?
|
four
|
|
|
What are the three types of hemoglobin?
|
A - two alpha, two beta
F - (fetal) - two alpha, two gamma A2 - two alpha, two delta |
|
|
how many oxygen molecules can bind to one hemoglobin molecue?
|
four (one to each heme group)
|
|
|
define anemia
|
a reduction in the circulating RBC mass below normal limits, usually defined by a decrease in hemoglobin or hematocrit
|
|
|
possible reasons for an increased hemoglobin level? (4)
|
1. dehydration
2. polycythemia vera 3. COPD 4. increased altitude |
|
|
definition of hematocrit?
|
ratio of volume of erythrocytes to that of the whole blood (packed cell volume)
|
|
|
relationship between hematocrit and hemoglobin?
|
hematocrit normally should be approximately 3x HgB
|
|
|
possible reasons for a decreased hematocrit level? (2)
|
1. anemia
2. pregnancy |
|
|
possible reasons for an increased hematocrit level?
|
1. polycythemia vera
2. COPD 3. smoking 4. dehydration 5. hypovolemia |
|
|
define Mean Corpuscular Volume (MCV)
|
average red blood cell volume
|
|
|
possible reasons for MCV to be decreased? (6)
|
1. iron deficiency anemia
2. thallasemias 3. anemia of chronic disease 4. sideroblastic anemia 5. chronic renal failure 6. lead poisoning |
|
|
possible reasons for MCV to be increased? (8)
|
1. Vitmamin B12 deficiency
2. folate deficiency 3. liver disease 4. alcohol abuse 5. reticulocytosis 6. hypothyroidism 7. marrow aplasia 8. myelofibrosis |
|
|
define red cell distribution width (RDW)
|
an indicator of VARIATION in RBC size
(used in combination with MCV to determine cause of anemia) |
|
|
normal RDW and elevated MCV could be________________? (1)
|
aplastic anemia
|
|
|
what type(s) of anemia could this be?
- normal RDW and elevated MCV? (3) |
1. anemia of chronic disease
2. blood loss anemia 3. hemolytic anemia |
|
|
what type(s) of anemia could this be?
- normal RDW and decreased MCV? (2) |
1. anemia of chronic disease
2. heterozygous thalassemia |
|
|
what type(s) of anemia could this be?
- elevated RDW and elevated MCV? (3) |
1. B12 deficiency
2. folate deficiency 3. immune hemolytic anemia |
|
|
what type(s) of anemia could this be?
- elevated RDW and normal MCV? (3) |
1. early iron deficiency
2. early B12 deficiency 3. early folate deficiency |
|
|
what type(s) of anemia could this be?
- elevated RDW and decreased MCV? (2) |
1. iron deficiency
2. RBC fragmentation |
|
|
define reticulocyte count
|
measure of recently released immature erthrocytes from marrow
|
|
|
what type(s) of anemia could this be?
increased reticulocyte count (2) |
1. hemolytic anemias
2. blood loss anemia |
|
|
what could cause the following:
- decreased reticulocyte count (5) |
1. aplastic anemia
2. marrow suppression 3. cirrhosis 4. transfusion 5. anemia of disordered maturation (iron deficiency, megaloblastic anemia, anemia of chronic disease) |
|
|
hypochromia is most often seen in which type of anemia?
|
iron deficiency anemia
|
|
|
polychromasia/polychromatophilia is associated with an increase in which lab value?
|
an increased reticulocyte count (blue-grey tint seen in RBCs)
|
|
|
what is polychromasia/polychromatophilia?
|
a blue/grey tint seen in RBCs
|
|
|
define anisocytosis. where is this seen?
|
variation in RBC SIZE
seen in severe anemias |
|
|
define poikilocytosis. where is this seen?
|
variation in RBC SHAPE
seen in severe anemias |
|
|
which cell type would be observed in the following:
1. liver disease 2. thalassemias 3. moderate to severe iron deficiency |
target cells
|
|
|
which cell type would be observed in the following:
- hereditary spherocytosis - autoimmune hemolytic anemia |
spherocytes
|
|
|
which cell type would be observed in the following:
- microangiopathic hemolytic anemia - post-severe burns |
Schistocytes
|
|
|
which cell type would be observed in the following:
- abetalipoprotienemia - liver disease - hemolytic anemias |
Acanthocytes
|
|
|
which cell type would be observed in the following:
- megaloblastic anemia |
Macrocytic ovalocytes
|
|
|
which cell type would be observed in the following:
- hereditary ovalocytosis |
normochromic ovalocyte
|
|
|
which cell type would be observed in the following:
- hereditary elliptocytosis - megaloblastic anemias |
Elliptocytes
|
|
|
which cell type would be observed in the following:
- myeloid metaplasia - hypersplenism - leukoerythroblastic response |
tear drops
|
|
|
what is the leukoerythroblastic response and what does it suggest?
|
presence of normoblasts and immature granulocytes in the circulation
indicative of a space occupying lesion in the bone marrow |
|
|
which cell type would be observed in the following:
- alcoholic liver disease |
stomatocytes
|
|
|
which cell type would be observed in the following:
- multiple myeloma - waldenstrom's macroglobulinemia |
Rouleaux formation
|
|
|
which cell type would be observed in the following:
- autoimmune hemolytic anemia - cold agglutinin disease |
agglutination
|
|
|
which cell type would be observed in the following:
- post splenectomy - hemolytic anemia |
Howell-Jolly bodies
|
|
|
what are Howell-Jolly bodies?
|
remnants of DNA within RBCs
|
|
|
when are nucleated RBCs within peripheral blood:
1. normal 2. abnormal |
1. fetus or young infant
2. healthy adult |
|
|
under what circumstances would nucleated RBCs be seen in an adult?
|
1. increased demands placed on marrow
2. extramedullary hematopoiesis 3. marrow replacement |
|
|
what are the three major pathophysiologic mechanisms behind an anemia?
|
1. decreased RBC production
2. increased RBC destruction (often secondary to defective RBCs) 3. acute blood loss |
|
|
pathophysiology of aplastic anemia?
|
suppression of pluripotent STEM CELLS due to
- somatic mutation - immune (T-cell mediated) mechanism |
|
|
4 possible etiologies of aplastic anemia?
|
1. drugs or chemicals
2. irradiation 3. infections 4. inherited |
|
|
in aplastic anemia:
1. morphology of bone marrow? 2. morphology of blood? |
1. hypocellular
2. pancytopenia |
|
|
3 clinical features of aplastic anemia?
|
infections
bleeding pallor |
|
|
treatment for aplastic anemia?
|
bone marrow transplant
-or- immunosuppressive therapy |
|
|
what is pure red cell aplasia?
|
absence of RBC precursors
|
|
|
3 possible causes of red cell aplasia?
|
1. drug induced
2. virus induced 3. neoplasms |
|
|
what is one the most common causes of anemia?
|
iron deficiency
|
|
|
4 possible etiologies of iron deficiency anemia?
|
1. low dietary intake
2. malabsorption 3. increased iron demands (pregnancy, infancy) 4. chronic blood loss (GI bleed, menorrhagia) |
|
|
regarding iron-deficiency anemia:
1. morphology of peripheral blood? 2. morphology of bone marrow? |
1. microcytic, hypochromic RBCs
2. normoblastic hyperplasia |
|
|
what is the Plummer-Vison triad observed in iron deficiency anemia?
|
1. hypochromic, microcytic anemia
2. atrophic glossitis 3. esophageal webs |
|
|
what is koilonchia?
|
spoon shaped nails - seen in iron deficiency anemia
|
|
|
describe the following levels in a patient with iron deficiency anemia:
1. serum iron concentration 2. ferritin concentration 3. TIBC 4. plasma transferrin saturation |
1. decreased
2. decreased 3. increased 4. decreased |
|
|
treatment for iron deficiency anemia?
|
iron supplementation
|
|
|
pathophysiology of anemia of chronic disease?
|
abnormal iron metabolism (ineffective use of iron from macrophage stores)
|
|
|
regarding anemia of chronic disease:
morphology of peripheral blood smear? |
normocytic, normochromic anemia
-or- microcytic, hypochromic anemia |
|
|
anemia of chronic disease is seen in association with what three stimulants?
|
1. chronic microbial infections
2. chronic immune disorders 3. neoplasms |
|
|
what is the pathophysiology of anemia of renal disease?
|
decreased production of erythropoietin by damaged kidneys.
|
|
|
what is the morphology of anemia of renal disease?
|
normochromic, normocytic anemia with echinocytes
|
|
|
what are echinocytes that are commonly seen in anemia of renal disease?
|
Burr cells
|
|
|
if renal disease has a hypertensive component, what additional cellular morphologies would be seen in anemia of renal disease?
|
red cell fragmentation
schistocytes |
|
|
treatment for anemia of renal disease?
|
EPO
|
|
|
what is myelophthisic anemia?
|
hypoproliferative anemia. usually caused by infiltration of the bone marrow by metastatic disease
|
|
|
what would be seen on the blood smear of a patient with myelophthisic anemia?
|
teardrop cells
immature WBC, RBC |
|
|
why is lead poisoning so "toxic"?
|
lead interferes with several enzymes involved with heme synthesis
|
|
|
what is a hallmark of lead poisoning seen in the marrow and peripheral blood smears?
|
basophilic stippling
ringed sideroblasts (due to impaired iron utilization) |
|
|
two main causes of megaloblastic anemia?
|
Vitamin B12 or folate deficiency
|
|
|
why are B12 and folate so important?
|
essential enzymes required for DNA synthesis
(lack of them is most noted in hematopoiesis) |
|
|
1. what is the main site of vitamin B12 absorption?
2. what is the main site of folate absorption? |
1. ileum
2. jejunum |
|
|
pathophysiology behind B12 or folate deficiency anemia?
|
- no DNA synthesis
- results in ineffective erythropoiesis and production of abnormal erythrocytes susceptible to hemolysis |
|
|
why would a gastrectomy result in a B12 deficiency?
|
loss of intrinsic factor
|
|
|
why would pernicious anemia be a result of vitamin B12 deficiency?
|
pernicious anemia is an autoimmune disorder that damages gastric parietal cells (cells that release intrinsic factor)
|
|
|
why would an ileal resection result in B12 deficiency?
|
ileum is where absorption of B12 occurs
|
|
|
why could pregnancy result in a B12 deficiency?
|
increased demand of B12
|
|
|
which two groups of "inadequate dieters" would possibly experience B12 deficiency?
|
alcoholics
vegans |
|
|
describe the morphology of the bone marrow seen in megaloblastic anemia
|
*megaloblastic erythroid hyperplasia
*giant myelocytes, metamyelocytes *hypersegmented neutrophils |
|
|
describe the effects of a B12 deficiency on the CNS
|
demyelination of dorsal and lateral tracts of spinal cord
|
|
|
what causes the majority of hemolytic anemias?
|
defective RBC synthesis
|
|
|
describe intravascular hemolysis
|
lysis of RBCs within vasculature. Would see hemoglobinuria and hemoglobinemia.
|
|
|
describe extravascular hemolysis
|
"normal" route for removal of senescent RBCs
- occurs mainly by macrophages within the spleen Main symptoms: anemia, jaundice |
|
|
what is acanthocytosis?
|
defect within the lipid bilayer of the RBC membrane.
*see spiny projections on the RBC surface (Spur cells) *results in hemolysis |
|
|
what is the most common cause of aquired acanthocytosis?
|
chronic liver disease
(increased cholesterol deposited in the lipid bilayer) |
|
|
1. hereditary spherocytosis is a defect in?
2. result? |
1. ankryn or spectrin
2. RBC is a sphere - no biconcave shape |
|
|
three presenting s/s of a patient with hereditary spherocytosis?
|
1. anemia
2. moderate splenomegaly 3. jaundice |
|
|
treatment to induce symptom relief in hereditary spherocytosis?
|
splenectomy
(corrects anemia and hemolysis) |
|
|
in regards to glucose-6-phosphate deficiency: why is G6PD needed?
|
G6PD protects RBCs from oxidative injuries
(it reduces glutathione) |
|
|
what happens when the RBC is exposed to oxidative stress?
|
hemoglobin is oxidized and denatured
|
|
|
inheritance of G6PD deficiency?
|
XLR
*appears to provide some protection from malaria, therefore seen in highest incidence in regions with endemic malaria. |
|
|
in G6PD deficiency, hemoglobin is oxidized and precipitates in the cell forming a?
|
Heinz body
|
|
|
what type of RBCs are seen in the PBS of a patient with G6PD deficiency?
|
Bite cells
|
|
|
two types of G6PD deficiencies and where they are seen?
|
1. G6PD A (African Americans)
2. G6PD Mediterranean (middle eastern descent) |
|
|
what food is known to cause hemolytic anemia in a person with G6PD deficiency?
|
Fava beans
|
|
|
pathophysiology of Beta thalassemia?
|
*deficient synthesis of structurally normal beta-globin chain
* alpha chains are still being synthesized -> therefore alpha chains combine to form unstable aggregates * leads to hemolysis |
|
|
what is B+ thalassemia?
|
1. reduced beta globin synthesis
(versus beta0 thalassemia=total absence of beta globin chains) |
|
|
why are skeletal abnormalities formed in beta thalassemia?
|
excessive hematopoiesis with marrow expansion encroaches upon cortical bone
(forms "halo" seen in Xray) |
|
|
describe the PBS in beta thalassemia
* what unique RBC type is seen here? |
hypochromic, microcytic anemia
*target cells are seen |
|
|
what is a very common complication of major beta thalassemia?
|
iron overload due to numerous blood transfusions
|
|
|
what treatment may cure beta thalassemia major?
|
bone marrow transplant
|
|
|
compare beta thalassemia minor (heterozygote) to beta thal major (homozygote)
|
beta thal minor: usually asymptomatic
beta thal major: very symptomatic, weekly transfusions required, fatal if untreated |
|
|
pathophysiology of alpha thalassemia?
|
reduced synthesis of alpha-globin chains due to deletion of a-globin genes. accumulation of beta chains forms unstable aggregates that precipitate and damage RBCs. Results in hemolysis.
|
|
|
describe what happens when:
1. one alpha chain is missing 2. two a-globin genes are missing 3. three a-globin genes are missing 4. four a-globin genes are missing |
1. silent carrier, asymptomatic
2. a-thallasemia trait, usually asymptomatic but see changes on PBS 3. Hemoglobin H disease - moderate to severe anemia, formation of hemoglobin H results in tissue hypoxia (hemoglobin H has a high affinity for oxygen) 4. Hyrops Fetalis (incompatible with life - all four a-chains missing) |
|
|
sickle cell disease is the result of a point mutation in the globin gene: what is the result of this mutation?
|
formation of hemoglobin S (HbS)
|
|
|
compare the HbS levels in:
1. homozygous SCD 2. heterozygous |
1. nearly all hemoglobin is HgS
2. about 40% is HgS |
|
|
what happens to the HgS molecule upon deoxygenation?
|
HgS aggregates and polymerizes, causing a "sickling phenomenon."
|
|
|
three characteristics of the "Sickling phenomenon?"
|
1. chronic hemolytic state
2. oxidative damage to sickled and normal appearing RBCs 3. microvascular occlusions |
|
|
are sickle cells seen in the PBS of heterozygous individuals?
|
No
(not under normal conditions) |
|
|
describe the changes to the spleen in the early and late phase of sickle cell disease.
|
1. early phase - spleen enlarged (trapping of sickle cells in splenic cords)
2. late phase - autosplenectomy (spleen becomes scarred and shrinks in size) |
|
|
what is seen in the bone marrow of sicke cell disease?
|
normoblastic hyperplasia
|
|
|
describe the vaso-occlusive crises seen in sickle cell disease.
|
episodes of ischemic necrosis, extremely painful, affect bones, lungs, liver, brain, penis, spleen
|
|
|
two things that can trigger an aplastic crisis in a person with sickle cell disease?
|
1. folate deficiency
2. parvovirus |
|
|
why do infants not manifest sickle cell disease until 5-6 months of age?
|
that is the age that fetal hemoglobin disappears.
|
|
|
why is that patients with Hemoglobin C/Hemoglobin S disease have milder courses than pure Hemoglobin S (sickle cell disease)?
|
* HgC crystallizes in the RBC leading to cellular dehydration and decreased deformity.
*reduced oxygen affinity improves delivery to tissue and decreases the effects of sickle cell disease. |
|
|
what (in general) goes wrong in immune mediated anemias?
|
antibodies directed against antigens on the RBC surface cause hemolysis.
|
|
|
what test is used for the diagnosis of immune mediated anemias?
|
Coomb's test
(tests for antibodies present on the RBC after serum is incubated with anti-human globulin) |
|
|
when would an indirect Coombs test be used?
|
when looking for antibodies that are not attached to RBC, just in serum
|
|
|
three types of immune mediated anemias?
|
1. Warm antibody type
2. Cold agglutination type 3. Cold hemolysins type |
|
|
what is the most common type of immune mediated anemia?
|
warm autoimmune hemolytic anemia
|
|
|
pathophysiology of autoimmune hemolytic anemia (warm)?
|
* IgG coats the RBCs but are not compliment fixing. Results in RBCs that are spheroidal shaped
|
|
|
2 etiologies of warm antibody type of immune mediated anemia?
|
50% idiopathic
drugs |
|
|
treatment for autoimmune hemolytic anemia (warm)?
|
immunosuppression
may require splenectomy |
|
|
what type of hemolysis occurs in autoimmune hemolytic anemia (warm)?
|
extravascular hemolysis (spleen)
|
|
|
which type of antibodies coat the RBCs in cold agglutination disease?
|
IgM - causes agglutination at low temperatures
*will fix compliment* |
|
|
type of hemolysis seen in cold agglutination disease?
|
extravascular or intravascular
|
|
|
cold agglutination disease may be idiopathic or secondary to infection. what two infectious agents have been linked to this?
|
EBV
mycoplasma |
|
|
why does agglutination not occur in Paroxysmal Cold Hemoglobinuria?
|
Abs are IgG type, not IgM.
|
|
|
pathophysiology of Cold Hemolysin Disease (Paroxysmal Cold Hemoglobinuria)?
|
IgG Abs attach to RBCs and bind compliment at low temperatures: when temperature rises, hemolysis occurs.
|
|
|
1. Cold Hemolysin disease(PCH) has historically been associated with what infection?
2. now it is more common with what? |
1. syphillis
2. viral infections |
|
|
what are the two settings in which alloimune hemolytic anemia can occur?
|
1. imcompatible blood transfusion
2. hemolytic disease of the newborn |
|
|
what is the difference between:
1. immediate hemolytic transfusion rxn and 2. delayed hemolytic transfusion rxn? |
1. immediate - due to major (ABO) incompatibility. usally fatal
2. delayed - due to minor red cell antigen, less serious |
|
|
which is more severe: ABO type or Rh type hemolytic disease of the newborn?
|
Rh type is more severe
(ABO incompatibility just produces mild hemolysis in the fetus) |
|
|
difference between microangiopathic and macroangiopathic hemolytic anemia?
(hint-both are intravascular hemolysis) |
1. microangiopathic - in capillaries (usually from turbulent flow pattern, DIC, TTP), also malignant HTN or march hematuria
2. macroangiopathies occur in large vessels. usally direct RBC trauma (ie. prosthetic valve) |
|
|
what type of hemolysis is seen in both micro and macroangiopathic hemolytic anemia?
|
intravascular
|
|
|
what types of RBCs are seen in the PBS of micro and macroangiopathic hemolytic anemia? (4)
|
1. schistocytes
2. burr cells 3. helmet cells 4. triangle cells |
|
|
what is Paroxysmal Nocturnal Hemoglobinuria (PNH)?
|
rare disorder involving disruption of synthesis of a RBC membrane component. this component regulates compliment activity: without is there is an increase in compliment mediated lysis
|
|
|
clinical s/s of PNH?
|
1. intermittent intravascular hemolysis (intermittent hematuria)
2. venous thromboses 3. infection |
|
|
what type of infection could cause hemolysis?
|
plasmodium
|
|
|
why would severe thermal burns cause hemolytic anemia?
|
heat from burn disrupts RBC membane
PBS shows schistocytes, microspherocytes |
|
|
why is it not possible to estimate blood loss from trauma or surgery with a CBC immediately after the event?
|
no change in CBC for the first few hours
(HGB and HCT stay the same) |
|
|
describe the CBC changes hours after an acute blood loss
|
normochromic, normocytic anemia (decreased RBCs)
-increase in WBC |
|
|
why is reticulocytosis seen in a CBC days after an acute blood loss?
|
erythropoietin was increased to increase RBC production (rxn. to blood loss).
|
|
|
what is polycythemia vera?
|
increase in RBCs (and a corresponding increase in hemoglobin)
|
|
|
what is the cause of relative polycythemia?
|
hemoconcentration because of a decrease in plasma volume (ie. dehydration)
|
|
|
causes of absolute polycythemia? (2)
|
1. abnormality of precursor lines
2. increase in EPO levels (could be due to hypoxia or EPO secreting tumor) |
|
|
what are the 3 microcytic anemias?
|
1. iron deficiency anemia
2. thalassemia 3. anemia of chronic disease |
|
|
what are the 6 normocytic anemias?
|
1. aplastic anemia
2. pure red cell aplasia 3. anemia of chronic disease 4. myelophthisic anemia 5. chronic renal failure 6. acute blood loss |
|
|
what are the 4 macrocytic anemias?
|
1. megaloblastic anemias (B12 and folate deficiency)
2. reticulocytosis 3. liver disease 4. hypothyroidism |
|
|
HIV belongs to which:
1. family 2. genus |
1. retroviridae
2. lentivirus |
|
|
what are the two kinds of HIV and what is the difference between the two?
|
1. HIV-1 ("one we are worried about")
2. HIV-2 (in Africa) |
|
|
describe the virion and genome of HIV
|
enveloped virion
(+)ssRNA diploid genome |
|
|
what is the viral attachment protein called and where on the virus cell is it found?
|
gp120
seen as spikes on the virion surface |
|
|
is the reverse transcriptase host or virion associated?
|
virion associated
|
|
|
retroviruses typically have three gene groups. what does each of the following groups encode?
1. env 2. pol 3. gag |
1. transmembrane and surface glycoproteins
2. reverse transcriptase, integrase, protease 3. group antigens (matrix and capsid proteins) |
|
|
which two HIV proteins are produced as a polyprotein?
|
gag and pol
|
|
|
regarding HIVs accesory genes:
function of... 1. tat and rev? 2. vpu, nef 3. vpr |
1. regulatory genes
2. downregulates CD4 production 3. blows holes in host cell nucleus, allowing virus to enter |
|
|
during infection, the gp120 protein binds to the CD4 molecule. Conformational changes allow binding to the coreceptor. what are the two possible coreceptors?
|
CCR5
CXCR4 |
|
|
which glycoprotein mediates fusion of the viral envelope with the cell membrane?
|
gp41
|
|
|
describe the synthesis of the dsDNA strand and the long terminal repeats from the viral RNA genome.
|
* reverse transcriptase makes a dsDNA copy of the RNA genome
*as DNA genome is synthesized, RNA genome is being degraded *the end portion of the RNA genome is saved and duplicated in the dsDNA - it is these RNA segments that generate LTRs. |
|
|
what enzyme integrates the viral dsDNA into the host DNA?
|
viral integrase
|
|
|
what is the integrated copy of the viral genome called?
|
provirus
|
|
|
what types of signals are contained in the LTRs?
|
promoter and other regulatory signals
|
|
|
what happens to the provirus?
|
cellular RNA polymerase synthesizes viral mRNA
|
|
|
why does HIV rapidly acquire resistance to antiviral drugs?
|
the HIV reverse transcriptase is highly error prone. combined with the vast numbers of progeny, resistance is easily acquired
|
|
|
regarding transcription of the provirus and regulatory protiens:
1. tat encourages production of _____ mRNA 2. rev encourages production of ____mRNA |
1. spliced
2. unspliced |
|
|
what is a critical thing that must occur in order for the HIV virus to maturate?
|
protease cleavage of the polyprotiens
|
|
|
what is the incubation period in a primary HIV infection?
|
2-4 wks
|
|
|
in a primary HIV infection, how long does it take for the symtoms to resolve?
|
1 month
|
|
|
on average, how long is the asymptomatic period that follows a primary HIV infection?
|
8-10 yrs
|
|
|
in a primary HIV infection: what type of cells carry the virus from the mucosal surface to the lymph nodes?
|
dendritic cells
|
|
|
during the "latent period," what exactly is latent?
|
clinical symptoms are latent
VIRAL REPLICATION IS NOT (viral replication continues at a very high rate, immune system can keep up) |
|
|
most common test for diagnosis of HIV is?
|
ELISA
|
|
|
how fast after infection can the ELISA test detect HIV?
|
6-12 weeks
|
|
|
what is done as a confirmatory test to exclude false positives? (3 methods)
|
*Western Blot (to env, gag or pol proteins)
*immunoflourescence *PCR |
|
|
basic MOA of:
1. nucleoside RT inhibitors 2. non-nucleoside RT inhibitors 3. protease inhibitors 4. fusion inhibitors |
1. chain terminators
2. interfere with RT 3. prevent viral maturation 4. prevent viral entry |
|
|
what are the 3 most common combinations used in HAART therapy?
|
1. two nRTIs plus one PI
2. two nRTIs plus one NNRTI 3. two nRTI plus ritonovir plus another PI |
|
|
HAART still has a ___% failure rate?
|
20%
|
|
|
adverse effects of PIs? (2)
|
1. visceral fat accumulation
2. insulin resistance |
|
|
adverse effect of nRTIs?
|
lipoatrophy
|
|
|
what are the three primary blood cell components and therefore the targets of hematinic therapy?
|
1. erythrocytes
2. platelets 3. granulocytes (leukocytes) |
|
|
in regards to regular hematopoiesis, what is the function of:
1. SCF (stem cell factor) 2. erythropoietin 3. IL-12 4. GM-CSF 5. G-CSF 6. M-CSF |
1. stimulates pluripotent stem cells
2. stimulates proliferation of erythroid progenitors ultimately increasing RBC production 3. promotes differentiation and maturation of all cell lines 4. stimulates maturation of granulocytes 5. stimulates maturation (mainly) of neutrophils 6. stimulates maturation of macrophages |
|
|
sargramostim is the pharmacological equivalent to which colony stimulating factor?
|
GM-CSF
|
|
|
filgrastim is the pharmacological equivalent to which colony stimulating factor?
|
G-CSF
|
|
|
oprevlekin is the pharmacological equivalent to which stimulating factor?
|
IL-11
|
|
|
which HIV drug is well known for causing neutropenia?
-treatment? |
zidofudine (HIV drug)
- GM-CSF, G-CSF, EPO (if global anemia is induced) |
|
|
where is 90% of EPO made?
|
in the peritubular cells of the renal cortex
|
|
|
1. dietary iron is in what form?
2. iron is best absorbed in what form? |
1. Ferric (Fe3+)
2. Ferrous (Fe2+) (the stomach acid reduces ferric to ferrous) |
|
|
where is iron primarily absorbed?
|
duodenum
|
|
|
two anemias that cause iron defiency?
|
1. iron deficiency anemia
2. anemia of chronic disease |
|
|
how do you differentiate between iron deficiency anemia and anemia of chronic disease?
hint: serum iron and TIBC values |
1. iron deficiency: serum iron is low and TIBC is elevated
2. anemia of chronic disease: serum iron is low and TIBC is low |
|
|
serum ferritin is proportional to?
|
total iron stores in the body
|
|
|
why should iron be taken on an empty stomach?
|
food decreases the absorption
(less stomach acid to reduce ferric to ferrous) |
|
|
most adverse effects to iron therapy are?
|
GI
|
|
|
two drugs that are known to cause a folic acid deficiency?
|
phenytoin
phenobarbital |
|
|
ideal treatment for:
1. severe chronic neutropenia? 2. myelodysplastic syndrome that has a low WBC count? 3. anemia of chronic disease? 4. bone marrow suppression (ie. aplastic anemia) 5. thrombocytopenia? |
1. G-CSF (possibly +GM-CSF)
2. GM-CSF 3. EPO and iron 4. G-CSF and GM-CSF 5. IL-11 |
|
|
which has the adverse effect of capillary leak syndrome?
|
G-CSF, GM-CSF
|
|
|
which has the adverse effect of papilledema, fluid retention, transient atrial arrythmias?
|
IL-11
|

