![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
19 Cards in this Set
- Front
- Back
|
Three particles of an atom |
1. Proton - positive charge, same number as electrons, make up nucleus. 2. Neutrons - neutral charge, Atomic mass = number of neutrons, make up nucleus. 3. Electrons - negative charge, surrounds atoms nucleus. |
|
|
What happens when NaOH and HCl are dissolved in water |
NaOH+HCl = Salt(NaCl)+H2O |
|
|
PH Scale |
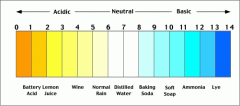
1-14, 7 is neutral
|
|
|
Logorithmic |
PH Scale is logarithmic, 1 - 2 - 3 - 4... x10 x10 x10 |
|
|
Role of Enzymes in living things |
change the rate of chemical reactions in the body |
|
|
Importance of water in living things |
All chemical processes occur in water(in living things) |
|
|
Water molecule's structure |
H2O |
|
|
Special properties of water |
- expands when frozen - freezes from the top first |
|
|
Dehydration Synthesis |
The making of something by losing water in the equation |
|
|
Hydrolysis |
The making of something by adding water to the equation |
|
|
Substances that are acids |
PH under 7, vinegar, HCl... |
|
|
Substances that are bases |
PH over 7, soap, NaOH, ammonia… |
|
|
What happens when acids and bases are a) mixed together b) placed in water |
a) Produce Salt b) Produce Water |
|
|
Chemical activity of an atom is determined by... |
electron arrangement |
|
|
Metals vs. Non-metals |
Metals - lose electrons Non-metals - gain electrons |
|
|
Cation vs. Anion |
Cation - ion with positive charge Anion - ion with negative charge |
|
|
Characteristics of Acids |
Sour, PH under 7 |
|
|
Characteristics of Bases |
Bitter, PH over 7 |
|
|
Saturated vs. Unsaturated Fats |
Saturated Fat - Fully surrounded and unable to connect with other substances Unsaturated Fat - Open to mixing with other substances |

