![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
39 Cards in this Set
- Front
- Back
|
Briefly describe Carl Woese’s contribution to the classification of microorganisms. Be
sure to mention what technologies Woese and his colleagues took advantage of and what component of cells they compared to make their decisions about the relative relatedness of different species. |
Using nucleic acid technologies that determined the sequence bases in ribosomal RNA (rRNA), Carl Woese and his colleagues compared a wide variety of organisms from which they concluded that prokaryotes once thought to be part of the plant kingdom are as different from other prokaryotes as they are from eukaryotes, thus separating cells into the current three domains of Eurkaryotes, Prokaryotes, and Archaea.
|
|
|
List two phenotypic methods that can be used to identify microorganisms and two
genotypic methods that can be used to identify prokaryotes. |
Phenotypic Methods to ID Microorganisms:
a- Microscope Morphology b- Metabolic Differenes Genotypic Methods to ID Prokaryotes: a- DNA probes b- Polymerase Chain Reaction (PCR) |
|
|
You are given an unknown sample containing a pure culture of a single microorganism.
Based on Table 10.2, name two cell features you could examine that would allow you to unambiguously determine in which of the three domains the unknown microorganism belongs. Explain why the features you chose would make this unambiguos identification possible. |
a- Peptidoglycan Cell Wall
–> If it is present the cells are bacteria. If peptidoglycan is not present in the cell wall than cells are either Archaea or Eucarya. |
|
|
You are given an unknown sample containing a pure culture of a single bacterial strain.
List three biochemical tests you might perform to learn more about the unknown bacterial strain. Be sure to explain what each biochemical test will allow you to learn about the unknown strain. |
a- Catalase Test –> detects catalase enzyme activity by forming BUBBLES when HYDROGEN PEROXIDE is BROKEN DOWN TO O2 and WATER.
–>Nearly all bacterial that grow in presence of Oxygen, excluding lactic acid bacteria, are catalase positive. b- Methyl Red –> a pH indicator that turns RED in presence of MIXED ACIDS, the characteristic end products of a particular fermentation pathway. –> Distinguishes between bacteria that use or do not use this pathway |
|
|
You are the chief clinical microbiologist at a major hospital. Samples from two patients,
both suffering from Escherichia coli 0157:H7 infections are sent to your lab. Describe two methods your team would employ to characterize the two samples in order to determine whether or not they were both the identical strain of Escherichia coli 0157:H7 (suggesting food poisoning from the same source) or two differnt strains of Escherichia coli 0157:H7 (suggesting food poisonon from two independent sources). Hint: use Table 10.5. |
a- Serological Typing: to determine if the samples contain the same proteins & carbohydrates
b- Pulse-Field Gel Electrophoresis: (Genomic Typing) The enzyme-digested DNA will make patterns in the gel that can be used to determine whether or not the 2 samples are identical strains. |
|
|
Why is sequencing of the 16S ribosomal gene used as the standard for determining the
relatedness of species of microorganism? |
(Page 255)
a- their function is so highly constrained & essential that the nucleotide sequence changes that can occur while still allowing the ribosome to operate are limited b- the 5S molecule is too small & the 23S molecule is too large to serve the same function c- the regions that do vary are so different they can be used to identify an organism. |
|
|
What is the difference between oxygenic phototrophs and anoxygenic phototrophs? Give
an example of a microorganism from both classes. |
Anoxygenic Phototrophs:
- Use hydrogen sulfide or organic compounds rather than water as a source of electrons when making reducing power in the form of NADPH. - Do not produce O2.. - Have bacterioclorohpyll that absorb wavelength of light not used by other bacteria - Purple sulfur bacteria (ex Chromatium) , purple non-sulfur bacteria (ex Rhodobacter), green sulfur bacteria (ex Chlorobium), green non-sulfur bacteria (ex chloroflexus). Oxygenic Phototrophs: - Use water as a source of electrons for reducing power. - Produce O2. - Use sunlight to convert CO2 into organic compounds.- Cyanobacteria - Have phycobiliprotiens that absorb wavelengths of light chlorophyll doesn't - Nitrogen-fixing cyanobacteria (ex Anabaena azaollea) |
|
|
Name two genera of bactertia that contain species capable of forming endospores.
Why/when might a bacterium benefit from forming an endospore? |
Bacillus and Clostridium. These can form endospores to survive under extreme conditions (heat, toxicity, UV radiation) – not in detail in the book
|
|
|
Give an example of a soil microorganism that forms a symbiotic relationship with plants.
What do the bacteria gain from the symbiotic relation ship, and what does the plant gain from the symbiotic relationship. |
Rhizobium, live in root nodules of legumes. The plant synthesizes the protein leghemoglobin which binds O2 and regulates its concentration in the nodule protecting the O2 -sensitive nitrogenase by keeping free O2 at a low level. Bacterioids fix nitrogen releasing ammonia that then diffuses into root cell where it is assimilated into amino acids for use by the plant. In return, the bacteria receive nutrients from plant. How do plants benefit? – Pg 779.
|
|
|
You are an oceanographer and have recently discovered what you think is a new species
of fish. This unusual species of fish has several “light organs” that glow brightly when the fish come out at night to feed. You transport the fish back to your lab in Brooklyn. To be safe, you add a broad spectrum antibiotic to the storage tank in which you are transporting the fish, to make sure the tank does not become contaminated in the long trip home. The fish arrive safely and healthy to your lab, but the first night you notice that their “light organs” are no longer glowing brightly. What might explain this? |
The bioluminescence is caused by bacteria living within these "light organs". Adding an antibiotic kills the light emitting bacteria and the organs no longer glow.
|
|
|
You have just returned to your lab from Yellowstone National Park where you collected
several bacterial species from the hot, acidic, natural springs there. You use PCR to amplify several enzymes involved in DNA replication from the species isolated in Yellowtone National Park. You clone these genes into a laboratory strain of E. coli. You then test the activity of these enzymes and find, very surprisingly, little or no enzymatic activity. Explain why this might be. |
-Enzymes are only activated at high temperatures
- Enzymes require a more acidic environment than E. coli resides in / is being tested in |
|
|
Why is a virus or bacteriophage not considered a living organism? Give an approximate size
range that most phage would fall in. |
because they are non-living entities that infect all forms of life, including all members of the bacteria, archaea, and eucaryea. the approximate size range that most phage would fall in is 10-500nm.
|
|
|
The genetic material of bacteriophages can be divided into four distinct types. What are the four
types? |
double-stranded DNA, double-stranded RNA, single-stranded DNA and single-stranded RNA.
|
|
|
List the six steps of the life cycle of bacteriophage T4 in E. Coli(see figure 13.5). Make sure to
provide these steps in the sequence that they occur and how each step makes the infection cycle of the phage possible. |
Step1, Attachment: the phage attach to specific receptors on the cell wall of E.coli.
Step2, Penetration: after attachment, phage DNA is injected into the bacterial cell, leaving the phage coat outside Step 3, Transcription: Phage DNA is transcribed, producing phage mRNA, which is translated to phage proteins. Step 4, Replication of phage DNA and synthesis of proteins: phage coat proteins, other protein components, and DNA are produced separately. Host DNA degraded. Step 5, Assembly: Phage components are assembled into mature virions. Step 6, Release: The bacterial cell lyses and releases many infective phage. |
|
|
Draw and label the components of the T4 bacteriophage. Where would you expect to find phage
proteins that recognize specific host bacterial receptor sites? |
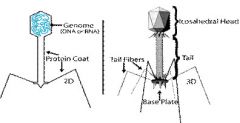
b) you can expect to find phage protiens that recognize specific host bacterial receptor sites in the replication of T4 Bacteriophage in E.Coli.
|
|
|
Give an example of a lytic phage, a lysogenic phage, and a filamentous phage. How do their
replication cycles within a host bacterium differ? |
Lytic phage- T4 bacteriophage, Lysogenic phage- phage-encoded tRNA,
Filamentous phage- M13. B) the difference is the Lytic phage is the phage that lyse the cell, the filamentous phage is phages that takeover only some of the metabolic processes of the cell, while a Lysogenic phage is a phage that does not go into a lytic cycle but instead either integrates into the host bacteria's chromosome or "lives" as a stable plasmid within the host cell. |
|
|
Provide a detailed description of the life cycle of λ phage. Make sure you clearly explain how the phage DNA integrates into the host chromosome, how the prophage state is maintained, the
process of phage induction, and the how the prophage prevents re-infection of the bacterium by another λ phage. |
λ phage(lambda phage) is a temperate bacteriophage that infects Escherichia coli. Once the phage is inside its host, it may integrate itself into the host's DNA. In this state, λ is called a prophage and stays resident within the host's genome, without apparent harm. Thus the prophage is duplicated with every cell division of the host. This is referred to as the "lysogenic pathway". The DNA of the prophage that is expressed in that state codes for proteins that look out for signs of stress in the host cell. Stress can be from starvation, poisons (like antibiotics), or other factors that can damage or destroy the host. At that point, the prophage reactivates, excises itself from the DNA of the host cell and enters its lytic cycle. The reactivated phage takes apart the host's DNA and produces large amounts of its own mRNA, so as to produce prodigious amounts of phage units. When all resources of the host are depleted from building new phages, the host cell is lysed (the cell wall is broken down), and the new phages are released.
|
|
|
Describe lysogenic conversion. Give an example of two common pathogens that result from
lysogenic conversion. |
Lysogenic conversion is when a temperate phage induces a change in the phenotype of the bacteria infected that is not part of a usual phage cycle. Changes can often involve the external membrane of the cell by making it impervious to other phages or even by increasing the pathogenic capability of the bacteria for a host.
b) Corynebacterium diphtheriae produces the toxin of diphtheria only when it is infected by the phage β. In this case, the gene that codes for the toxin is carried by the phage, not the bacteria. |
|
|
Describe how specialized and generalized transduction differ? Provide a figure to illustrate the
difference between specialized and generalized transduction. |
The difference is in generalized transduction, any bacterial gene can be transferred, while in specialized transduction only a few specific genes can be transferred. View figure 13.11 on p.331
|
|
|
Describe the restriction modification system and what role it serves for a bacterium.
|
restriction modification system is used by bacteria, and perhaps other prokaryotic organisms to protect themselves from foreign DNA, such as bacteriophages. Bacteria have Restriction enzymes, also called restriction endonucleases. These cleave double stranded DNA at specific points. The point of cleavage depends on the restriction enzyme. This effectively destroys the foreign DNA, which is then degraded further by other endonucleases. This phenomenon is called restriction.Restriction enzymes are highly specific. They only cleave at specific points. They recognize specific sequences of DNA which are usually 4-6 base pairs long, and often palindromic. Given that the sequences they recognize are very short, the bacterium itself will almost certainly have many of these sequences present in its own DNA. Therefore, the host must have a system to protect its own DNA. It achieves this by modifying it. It adds methyl groups to it to block the action of the restriction enzymes.
|
|
|
Describe the stages in the generalized life cycle for herpes
simplex virus type I infection of a typical eukaryotic host cell. |
This is a latent infection which means they are persistant infections in which a symptomless period is followed by reactivation of the virus with accompanying symptoms. HSV-1’s Initial infection might not lead to any symptoms, but cold sores and fever blisters, which is the initial acute infection, then it infects the sensory nerve cells, where it remains in a non-infectious form without causing symtoms of disease then could be activated by condition as menstruation, fever, or sunburn.
|
|
|
Define the following six terms related to the cultivation of
host cells that one would use to grow and maintain viruses: tissue culture, primary culture, monolayer, cytopathic effect, inclusion body, and lysate. |
Tissue culture: culture of plant or animal cells that grows in an enriched medium outside the plant or animal.
Primary Culture: cells taken and grown directly from the tissue of an animal Monolayer: is a single layer of cells. Cytopathic effect: observable change in a cell’s appearance produced by virus that is propagated in tissue culture cells. Inclusion body: this is when the cytopathic effect is localized to particular sites within infected cells. Lysate: this is liquid containing the virions. |
|
|
Describe two methods that are typically used to quantify
virus concentrations. |
a)Plaque Assay: which involves determining the number of viruses in solution by adding a known volume of the solution to actively metabolizing cells in a Petri dish. The infection, lysis, and subsequent infection of surrounding cells leads to a clear zone or plague surrounded by the uninfected cells. Each plaque represents one virion, initially infecting one cell.
b) Counting of Virions with the Electron Microscope: which involves pure preparations of virions, their concentration may be readily determine by counting the number of virions in a specimen prepared for the electron microscope. This method distinguish between infective and non-infective virions. From their shape and size, it also helps find the identity of the virus. |
|
|
Describe the steps required for retroviruses, such as HIV,
to transcribe their RNA genome into viral proteins. Make sure to list the critical and unique properties of the reverse transcriptase enzyme. |
The steps for retroviruses to transcribe their RNA genome into viral proteins is by by first having the reverse transcriptase make DNA:RNA and then DNA:DNA double helix. after there’s integration of DNA copy into host chromosome, then there’s transcription of viral genes into many RNA copies. The RNA is then transcribed by the host cell RNA polymerase to produce one long mRNA molecule that contains all of the viral information. This mRNA molecule is translated into long polyprotein, which is then cleaved by a viral-encoded protease to yield the individual proteins. Following cleavage, the individual proteins fold to their proper conformation resulting in the proteins that make up the virion.
|
|
|
Describe in detail the process of influenza virus adsorption and entry to a host cell, and the exit of newly formed viruses from the host cell.
|
The process of Influenza virus, is the protein which recognizes a target protein on the cell, changes its shape when it contacts the host cell. Following fusion, the nucleocapsid is released directly into the cytoplasm, where the nucleic acid separates from the protein coat.
|
|
|
What mechanisms explain how some viruses can be associated with cancer? List three viruses that are associated with cancer, and name the type of cancer with which they are associated.
|
The mechanism that explains how viruses can be associated with cancer is integration of all or part of the virus genome into the host chromosome. Following integration the transforming genes or oncogenes are expressed, resulting in uncontrolled growth of the host cells.
|
|
|
Define the following terms relating to types of viral infections: acute infection, latent infection,
persistent infection, chronic infection. |
Acute Infection: are usually of relatively short duration, and the host organism may develop long-lasting immunity.
Latent Infection: are pesistant infections in which a symptomless period is followed by reactivation of the virus with accompanying symptoms. Persistent Infection: the viruses are continually present in the body and remains in the body for long periods of time, often without causing symptoms of disease. Chronic Infection: Infections that develop slowly and persist for months or years. |
|
|
Describe the structure, organization, and role of the viral coat proteins. How do enveloped viruses differ from non-enveloped viruses? Starting from initiation of synthesis at the ribosome,
describe the pathway of virally coded spike proteins, such as neuraminidase and hemagglutinin, and how become incorporated into the viral envelope? |
The viral capsid together with the nucleic acid packed within the protein coat is called the nucleocapsid. The capsid is composed of many identical protein subunits called capsomeres.
b.) They have an additional double layer of lipids covering over the capsid protein & just inside this layer is a matrix protein not found in naked virusues. |
|
|
Give examples of two prion diseases that affect humans. What is unique about the infectious mechanism of prion diseases? What host cell type do most prion diseases identified to date target?
|
a.)Mad-cow disease & Creutzfeldt-Jakod disease.
B.)How can they replicate if they do not contain any nucleic acid. c.)Neurons and brain tissue. |
|
|
After obtaining your PhD in virology, you begin work at a pharmaceutical company in their HIV division. In your introduction to the HIV division your co-workers ask you what you think are the three best targets in HIV for desiging potential anti-HIV drugs against. List the three targets you suggest and why they make good targets for drug therapies against HIV.
|
1. Inhibitors to prevent the protease from yielding any viral proteins.
2. Developing a medicine to degrade the reverse transcriptase because without this enzyme, the HIV RNA genome can’t be converted into a double-standed DNA & be integrated into the host chromosome. 3. Can’t think of a 3rd target. |
|
|
Describe what has recently been learned about the 1918 “Spanish flu” strain, and how this new
information was obtained. |
- By sequencing the RNA genome of various strains of the influenza virion, and observing the tissues obtained from the preserved bodies.
- The hemagglitinin gene of the Spanish flu virion originated by recombination between a swine influenza virion and a human-lineage virion. The end of the protein that binds to receptors on the host cell was encoded by the swine-lineage influenza, whereas the rest of the molecule was encoded by a gene that came from human-lineage influenza. |
|
|
The observation that viruses can agglutinate red blood cells suggests to some people that both
viruses and red blood cells must have multiple binding sites. Is this a good argument? Why or why not? |
Yes because the only reason for viruses to clump red blood cells is because they interact with the surfaces of the cells.
|
|
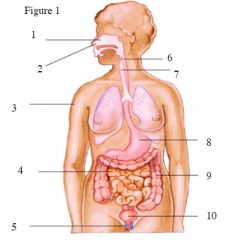
Using the numbers in figure 1,
name the first-line physical barrier being pointed to and how each first-line defense acts to prevent infection. |
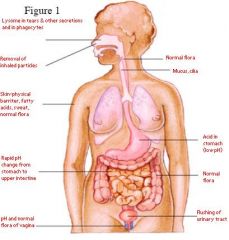
Answer:
|
|
|
List six different types of immune cells and explain their basic function in immune responses. Where in the adult human do leukocytes derive?
|
1-Red Blood Cells carry oxygen in the blood.
2-Platelets important for blood clotting 3-White Cells important in all host defenses 4-Granulocytes 5-Lymphocytes 6-Mononuclear Phagocytes 7-Dendritic Cells Found in the Hematopoietic stem cell in bone marrow. |
|
|
What are the five signs associated with inflammation? What is the term for the process by which neutrophils migrate out of the blood vessels and into local connective tissue at the site of an infection?
|
Five signs associated with inflammation are swelling, redness, heat, and loss of function.
The term is called diapedesis |
|
|
What are three independent pathways (direct or indirect) that complement proteins contribute to in order to fight off bacterial pathogens? This question is not asking for three different pathways
for activation of complement factors, but rather three different ways in which complement proteins can function to fight off bacterial pathogens. |
*Inflammation- complement components C3a and C5a a potent chemoattractant drawing phagocytes into the area where complements activated induce changes in endothelial cells that line the blood vessels and in mast cells.
*Lysis of foreign cells- complexes of C5b, C6, C7, C8 and multiple C9 molecules spontaneously assemble in the membranes of the cells, forming doughnut shaped structures each called a membrane attack complex(MAC). *Opsonization- complement protein C3b binds to foreign material –phagocytes more easily “grab’ particles coated with C3b because phagocytic cells have receptors for the molecule on their surface. |
|
|
Describe the steps involved in the phagocytosis and killing of a bacterial pathogen by a
macrophage. |
*Chemotaxis- cells are recruited to the site of infection or tissue damage by certain stimuli that acts as chemoattractants.
* Recognition and attachment- cells use various receptors to bind invading microbes either directly or indirectly. Direct binding occurs through receptors that recognize patterns associated with compounds found on microbes. Indirect binding occurs when a particle has first been opsonized dramatically enhancing the phagocytes’ ability to attach and engulf the material. *Engulfment- cell engulfs invader forming a membrane bound vacuole called a phagosome. *Fusion of the phagosome with the lysosome- the phagosomeis transported along the cytoskeleton to a point where it can fuse with lysosomes, membrane bound filled various digestive enzymes, including proteases and lysozyme. *Destruction and digestion- within phagolysosme oxygen consumption increases enormously as sugars are metabolized via aerobic respiration with the production of highly toxic oxygen products such as superoxide, hydrogen peroxide, singlet oxygen and hydroxyl radicals. *Excoytosis- following the digestion of the microorganisms the membrane bound vesicle containing the digested material fuses with the plasma membrane. (*)When these receptors are triggered the macrophage produces pro inflammatory cytokines to alert and stimulate various other cells of the immune system. Macrophages can increase their otherwise limited killing power with the assistance of certain T cells to become activated macrophages. This an example of the cooperation between the innate and adaptive host defenses. |
|
|
Interferons may be secreted by a virally infected cell. These interferons then trigger both the production of mRNA degrading enzymes and coordinated cell death (apoptosis) in adjacent cells
that may become infected by the virus. If new virions are released from the originally infected cell and go on to kill adjacent cells any ways, what is the value of triggering cell death in adjacent cells via the interferon-induced route? |
*When cells bind interferon only the infected ones are sacrificed. Their uninfected counterparts remain functional, but are poised to cease vital functions should they too become infected.
|
|
|
Describe the pioneering work and contribution to the field of immunology made by Elie
Metchnikoff. |
Elie Metchnikoff theorized that specialized cells within the body destroyed invading organisms. His ideas rose from observations he made while studying the transparent immature larval in starfish in Sicily in 1882. He observed the larvae under a microscope and could see ameba-cells within the bodies. He reasoned that certain cells present in animals were responsible for ingesting and destroying foreign material.
|

