![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
22 Cards in this Set
- Front
- Back
|
Carbohydrates |
(CH20)n; smallest units of these are monosaccharides and are also known as simple sugars (ex. glucose and fructose). Monosaccharides can join together to form disaccharides (2), oligosaccharides (several), or polysaccharides (thousands) of simple sugars. |
|
|
Lipids |
diverse group that have low or no solubility in water. Common ones are steroids, phospholipids, and fats and oils. Saturated if tails carry max number of hydrogen atoms. |
|
|
Proteins |
long chains of amino acids, thousands of chains make up proteins once folded in 3D structure. Formed through peptide bonds between amino acids. |
|
|
Amino Acid Structure |
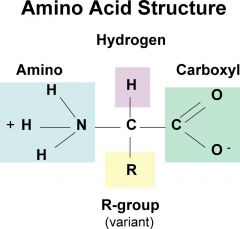
|
|
|
Nucleic Acids |
DNA and RNA. DNA is instructions for constructing each organism on Earth. Made by double helix of two nucleotide monomers. Contains 5 carbon sugar (deoxyribose), phosphate group, and nitrogenous base. |
|
|
Purine Ring |
Guanine and Adenine. Both contain two interlocking rings. |
|
|
Pyrimidine Ring |
Cytosine and Thymine. Only have one ring. |
|
|
Phosphodiester Bond |
the bond linking nucleotides. |
|
|
Positive vs. Negative Control |
Positive control = a response is expected. Negative control = no response is expected (water in this experiment). |
|
|
Iodine Test |
positive result is deep blue-black colour. Tests for starch. |
|
|
Benedict's Test For Reducing Sugars |
when heated with sugars that contain aldehyde or ketone functional groups, this causes Cu2+ to reduce to Cu+ thus changing the colour of solution. Sucrose and Starch do not test positive though because they are consumed during the formation. Positive test = green (weak), yellow (stronger), orange, brown, or red (very strongly positive) precipitate. Used to test for sugars in diabetes urine. |
|
|
Biuret Test for Proteins |
detects bonds that form between amino acids (peptide bonds). Turns solution violet if positive. |
|
|
Sudan IV Test for Lipids |
Soluble in lipids. When it dissolves, is causes the solution to turn pink or red with flakes. |
|
|
Acid Hydrolysis |
breaking down macromolecules into their monomers in the presence of heat and acid. |
|
|
Electrophoresis |
separating a mixed population of molecules to isolate only one part desired. Charged molecules move through a gel-like matrix under the influence of an electric field. |
|
|
Sieving |
Shorter DNA molecules move faster and migrate farther than longer ones because shorter molecules migrate more easily through the pores of the gel. |
|
|
Agarose |
sieving gel made from red algae. |
|
|
Hydrogen Ion Concentration |
usually expressed in negative exponents of ten. |
|
|
pH Scale |
designed to remove negative exponents for hydrogen ion concentration measurements. Neutral is 7 pH. Below 7 acidic. Above 7 basic. |
|
|
Strong Acid / Base |
a substance that will completely dissociate in water. |
|
|
Weak Acid / Base |
will reach equilibrium in which some of the molecules are dissociated but other not. |
|
|
Buffers |
maintain pH of a solution by minimizing changes in the concentration of hydrogen ions. Does not make solution neutral, only used to maintain current pH. |

