![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
26 Cards in this Set
- Front
- Back
|
Radioactivity is... |
the release of high energy radiation from a radioactive source. |
|
|
Occurs when... |
changes happen in the nuclei |
|
|
what are the 6 types of radiation |
x-rays, gamma rays, microwaves, ultraviolet waves, radio waves, infrared waves |
|
|
Which is the most dangerous |
gamma rays |
|
|
isotopes are... |
different types of one element that have the same number of protons, different number of neutrons |
|
|
atomic # of an isotope never... |
changes |
|
|
istopes have different... |
mass numbers |
|
|
mass #= |
#protons-#neutrons |
|
|
(2 ways) #neutrons = |
mass # -#protons or mass#- atomic# |
|
|
different isotopes of an element have the same ------1 but different-------2 |
1. atomic # 2. mass #'s |
|

|

|
|
|
in Cl-35 what does the 35 represent |
atomic mass |
|
|
when a radioactive elements emits radiation... these elements have nuclei that are... |
a change occurs in its nucleus and becomes a new element. unstable. |
|
|
To become more stable an element must |
emit radiation |
|
|
positively charged |
alpha |
|
|
negatively charged |
beta |
|
|
neutral particles |
gamma rays |
|
|
symbols that represent alpha particles |
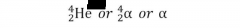
|
|
|
which particle cant penetrate a peice of paper |
alpha |
|
|
beta symbols are |
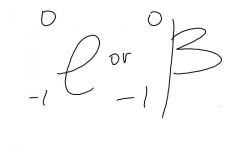
|
|
|
which particle can be blocked by aluminum foil |
beta |
|
|
in beta decay... |
neutron is changed into a proton and an electron, electron is emitted so with one more proton it becomes a new element with same mass number |
|
|
gamma ray symbols |

and look for a * for high energy |
|
|
no mass or charge are which particle |
gamma |
|
|
thick blocks of concrete or lead block which particle |
gamma |
|
|
omit |
omit |

