![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
6 Cards in this Set
- Front
- Back
|
Physical changes |
-No new substances form |
|
|
Chemical changes |
-New substances are created. -Atoms rearrange creating new chemical bonds -Mass is conserved! -Change occurs in the electron cloud region of the atom -Signs of a chemical reaction occur: 1) New colors, new odors, large energy changes 2) Bubbling without heating- New gas produced -Examples: tarnishing, oxidizing, burning wood, |
|
|
Nuclear changes |
-Some atoms are destroyed while new atoms are created. -Change occurs in the nucleus - Some mass is converted to energy. - Huge amounts of energy is released! - Examples: fission of uranium, radioactive decay of lead-22, fusion of hydrogen to make helium on the sun. |
|
|
More on Fusion & Fission |
- Nuclear Fusion occurs when 2 or more smaller nuclei join together. -Nuclear Fusion occurs in the sun and in other stars. - Nuclear Fission occurs when an atom get split. -Opposite of nuclear fusion - Nuclear Reactions change atomic potential energy to thermal energy(heat) and radiant energy(light). |
|
|
Nuclear vs. Chemical |
- If a reaction is Nuclear... less than 0.1% of the mass initially present is transformed into other forms of energy. Uses E=mc^2 - If a reaction is chemical... The percentage is much less So small that practically zero mass is converted to energy ----Follows law of conservations of mass |
|
|
Exothermic VS. Endothermic |
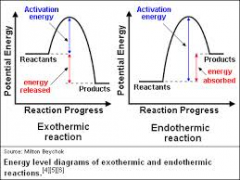
|

