![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
112 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Vital Force |
Thought to be the reason organic compounds could not be synthesized in a lab |
|
|
|
Organic Chemistry |
The study of carbon compounds |
|
|
|
Carbon |
Group 4A element that can share 4 valence electrons and form 4 covalent bonds |
|
|
|
Atom Structure |
Made of a positively charged nucleus surrounded by negatively charged electron cloud |
|
|
|
Atom diameter |
2 × 10 ^-10 or 200 picometers (pm) |
|
|
|
Angstrom |
represents the unit Aº= 10^-10 m or 100 picometer |
|
|
|
Atomic number (Z) |
the number of protons in the atom's nucleus; the same for all atoms of a given element |
|
|
|
Mass Number (A) |
The sum of protons & neutrons |
|
|
|
Isotopes |
Atoms of the same element that have a different number of neutrons ( different Mass numbers) |
|
|
|
Atomic mass ( atomic weight) |
Weighted average mass of an element the naturally occurring isotopes |
|
|
|
Quantum Mechanics |
Describe the electron energies and locations by a wave equation (orbital,) |
|
|
|
Orbital (triton) |
The wave function in quantum mechanics; a plot of these describes where electron most likely to be. Each can hold 2 electrons |
|
|
|
S & P Orbital |
Most important orbitals in organic and biological chemistry |
|
|
|
How many kinds of orbitals for electrons in hydrongen atom |
There are four orbitals s, p, d, f |
|
|
|
Shell (n) |
Groupings of orbitals that vary in size and energy. Correspond to the periods on the table |
|
|
|
1st Shell (1s) |
This shell has only one s orbital Capacity 2 electrons |
|
|
|
2nd Shell (2s) |
This shell contains one s orbital (2s)x1 and p orbitals (2p)x3 Capacity is 8 electrons |
|
|
|
3rd Shell |
This shell has one s orbital (3s) x1, p orbitals (3p) x3, and d orbitals (3d) ×5 Capacity 18 electrons |
|
|
|
P orbitals |
Each shell has three of these orbitals of equal energy and separated by an area of zero electron density, a node |
|
|
|
Node |
An area of zero electron density in a p orbital |
|
|
|
Ground State Electron Configuration |
Rule of atomic structure lowest energy arrangement of atom lists orbitals occupied by electrons |
|
|
|
Aufbau Principle |
The first rule of electron config: the "build-up"- lowest energy orbitals fill first |
|
|
|
Pauli Exclusion Principle |
Rule 2 of electron config: only two electrons of opposite spin can occupy an orbital to have unique wave equations |
|
|
|
Hund's Rule |
Rule 3 of electron config: if two or more empty orbitals of equal energy are available electrons occupy each with spins parallel until all orbitals have one electron |
|
|
|
Kekulé and Couper |
Each observed that carbon always has 4 bonds |
|
|
|
Tetrahedral Carbon Atom |
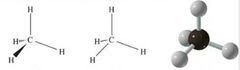
|
|
|
|
Chemical Bonding Theory |
Atoms form bonds because compound are more stable than separate atoms |
|
|
|
Covalent bond |
The bond organic compounds get from sharing electrons |
|
|
|
Ionic bonds |
This type of bond results from electron transfers (btwn metal &nm) Difference in EN > 2 |
|
|
|
Kekulé Structures |
Line bond structures: the line rep a 2 electron covalent bond |
|
|
|
Lewis Structure |
Electron dot structures where each dot rep one valence electron |
|
|
|
Nitrogen |
This element has 5 valence electrons (2s^2) ( 2p^3) but forms only 3 bonds NH3 |
|
|
|
Oxygen |
Has 6 valence electrons 2s2 2p4 but forms only two bonds H2O |
|
|
|
Molecular Geom- Linear |
Ax2 @180º |
|
|
|
Molecular Geom- Bent |
AX2E @ 120º |
|
|
|
Molecular Geom- Triangular Planar |
AX3 @120º |
|
|
|
Molecular Geom- Tetrahedral |
AX4 @109.5º |
|
|
|
Molecular Geom- Trigonal Pyramidal |
AX3E @ 107º |
|
|
|
Molecular Geom- Angular Bent |
AX2E2 @ 104.5º |
|
|
|
Lone pairs |
Another term for valence electrons not used in bonding (nonbonding electrons) |
|
|
|
Valence Bonding Theory |
This theory states that atoms in a covalent bond share electron density through the overlapping of their valence Atomic orbitals |
|
|
|
2SP2 Hybridization |
AX2 |
|
|
|
3SP2 Hybridization |
AX3 AX2E |
|
|
|
4SP3 Hybridization |
AX4 AX3E AX2E2 |
|
|
|
Tetrahedral |
109.5º. AX4 |
|
|
|
Bent |
120º. AX2E |
|
|
|
Linear |
180º AX2 |
|
|
|
Trigonal pyramid |
107º AX3E |
|
|
|
Angular Bent |
104.5º. AX2E2 |
|
|
|
Sigma Bond |
Single Bond created by overlapping of s or p orbitals during hybridization |
|
|
|
Major Resonance Structure |
The major resonance structure is one with the maximum number of atoms with zero formal charge and also one in which negative formal charges are on more electronegative atoms. |
|
|
|
ELECTRON DOMAIN GEOMETRY |
The electron pairs around a central atom (A) in a Lewis structure arrange themselves so as to minimize the electrostatic repulsions among the them. The resulting arrangement is called ... |
|
|
|
MOLECULAR GEOMETRY |
If we ignore the lone pairs, the arrangement of bond pairs result in geometry is called ____ and that is the shape of the molecule. |
|
|
|
Bond Angle |
The angle between bond pairs in a molecular geometric shape |
|
|
|
Molecular Orbital (MO) Theory |
Bonds result from combination of atomic orbitals to give molecular orbitals which belong to the entire molecule |
|
|
|
Sigma Bond |
A single bond formed by head-on interaction in a circular cross-section due to s or p overlap *in Diatomic molecules* |
|
|
|
Pi Bond |
A bond that forms bc of parallel overlap of singly occupied p orbital *polyatomic molecules* need this bond for hybridization |
|
|
|
Hybridization |
This occurs by the sharing of orbitals to form bonds in polyatomic molecules |
|
|
|
Degenerate Orbital |
Another term for hybrid orbital. Meaning energy level will be closer to the p |
|
|
|
Single bonds |
These bonds are long (1sigma) |
|
|
|
Double bonds |
This bond consists of 1 sigma + 1 pi bond |
|
|
|
Triple bond |
Short bond that contains 1sigma +2 pi bonds |
|
|
|
Polar covalent bonds |
Form when Bonding electrons are attracted more strongly by one atom than the other *think north and south pole* |
|
|
|
Electronegativity (EN) |
Intrinsic ability of an atom to attract the shared electrons in a covalent bond Difference in this produce bond polarity |
|
|
|
Nonpolar Covalent Bond |
Bond that forms btwn atoms with the same or very similar EN EN < 0.4 |
|
|
|
Inductive Effect |
Shifting of electrons in a bond response to EN of nearby atoms |
|
|
|
Polar covalent bonds |
Bond btwn atom with a a low EN to one that is high EN >0.5 |
|
|
|
Dipole |
When an arrow shows which atom is more EN |
|
|
|
Dipole moment |
Net molecular polarity due to the difference in the summed charges *n/a in symmetrical molecules bc they have one in opposite directions which cancels out" ie) benzene, ethane, methane, co2 |
|
|
|
Dipolar |
A term used to describe a neutral molecule whose central atoms have bith a positive and negative charge |
|
|
|
Arrhenius Acid |
Examines behaviors in water H is released in water |
|
|
|
Arrhenius base |
In water these bases increase in OH |
|
|
|
Brönstead Acid |
Acids are proton (H+) donors These molecules have H attached to a more EN atom |
|
|
|
Brönstead Base |
Bases are H+ acceptors EN atoms w/ lone pairs |
|
|
|
Lewis Acid |
Acids are on e (electrophile) |
|
|
|
Lewis Base |
Bases are e rich (nucleophile)
Base Bonus |
|
|
|
Carboxl group |

|
|
|
|
Hydroxl group |
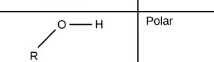
|
|
|
|
Carbonyl group |
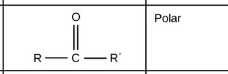
|
|
|
|
Aromatic hydrocarbon (Arene) [Benzene Ring ] |
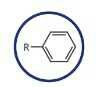
|
|
|
|
Benze nomenclature |
-yl benzene |
|
|
|
Thiol group |
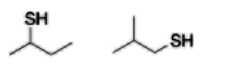
|
|
|
|
Amine group (-amine) |
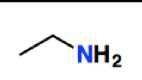
|
|
|
|
Imine group ( -imine) |
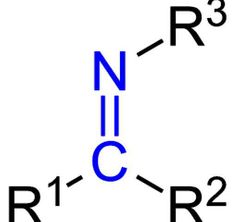
|
|
|
|
Nitrile group (-nitril) |
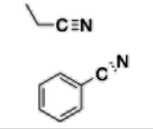
|
|
|
|
Akyl halide group (-oyl halide) |
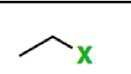
|
|
|
|
Amide group (-amide) |
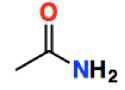
|
|
|
|
Ester (-ethyl, -oate) |
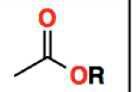
|
|
|
|
Carboxylic acid (- oic acid) |
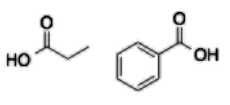
|
|
|
|
Acid anhydride (-oic anhydride) |
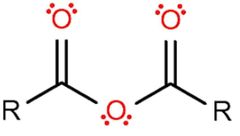
|
|
|
|
Ketone (-one) |
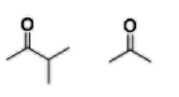
|
|
|
|
Aldehyde (-al) |
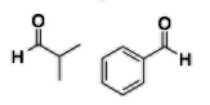
|
|
|
|
Haloalkane (halo-) |

|
|
|
|
Ether (-oxy, -ane) |
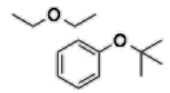
|
|
|
|
Alcohol (-ol) |
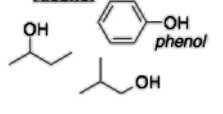
|
|
|
|
Alkene group (-ene) *hydrocarbon* |
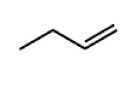
|
Hc dbl bond |
|
|
Alkyne group (-yne) *hydrocarbon* |
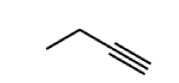
|
hc triple bond |
|
|
Alkanes *hydrocarbon* |

Not a functional group |
hc single bond |
|
|
Azo compound (azo-) |
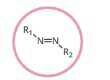
|
|
|
|
Isocyanate (-yl isocyanate) |

|
|
|
|
Amino group |

|
|
|
|
Phosphate group |

|
|
|
|
Sulfhydrl |
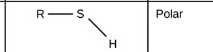
|
|
|
|
Methyl group |
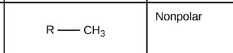
|
|
|
|
Steriochemistry |
The branch of chemistry concerned with the 3D arrangement of atoms and molecules and the effects on its chemical reactions |
|
|
|
Constitutional isomer |
Molecules with the same molecular formula but with atoms connected to each other in different ways |
|
|
|
Stereoisomer |
Molecules with the same atom connectivity but with different orientations of those atoms in space |
|
|
|
Conformation |
Different arrangement of atoms resulting from Bond rotation |
|
|
|
Steric strain |
Repulsive interaction occurring between atoms that are forced closer together than their atomic radii allow |
|
|
|
Alkyl Alphabetical Order |
butyl sec-butyl t-butyl ethyl isobutyl Isopropyl methyl propyl |
|
|
|
Staggered (Anti) conformation |
Mot stable conformation All 6 C-H are far away (180º) |
|
|
|
Eclipsed (Gauche) conformation |
Least stable conformation All 6 C-H are close =60º |
|

