![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
98 Cards in this Set
- Front
- Back
|
Identify the biological role of membranes |
Organization, compartmentilization, selective filter, communication via protein receptors, individuality, energy transduction |
|
|
How do prokaryotes differ from Eukaryotes |
Eukaryotes--Have nucleus and other organelles Prokaryotes--no nucleus (bacteria) |
|
|
What is the function of compartmentalization |
Collect/concentrate enzymes, facilitates communication of cells with surrounding environment |
|
|
Membrane Asymmetry |
When the outer membrane has a different composition than the inner membrane, assists with membrane functionality |
|
|
Non-Polar, Alphatic Amino Acids |
Glycine, Alanine, Proline, Valine, Leucine, Isolucine |
|
|
Aromatic, Non-polar amino acids |
Phenylalnine, Tyrosine, Tryptophan |
|
|
Polar Uncharged amino acids |
aspargine, glutamine, serine, threonine |
|
|
Sulfur containing amino acids |
Methionine, lysteine |
|
|
Charged Negative/Acidic Amino Acids |
Aspartate, Glutamate |
|
|
Charged Positive Basic Amino Acids |
Arginine, Lysine, Histidine |
|
|
Integral proteins |
bury deep into non-polar regions/used for transport and signal transduction |
|
|
Peripheral proteins |
bind to the tops of membranes through non-covalent, ionic, or hydrogen bonds--receptors and enzymes |
|
|
components of the fluid mosaic model |
phospholipids, cholesterol, lipids, carbohydrates |
|
|
what components of a membrane affect fluidity |
cholesterol, lipids, and proteins |
|
|
Major polar membrane lipids |
Glycerophospholipids and Sphingolipids |
|
|
Glycerophospholipids |
phosphatidic acid is the foundation molecule, 2 fatty acids in eser bond to glycerol and a phosphate |
|
|
Phingolipids |
sphingosine backbone, 1 fatty acid and polar glucose or galactose |
|
|
phosphatidylcholine differs from sphingomyelin |
phosphatidylcholine has a choline head group, clycerophosphoric acid and 2 fatty acids Sphingoymelin has a phosphocoholine, a ceramide, and a fatty acid |
|
|
How does the orientation of a phospholibid differ if it in water versus it being in oil |
direction of the polar heads--on the outside in water, inside with oil |
|
|
What compounds belong to the class of glycerophospholipids |
Phosphatidic acid, Phosphatidylethanolamine, Phosphatidylcholine, Phosphatidylserine, Phosphatidylinositol |
|
|
What compounds belong to the class of sphingolipids |
Cermide, sphingomyelin, ganglioside, glucosylcerebroside |
|
|
Cholesterol is derived from what compound |
Isoprene |
|
|
Cholesterold is the precursor molecule for what classes of compounds |
Steroid hormones and bile acids |
|
|
What are the characteristics of proteins in plasma membranes? |
channels/transporters for movement of compounds across membranes, receptors for hormones and neurotransmitters, provide structure
|
|
|
What is the glycocalyx and how does it function |
Exposed carboyhydrate chains that are hydrophilic--protect cell from digestion, restrict hydrophobic compounds from entering |
|
|
What is the function of aquaporin |
allows faster movement of H20 |
|
|
How does glucose permease effectuate glucose absorption in the red blood cell |
Hydrophobic region coils into alpha helix which allows for glucose transport |
|
|
Molecules that can diffuse across membranes |
Gases like CO2, 02, N2, CH4, small polar molecules (water, ethanol), and hydrophobic molecules (benzene) |
|
|
Cannot diffuse across membranes |
Large polar molecules (glucose), charged molecules (H+, Cl-, Ca2+, Na2+) |
|
|
Carrier mediated transport vs. diffusion |
diffusion molecules move along electrochemical gradient, carrier mediated move against the gradient, has specificity, competition and saturation |
|
|
Voltage gated channels |
transmembrane proteins form a pore for ions that responds to stimuli to open and close, Na+ gates are closed, depolarization opens them |
|
|
Ligand gated channels |
Opens or closes in response to a chemical messenger like a ligand |
|
|
The sodium Potassium Pump |
3 sodium ions enter for each 2 potassium ions that leave, atp hydrolysis is used to allow sodium to be released, potassiums bind and trigger hydrolysis of the bound phosphate group and return to original composition |
|
|
Sodium CoTransport or Symport |
glucose enters with sodium into cell, sodium then leaves, and postassium pumps in to maintain intra and extracellular concentrations |
|
|
what are lysosomes, what do they do |
digestive organelles, prevent loss of hydrolyctic enzymes into cytosol, eliminate unwanted materials, destroy infection bacteria and yeast, recover from injury, tissue reomodeling, cell turnover |
|
|
identify lysosomal hydrolases |
Proteases, Glycosidases, glucuronidases, sulfatases, deoxyribonucelases, ribonuclease, phosphatase, lipase, phopholipase |
|
|
Proteases |
break down proteins |
|
|
Glycosidases, Glucuronidases, and Sulfatases |
Break down carboyhdrates |
|
|
Deoxyribonuclease |
breaks down DNA
|
|
|
Ribonuclease |
Breaks down RNA |
|
|
Phosphatase |
Inorganic phosphate |
|
|
lipase |
breaks down triacylglycerols |
|
|
Endocytosis |
Particles to be transported are enclosed in small portions of cell membrane to form vesicles--uses ATP |
|
|
Exocytosis |
release secreted proteins into the exterior (expulsion of waste) |
|
|
Functions of the Golgi Apparatus |
posttranslational modification of proteins |
|
|
Endoplasmic Reticulum |
Smooth ER- synthesis of lipids and steroids Rough ER- synthesis of proteins |
|
|
What is the function of kinesin motor proteins |
Moves vesicles along tubules |
|
|
Cytoskeleton and how does it function |
The structure of the cell, organized by microtublules, thin filaments, and intermediate filaments--uses actin and tubulin for cellular movement |
|
|
General features of a chemical messenger |
Secreted by specific cells in response to stimulus, transported through blood or extracellular fluid, messenger binds to target cell receptor, messenger binding elicits response |
|
|
Types of chemical messengers |
Nervous, Endocrine, Cytokines, Eicosanoids |
|
|
Nervous chemical messengers |
biogenic amines/neuropeptides |
|
|
5 types of biogenic amine neurotransmitters |
Catecholamines: Dopamine, Norepinephrine, and epinephrine, as well as histamine and serotonin |
|
|
Endocrine chemical messengers |
hormones from endocrine glands, estrogen |
|
|
Cytokine chemical messengers |
cell to cell communication--interferons, interleukins |
|
|
Eicosanoids |
made by oxidation of 20 carbon-fatty acids, derived from omega 3 or omega 6 fatty acids--prostoglandins
|
|
|
Intracellular Receptors |
hydrophobic molecules, gene specific transcription factors like steroid hormones, thyroid hormone, etc |
|
|
Plasma membrane receptors |
has an external domain, membrane spanning domains that are alpha helices, and intracellular domain that initiates signal transduction |
|
|
Categories of plasma membrane receptors |
Ion channel linked receptors, Kinase or activated Kinase receptors, Heptahelical receptor |
|
|
Ion Channel Linked Receptors |
cell membrane bound receptors--ligand gated channels, conformational change occurs upon ligand binding |
|
|
Kinase or activated Kinase receptors |
intracellular domain is activated when messenger binds to extracellular domain and amino acid on receptor is phosphorylated--response is propogated through signal transduction proteins (tyrosine kinase receptor) |
|
|
Heptahelical Receptor |
G proteins (guanosine nuceleotide-binding proteins), have 7 membrane spanning helices, work through second messengers like cyclic AMP |
|
|
9 functions of water |
transport nutrients and waste, provide medium for chemical reactions, act as a reducing agent, act as a solvent, lubricates and cushions joints, absorbs shock and maintains temperature, buffering substances, maintains blood volume, aids in digestion, absorption, metabolism of nutrients |
|
|
hydrogen bonding and why is it important in water |
chemical bond formed between a hydrogen atom and an oxygen, nitrogen, or flourine. Holds water in its liquid and solid states (easily broken) |
|
|
How does Hydrogen Bonding effect the state of water |
The strength of the hydrogen bonds leads to unusually high melting temperature--the bonds form crystalline patterns in ice where all 4 hydrogen bonds participate vs. 2-3 hydrogen bonds in liquid form |
|
|
How does hydrogen bonding compare to the strength of a covalent bond |
Hydrogen bond is much weaker, need about 4-4.77 calories to break, need 110 calories to break covalent bond |
|
|
Why is the fact that water becomes less dense when frozen beneficial for marine life |
Only the top layer of a river or lake freezes which allows the water below to remain unfrozen and support life, it also traps oxygen under the layer of ice |
|
|
What is the specific heat of water |
1.00 cal/g C |
|
|
Examples of organic molecules that form hydrogen bonds with water |
alcohol, amides, amines, esters, carbonyl |
|
|
Examples of hydrogen bonding between molecules without water being present |
two peptides, two complementary base pairs of DNA |
|
|
examples of organic molecules that water can dissolve |
polar compounds--alcohol, amides, amines, esters and ionic compounds (salts) |
|
|
how are ionic compounds dissolved by water |
because they are polar and their individual ions can be saturated with water |
|
|
What is waters interaction with non-polar compounds |
not water soluble, hydrophobic |
|
|
why is water a good solvent |
low viscosity, smaller relative to organic compounds, can dissolve polar compounds and compounds that ionize |
|
|
How does water affect thermoregulation in the body |
high heat of fusion so a lot of energy required to raise temperature , high heat of vaporization |
|
|
What are electrolytes? |
ionic salts |
|
|
examples of electrolytes |
Na, K, Ca, Mg, HC03, SO, PO |
|
|
what is osmolality |
the measure of the moles of solute per kilogram of solvent
|
|
|
Why is osmolality important? |
measures the body's electrolyte-water balance |
|
|
What is water's relation to pH |
water dissociates or ionizes forming a hydroxyl molecule (OH) and hydrogen atom (H+) |
|
|
what is pH? |
pH is the negative log of the proton concentration |
|
|
what is an acid? |
any substance with a pH below 7 (releases a proton) |
|
|
what is a base? |
any substance with a pH higher than 7 (accepts a proton) |
|
|
Examples of acidic compounds |
battery acid, lemon juice, vinegar, beer, coffee |
|
|
Examples of basic compounds |
seawater, detergent, soapy water, hair remover, drain cleaner, blood plasma |
|
|
examples of neutral compounds |
distilled water/waters with salt dissolved |
|
|
why is the maintenance of normal pH essential for cells? |
critical enzymes are pH sensitive, can cause cell death |
|
|
Examples of common organic acids |
formic acid, carbonic acid, acetic acid, citric acid, lactic acid |
|
|
what is pK, what does it measure? |
pK is the negative log of the acid dissociation constant and the measure of acid strength low pK strong acid |
|
|
what is the henderson-hasselbalch equation? |
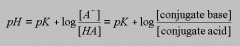
|
|
|
what does it do? |
used to calculate pH, pK, and individual acid and conjugate base concentrations and laboratory buffers mathematical calculation of all points on the titration curve |
|
|
What is a buffer? |
A buffer solution is an acid and its conjugate base or vice versa |
|
|
What is titration? |
titration is adding incremental amounts of base to an acid sample and monitoring the pH with a pH meter. used to determine the pK of the acid |
|
|
Examples of biological buffers |
carbonate-bicarbonate buffer system, phosphate buffer system, protein buffer system |
|
|
What is the major source of CO2 production? |
TCA cycle |
|
|
What is the chloride shift-whys it important? |
Cl- is brought into the RBC to keep neutral charge as HCO3- leaves RBC (blood pH is extremely important and needs to remain neutral) |
|
|
How is hydrochloric acid produced in parietal cells? |
CO2 enters cell from blood, combines with water with carbonic anhydrase, transformed into carbonic acid, which dissociates into H+ and bicarbonate (HCO3-). Bicarbonate is released pulling in Cl- from the blood. Hydrogen-potassium ATPase pulls the H+ into gastric duct gland and K+ across paritial cell. Cl-combines with H+ to form hydrochloric acid |
|
|
Reasons for dehydration |
inadequate water intake, excessive water loss through kidney lungs and skin burns, result of impaired kidney function, impaired response to hormones |
|
|
How is intracellular pH maintained |
Phosphate anions and ATP act as buffers, liver produces ketone bodies, lactic acid from muscles, |

