![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
244 Cards in this Set
- Front
- Back
|
Main characteristics of prion
diseases • • • • • • |
Main characteristics of prion
diseases • Neurological disease • Long incubation time • No immune response • No inflammation • Prion - protein infectious agent • Protein only - No nucleic acid detected • Resistant to heat, UV irradiation, chemicals, proteases |
|
|
• Icelandic farmers - known for two
centuries • 1934 - sheep to sheep experimental transmission by injection of infected brain • 1961 - sheep to mouse experimental transmission by injection of infected brain Symptoms • Pruritis • Biting, rubbing • Tremors • Weaving gait • Hind quarter paralysis • Loss of weight, death in 4-6 weeks |
Scrapie
|
|
|
prion Brain lesions
• • |
Brain lesions
• Astrocytosis • Vacuolization of neurons |
|
|
scrapie Transmission and pathogenesis
• • • • – – |
Transmission and pathogenesis
• Horizontal • Vertical - ewe to lamb • IP. 1-5 years • After infection, sequential spread into: – Lymphoid tissues, tonsils, intestines, and spleen – Brain |
|
|
• "Mad cow disease"
• Great Britain - 1986 • Other countries - Ireland, France, Germany, Switzerland, Japan • By 1997, over 172,000 confirmed cases in the UK |
Bovine Spongiform
Encephalopathy (BSE) |
|
|
• 1998 - 34 confirmed cases
• Younger patients - 19-45 years • No risk factors • Longer course (mean 14 months vs. standard 6 months) • Until 1989 ban - pooled homogenates of bovine brains were used as binders in sausages, hamburgers |
Variant Creutzfeldt -Jakob Disease
(vCJD) |
|
|
prion
• Deer and elk • CO and WY endemic • Death within few months of clinical signs |
Chronic Wasting Disease
(CWD) |
|
|
Problems with antiviral therapy
• • • |
Problems with antiviral therapy
• Intracellular parasite • Stage of viral disease • Drug resistance |
|
|
Drug targets in viral life cycle
• • • • • |
Drug targets in viral life cycle
• Attachment and penetration • Virus uncoating • Unique viral enzymes involved • Viral protein synthesis and processing • Assembly and release |
|
|
Interferons
• Induced by . 1. • 2. • 3. • _____________ specific • Not _______ specific |
Interferons
• Induced by viral nucleic acids • Viral infected cells • Antigens • Species specific • Not viral specific |
|
|
interferons Mechanism of action
• • • |
Mechanism of action
• Binding to cell surface receptor • Induction of antiviral effector molecules • Inhibition of viral mRNA translation |
|
|
Interferon biological effects
• • • • |
Interferon biological effects
• Initial viral defense • Important in recovery • Positive effects on T and B cells, Macrophages • Anticancer effects |
|
|
Antiviral Drugs two types
• • |
Antiviral Drugs
• Nucleoside analogs • Non-nucleoside analogs |
|
|
• Large 100-300 nm in
diameter, enveloped, icosahedrons • Linear, ds DNA genome, 150 kb • Latency • Large family of pathogens: mammals, birds, fish, reptiles and amphibians |
Herpesviridae
|
|
|
Current Classification of
Herpesviruses |
Current Classification of
Herpesviruses • Species, serial number • e.g., bovine herpesvirus 1 aka infectious bovine rhinotracheitis • e.g., ranid herpesvirus 1 aka Lucke frog herpesvirus |
|
|
• ________________ (subfamily)
– Variable host range – Rapid replication in cell culture – Rapid cytopathic effect in cell culture – Latency in sensory ganglia • Example: Bovine herpesvirus 1(BHV-1) |
• Alphaherpesvirinae (subfamily)
|
|
|
________________
(subfamily) – Restricted host range – Slow replication – Cytomegalia in cells – Latency in secretory glands, lymphoreticular cells, kidneys • Examples: Porcine herpesvirus-2, aka porcine cytomegalovirus |
• Betaherpesvirinae
|
|
|
_______________
(subfamily) • Restricted host range • Replication in lymphoblastoid (T or B) cells • Some lytic infections in epithelioid and fibroblastic cells • Latency in lymphoid tissue • Example: alcelaphine herpesvirus 1 |
• Gammaherpesvirinae
|
|
|
• Enveloped, lipid,
pleiomorphic, glycoproteins • Tegument, amorphous • Icosahedral nucleocapsid, T=16 • Fragile in the environment |
herpesvirinae
|
|
|
Genome
• Large ds DNA, 150 kbp • Linear in the virion • Circularizes in the infected cell • Encodes 70 to 200 proteins • Replication occurs in the nucleus |
herpesvirinae
|
|
|
herpesviridae
Latent Infection • Entry of herpesvirus into ________________ and transport of nucleocapsids to ______________ and _________ • Viral DNA occurs as _________ (5-10) in 1% of ___________ in a __________ • ___________________` (LAT) is expressed during latency, but function is not well understood |
Latent Infection
• Entry of herpesvirus into sensory nerve endings and transport of nucleocapsids to neuronal cell body and nucleus • Viral DNA occurs as episomes (5-10) in 1% of neurons in a ganglion • Latency-associated transcript (LAT) is expressed during latency, but function is not well understood |
|
|
herpesviridae
Reactivation • Stimulated by _______, _______, __________ associated with menstrual cycle, _________ • Limited replication occurs in __________ • Virions are transported down axons to _______________ cells • _________ replication occurs in tissues • Limited by _______ response |
Reactivation
• Stimulated by stress, UV light, hormonal changes associated with menstrual cycle, fever • Limited replication occurs in neurons • Virions are transported down axons to epithelial cells • Lytic replication occurs in tissues • Limited by immune response |
|

Alphaherpesviral Diseases
|
Alphaherpesviral Diseases
|
|
|
• Host range: Bovidae, wild ruminants
• Transmission: Aerosolized nasal secretions, genital secretions, semen • Latent in trigeminal and sacral ganglia • Lifelong periodic shedding • Vaccines: inactivated and MLV IBR vaccines prevent severe disease but not infection |
Bovine Herpesvirus-1
|
|
|
bovine herpesvirus-1 Diseases
1. 2. 3. 4. |
1. Infectious bovine rhinotracheitis (IBR),
“rednose”: respiratory disease/pneumonia in feedlot and dairy calves 2. Conjunctivitis, “pinkeye”: keratitis, corneal ulcers 3. Pustular vulvovaginitis, balanoposthitis: venereal transmission, AI with contaminated semen 4. Abortions: 4-7 months of gestation following respiratory infection of dam |
|
|
1. Rhinopneumonitis: fever, serous nasal
discharge 2. Subclinical infections are common 3. Outbreaks occur in young horses 4. Introduction of new stock, transport to shows, racetrack or breeding facilities 5. Exposure to latently infected horses 6. Latent in trigeminal ganglion |
Equine Herpesvirus-1&4
|
|
|
1. Abortions: dead foals, 4 to 11 months
gestation 2. Neonatal foal mortality: systemic disease, interstitial pneumonia 3. Cell-associated viremia following respiratory infection of the mare 4. Occurs 2 to 8 weeks post-infection 5. May occur regardless of vaccination status of the mare |
Equine Herpesvirus-1
|
|
|
1. Neurologic disease: ataxia, toe-dragging,
paralysis of bladder, tail, penis, paralysis 2. May/may not be preceded by respiratory signs 3. Can occur in outbreaks 4. Associated with specific strains 5. Pathogenesis: immune complex-related vasculitis accompanied by hemorrhage in the spinal cord and brain 6. Vaccination may increase risk of neurologic disease |
Equine Herpesvirus-1
|
|
|
1. Coital exanthema
2. Transmission: venereal, iatrogenic 3. Vesicles, pustules, ulcers of the vaginal mucosa, penis and prepuce 4. Depigmentation of vulva and perianal areas 5. Latent in sacral ganglion 6. Temperature restriction |
Equine Herpesvirus-3
|
|
|
1. Neonatal Mortality: systemic spread,
fever, abdominal pain, hemorrhagic enteritis, shock, rapid progression to death 2. Abortions: Focal necrosis of liver 3. Balanoposthitis: ulceration of preputial mucosa, introduced via infected bucks |
Caprine Herpesvirus-1
|
|
|
1. PHV-1, Aujeszky’s disease
2. Abortions: embryonic death and resorption, mummification, stillbirth, weak pigs 3. Respiratory/generalized disease: fever, sneezing, coughing, depression, lethargy, anorexia, vomiting, constipation 4. Neurologic disease: incoordination, circling, convulsions, salivation |
Pseudorabies
|
|
|
Pseudorabies - Pathogenesis
1. 2. 3. a. b. 4. 5. 6. 7. |
Pseudorabies - Pathogenesis
1. Ingestion/inhalation 2. Replication in oropharynx and tonsils 3. Viremia and spread to: a. Reproductive tract and fetuses b. Respiratory tract 4. Shedding via nasal and oral secretions by recovered swine 5. Spread via cranial nerves to ganglia via the axoplasm to the CNS 6. Non-suppurative meningoencephalitis and ganglioneuritis 7. Latent infection of ganglia |
|
|
herpesvirus
1. Decreasing mortality rates with increasing age 2. Decreased growth rates in recovered swine 3. Eradication program in the U.S. instituted in 1989 4. Currently, accelerated eradication program in progress (1999) |
Pseudorabies
|
|
|
1. “Mad itch”, disease in animals other
than swine, except tail-less apes 2. Virus in saliva of infected pigs, transmission via ingestion, inhalation, bites or wounds 3. Encephalitis, intense pruritus, selfmutilation, paralysis of jaws, salivation, frenzy 4. 100% fatality, duration of hours to 6 days |
Pseudorabies
|
|
|
1. Host Range: Felidae
2. Shedding by 30% of normal cats, aerosol/contact transmission 3. Latent in trigeminal ganglion 4. Reactivated by parturition, lactation, stress |
Feline Herpesvirus-1
|
|
|
feline virus
1. Respiratory disease: sneezing, serous to mucopurulent ocular and nasal discharge, glossitis 2. Ocular disease: keratitis, corneal ulcers 3. Neonatal mortality: necrotizing pneumonia 4. Vaccine reduces severity of disease |
Feline Herpesvirus-1
|
|
|
1. Host Range: Canidae
2. Seroprevalence: 20-30% 3. Transmission: oral-nasal route 4. Latent in sacral ganglia 5. Recrudescence at parturition and infection of pups via vaginal secretions 6. Disease in puppies may or may not recur in subsequent litters |
Canine Herpesvirus-1
|
|
|
1. Neonatal mortality: systemic,
hemorrhagic disease 2. Puppies under 4 weeks of age 3. Abdominal pain, crying, dyspnea, rapid progression to death 4. Viral replication at 33C, role of hypothermia in generalized disease in neonatal puppies |
Canine Herpesvirus-1
|
|
|
Alphaherpesvirus infections of
avian species 1. 2. 3. 4. 5. |
Alphaherpesvirus infections of
avian species 1. Gallid herpesvirus 1 aka infectious laryngotracheitis 2. Gallid herpesvirus 2 aka Marek’s disease 3. Anatid herpesvirus aka duck plague aka duck viral enteritis 4. Pacheco’s disease, psittacine herpesvirus-1 5. Great horned owl herpesvirus |
|
|
• Host range: chickens
• Etiology: Gallid Herpesvirus-2 • Ubiquitous • Serotypes: – 1, oncogenic viruses in chickens – 2, avirulent chicken viruses – 3, avirulent turkey viruses |
Marek’s Disease Virus
|
|
|
• Transmission: carrier birds
– Replicates in feather follicles – Shed in feather dander – Durable in the environment • Affects chickens 12-24 weeks of age, increased resistance with age • Genetic resistance or susceptibility – MHC alleles – Sensitivity of T cells to transformation |
Marek’s Disease Virus
|
|
|
• Lymphoproliferative disease, T
cell lymphoma – Sciatic nerve: paralysis – Liver, kidney, spleen: wasting, hemorrhage due to rupture – Iris: blindness • Vaccines: live THV, attenuated serotype I or serotype II viruses |
Marek’s Disease Virus
|
|
|
herpesvirus
• Outbreaks of acute death in wild and feral ducks • High mortality rate • Enzootic in N.A. • A reportable disease |
Duck Plague (Viral Enteritis)
|
|
|
Duck Plague
• Host range: Anseriformes • Transmission: • Prevention/control: |
Duck Plague
• Host range: Anseriformes • Transmission: – Direct contact with infected birds and their secretions – Contact with contaminated water • Prevention/control: – Separate domestic and wild waterfowl – Carcass disposal, disinfection – Vaccine is available under special circumstances |
|
|
herpesvirus
Clinical Presentation • Sudden death • Bloody or watery diarrhea • Dehydration • Oculonasal discharge • Photophobia • Drooping head and wings • Prolapsed penis |
duck plague
|
|
|
Psittacine Herpesviruses
• Host range: ______________ • Etiologic agents: – ______________ (PHV-1) – PD-like disease viruses • ______ • ______ • History of exposure to ________ |
Psittacine Herpesviruses
• Host range: psittacines • Etiologic agents: – Pacheco’s disease (PHV-1) – PD-like disease viruses • PHV-2 • PHV-3 • History of exposure to psittacines |
|
|
PHV Clinical Presentation
|
1. Sudden death
2. GI: Anorexia, vomiting, bloody diarrhea, increased thirst and urination 3. Respiratory: conjunctivitis 4. CNS: depression, tremors, ataxia, opisthotonos, seizures |
|
|
Other Avian Herpesviruses
• Raptors: – – • Cranes: – – • Passerines: – |
Other Avian Herpesviruses
• Raptors: owls, falcons – Weakness, regurgitation, diarrhea – Leukopenia, hepatosplenitis, hepatic necrosis • Cranes: Inclusion body hepatitis – Acute death – Splenohepatomegaly, hemorrhagic enteritis • Passerines: – Conjunctivitis, respiratory disease |
|
|
Betaherpesvirus infections
• _____________________, porcine cytomegalovirus, inclusion body rhinitis • Sneezing, coughing, serous to mucopurulent nasal discharge, openmouth breathing • Spread via aerosol/resp. secretions • Occasional stillbirths if gilt/sow is infected during pregnancy • Component of atrophic rhinitis |
Poricne herpesvirus 2
|
|
|
Gammaherpesvirus infections
• • • • |
Gammaherpesvirus infections
• Alcelaphine herpesvirus 1, Malignant catarrhal fever • Ovine herpesvirus 2, Malignant catarrhal fever • Bovine herpesvirus-4 • Equine herpesvirus-2, -5 |
|
|
herpesvirus
• Reservoir host: wildebeest, topi, hartebeest • Affected species: cattle, deer, antelope • Etiologic agent can be grown in cell culture • Transmission: shed by wildebeest particularly at calving • Occurs in Africa and in zoos |
Alcelaphine herpesvirus 1
|
|
|
herpesvirus
• Reservoir host: domestic sheep • Affected species: cattle, deer, bison • Etiologic agent has not been isolated • Transmission: virus is shed by sheep, increased transmission at lambing • Occurs in North America and Europe |
Ovine herpesvirus 2
|
|
|
Herpesvirus
• Sporadic, fatal disease of cattle, deer, bison and other ruminants • Characterized by profuse mucopurulent nasal/ocular discharge • Keratoconjunctivitis with corneal opacity • Lymphadenopathy • Vasculitis, immunopathologic lesion • Gastroenteritis, ulceration, hemorrhage |
Malignant Catarrhal Fever
Ovine herpesvirus 2 |
|
|
• Non-enveloped,
icosahedral (T=25) symmetry, 70-120 nm in diameter • Double-stranded, linear DNA genome 36-44 kb • Pathogens of mammals and birds |
Adenoviridae
|
|
|
• Discovered by Rowe (1953)
• Explanted tonsillar tissue in culture • Highly host-specific and hostadapted • Persistent infections • Relatively stable in the environment |
Adenoviridae
|
|
|
• ______________ (genus)
– Canine adenovirus-1, CAV-2 – Equine adenovirus – Deer adenovirus • ____________ (genus) – Hemorrhagic enteritis in turkeys – Marble spleen disease in pheasants – Quail bronchitis • Atadenovirus (pending) |
• Mastadenovirus (genus)
– Canine adenovirus-1, CAV-2 – Equine adenovirus – Deer adenovirus • Avidadenovirus (genus) – Hemorrhagic enteritis in turkeys – Marble spleen disease in pheasants – Quail bronchitis • Atadenovirus (pending) |
|
|
Adenoviridae Replication
|
• Entry via endocytosis
• DNA transport to nucleus • Early mRNA transcription • Early protein translation • DNA genome synthesis • Late mRNA transcription • Late protein translation • Assembly |
|
|
1. Canine adenovirus 1
2. Canine adenovirus 2 3. Deer adenovirus 4. Equine adenoviruses 5. Miscellaneous adenoviruses |
Mastadenoviruses
|
|
|
a. Etiology of infectious canine hepatitis
b. Host range: dogs, foxes, wolves, coyotes, badgers, bears, skunks c. Transmission: ingestion of virus in urine, feces and saliva d. Virus persists in kidneys and is shed in the urine of survivors for prolonged periods e. Severe, generalized disease: mortality in puppies of up to 100% |
Canine Adenovirus-1
|
|
|
1. Inapparent infections
2. Respiratory disease, component of kennel cough 3. Peracute disease, sudden death 4. Acute disease: fulminant hepatitis, enteritis +/- hemorrhage, encephalopathy 5. Immunopathologic diseases: anterior uveitis, “blue-eye”, glomerulonephritis |
Infectious canine hepatitis
(Canine Adenovirus-1) |
|
|
Infectious canine hepatitis
(Canine Adenovirus-1) Clinical Presentation (Acute) |
• Fever
• Leukopenia • Conjunctivitis, oculonasal discharge • Abdominal pain • Jaundice • Anemia, pale mucous membranes • Increased clotting time • Petechial hemorrhages • Disseminated intravascular coagulation |
|
|
Pathogenesis of Infectious canine hepatitis
(Canine Adenovirus-1) |
a. Ingestion
b. Replication in tonsils, Peyer’s patches c. Viremia d. Infects endothelial cells, DIC, hemorrhage e. Infects liver, hepatitis, jaundice, increased clotting time f. Infects kidney, nephritis |
|
|
Pathogenesis of “Blue-eye”
|
a. Immunopathologic consequence
observed in recovery phase 8 to 12 days post-infection or vaccination with MLV CAV-1 b. Virus-antibody complex formation c. Deposition in capillaries in the ciliary body d. Impedes corneal fluid flow e. Corneal edema |
|
|
Canine Adenovirus-1 Prevention and Control
|
• Vaccination, CAV-2
• Rare disease in the U.S. • Occurs in unvaccinated puppies • Prolonged shedding of CAV-1 by survivors • Lifelong immunity following natural infection |
|
|
a. Etiologic agent: mild to inapparent
respiratory disease, tonsillitis, pharyngitis, tracheitis, bronchitis b. Host Range: Canidae c. Antigenically similar to another dog virus, confers cross-protective immunity and used as vaccine virus d. Shed in urine leading to secondary “vaccination” and herd immunity |
Canine adenovirus-2
similar to CAV-1 |
|
|
a. Newly recognized (1993) hemorrhagic
disease in deer b. Host Range: Black-tailed deer, muledeer, white-tailed deer, moose, pronghorn c. Transmission: Direct contact d. Described in captive fawns and freeranging animals of different ages e. Systemic disease: vasculitis, endothelial necrosis, pulmonary edema, hemorrhagic enteritis, seizures, recumbency, death f. Localized disease: swollen muzzles, necrotizing stomatitis, oral abscesses, emaciation, death due to starvation |
Adenovirus Hemorrhagic
Disease of Deer |
|
|
a. 2 serotypes
b. Inapparent respiratory tract infections in normal horses c. Pneumonia, systemic spread to pancreas, kidneys, bladder and GI tract in immunocompromised hosts, e.g., severe combined immunodeficiency (SCID) in Arabians |
Equine adenoviruses
|
|
|
Miscellaneous adenoviruses
a. Bovine – ___________ b. Swine – _________ c. Ovine – ________ d. Caprine – _________ e. Rabbits - __________ |
Miscellaneous adenoviruses
a. Bovine – inapparent or mild respiratory disease b. Swine – “ c. Ovine – “ d. Caprine – “ e. Rabbits - diarrhea |
|
|
Aviadenoviruses
a. Ubiquitous viruses of birds b. Host range: ___________ c. Serogroups based on group __________ 1. Group I: ____________ 2. Group II: ___________ 3. Group III: _____________ d. _______ serotypes |
Aviadenoviruses
a. Ubiquitous viruses of birds b. Host range: domestic poultry, pigeons, psittacines, ratites, raptors c. Serogroups based on group antigen 1. Group I: quail bronchitis 2. Group II: turkey hemorrhagic enteritis 3. Group III: egg drop syndrome d. Multiple serotypes |
|
|
Aviadenoviruses
Clinical Presentation a. Quail Bronchitis:_________ b. Hemorrhagic enteritis:__________ c. Marble spleen disease:_____________ d. Inclusion body hepatitis:_________ e. Egg drop syndrome: ______________ |
Clinical Presentation
a. Quail Bronchitis: respiratory disease, rales, coughing, sneezing, conjunctivitis, mortality 10-100% b. Hemorrhagic enteritis: acute onset, depression, bloody diarrhea, high mortality c. Marble spleen disease: sudden death, dyspnea, bloody diarrhea d.Inclusion body hepatitis: acute onset of anemia, hepatitis, high mortality e. Egg drop syndrome: decreased pigment, malformed shells, decreased production |
|
|
Aviadenoviruses Transmission
|
• Shed in the feces of healthy birds
• Ingestion • Vertical transmission (?) IBH • Persistent infection, reactivation and intermittent shed • Durable in the environment • Strains tend to be more virulent for species other than “normal” host |
|
|
Aviadenoviruses Prevention and Control
|
• Vaccines:
– MSD for pheasants – HE for turkeys • Isolation, quarantine, all-in-all-out, disinfection, sanitation • Avoid feeding domestic poultry to falcons • Avoid contact with free-ranging pigeons and other birds |
|
|
Circovirus
• ________________DNA genome • Ambisense or __________ • Limited ______ and gene products • envelope +or-, shape:______ • _______ in the environment |
Circovirus
• Single-stranded, circular DNA genome • Ambisense or positive-sense • Limited ORFs and gene products • Non-enveloped icosahedron, 17 - 22 nm in diameter • Durable in the environment |
|
|
Circovirus Classification
|
• One genus:
– Chicken anemia virus – Psittacine beak and feather disease virus – Porcine circovirus 1 – Porcine circovirus-2 |
|
|
Circovirus
• Host range: chickens • Transmission: – Horizontal: direct contact, fomites – Vertical: hen to egg • Age: young birds, 2-3 weeks of age • Mortality: 10-50% • Immunity: maternal Abs are protective |
Chicken Anemia Virus
|
|
|
Circovirus
• Acute infection • Anorexia, lethargy, depression • Decreased hematopoiesis: anemia, pale birds • Hemorrhages: • Immunosuppression: atrophy of lymphoid organs/tissues, bone marrow, thymus, bursa, spleen |
Chicken Anemia Virus
|
|
|
Circovirus
• Host Range: Cockatoos, parrots and budgerigars, etc. • Transmission: Shed in feather follicular epithelium • Age: Fledglings and young birds |
Psittacine Beak and Feather
Disease |
|
|
• Dystrophic feathers
– Necrosis – annular constrictions – shaft hemorrhage, – feather loss • Beak dystrophy, reduced growth • Abnormal plumage or color |
Psittacine Beak and Feather
Disease (cirovirus) |
|
|
Circoviruses
• Canaries: |
Circoviruses
• Canaries: Black spot disease |
|
|
avian Circoviruses
Transmission and Control |
• Shed in feather dander and feces
• Durable in the environment, resistant to many disinfectants – Clean up organic material – 1:30 solution of bleach – Environmental testing, PCR • Maternal antibodies are protective against disease but not infection • Survivors are immune • No vaccines available |
|
|
Porcine Circoviruses
• Host species: ________ • Etiologic agents: – _________________ (pathogenicity unknown) – ________________ • Disease:_________________________ |
Porcine Circoviruses
• Host species: domestic swine • Etiologic agents: – Porcine circovirus-1 (pathogenicity unknown) – Porcine circovirus –2 • Disease: Porcine post-weaning wasting syndrome (PMWS) |
|
|
Porcine Circoviruses Clinical Presentation
|
• Specific pathogen-free herds
• Piglets 6 weeks of age • Weight loss, lymphadenopathy, dyspnea, diarrhea • 10% mortality • Disease is dependent on multiple factors and etiologic agents |
|
|
Parvoviridae
•size: • envelope+or- shape • ____________genome, 5kb • Pathogens of humans and animals |
Parvoviridae
• Small (18-26 nm in diameter) • Non-enveloped, icosahedral virus • Single-stranded DNA genome, 5kb • Pathogens of humans and animals |
|
|
parvovirus
Structure • T=1 icosahedron, 60 molecules of VP2 • Hollow cylinder at 5-fold axis of symmetry • “________” surrounding 5-fold axis • “__________” at the 3-fold axis is the viral attachment protein which determines tissue tropism and host range • “________” at 2-fold axis |
Structure
• T=1 icosahedron, 60 molecules of VP2 • Hollow cylinder at 5-fold axis of symmetry • “Canyon” surrounding 5-fold axis • “Spike” at the 3-fold axis is the viral attachment protein which determines tissue tropism and host range • “Dimple” at 2-fold axis |
|
|
parvo
Classification • Parvovirinae (subfamily) – __________ (genera) – __________ – ___________ • Densovirinae (subfamily) – ____________ – ____________ – ____________ |
Classification
• Parvovirinae (subfamily) – Parvovirus (genera) – Erythrovirus – Dependovirus • Densovirinae (subfamily) – Densovirus – Iteravirus – Brevidensovirus |
|
|
parvo Replication
|
• No viral polymerase encoded
• Requires host cell functions of late S or early G2 phase of cell cycle • Cellular DNA polymerase II extends ss vDNA using the hairpin and free 3’-OH as primer to make ds DNA intermediate • Cellular transcriptases synthesize mRNA • Alternate splicing generates multiple mRNAs |
|
|
parvo Target tissues
|
• Stem cells in bone marrow and
lymphoid organs: erythroid precursors, lymphocytes, neutrophils, monocytes & platelets • Stem cells in the crypts of Lieberkühn • Cells in the external granular layer of the cerebellum |
|
|
• Emerged in 1978
• Virgin-soil pandemics, high morbidity and mortality rates • Host Range: Canidae, felidae • Transmission: acquired by ingestion, shed in feces • Durable in the environment |
Canine parvovirus-2
|
|
|
dogs
• Hemorrhagic enteritis: 8 to 12 weeks of age: leukopenia, lymphopenia, fever, vomiting, anorexia, lethargy, depression, bloody diarrhea, dehydration, secondary septicemia, death • Neonatal infections: 4 to 8 weeks of age: sudden death, myocarditis, pulmonary edema |
Canine parvovirus-2
|
|
|
• Etiologic agent: feline panleukopenia
virus, feline distemper virus, feline enteritis, “cat fever”, feline ataxia • Host range: Felidae, some canids • Transmission: highly contagious, shed in feces, vomit, urine, saliva • Durable in the environment • Age-related severity of disease |
Feline Parvovirus
|
|
|
• Acute infection: fever (>104oF),
lethargy, depression, anorexia, mucopurulent oculonasal discharge – Panleukopenia: leukopenia, lymphopenia – Enteritis: bloody diarrhea, dehydration – Peracute death |
Feline Parvovirus
|
|
|
• Fetal or neonatal infection: in utero
to 4 weeks of age – cerebellar hypoplasia, ataxia, hypermetria – Hydrocephalus, circling – Microphthalmia, blindness • Early gestation: abortion, resorption |
Feline Parvovirus
|
|
|
parvo Prevention and Control
|
• Vaccination: MLV vaccines
• Maternal antibody: 6 to 16 weeks of age, protects, interferes with vaccination • Sanitation/disinfection: 1:10-1:30 dilution of hypochlorite/bleach, • Isolation: Separation of infected cats, duration of virus shed = months post-recovery |
|
|
Aleutian Disease of Mink
• Host range:_______________ • Etiologic agent: _________ • Diseases:_______________________________________________________ |
Aleutian Disease of Mink
• Host range: Mink, ferrets, skunks, raccoons • Etiologic agent: Aleutian Mink Disease virus • Diseases: – Neonates: acute, fatal interstitial pneumonia – Adults: persistent infection, chronic immune complexmediated disease |
|
|
Aleutian Mink
Disease virus:Immune-complex disease • Aleutian coat color homozygous mutants with a defect of ____________________ • Failure of antibody to neutralize ____ and “__________” phagocytosis of immune complexes by ____________ • Chronic plasmacytosis, hypergammaglobulinemia, splenomegaly, lymphadenopathy, vasculitis, glomerulonephritis, arteritis, anemia |
Immune-complex disease
• Aleutian coat color homozygous mutants with a defect of lysosomal membranes (Chediak-Higashi) • Failure of antibody to neutralize ADV and “frustrated” phagocytosis of immune complexes by macrophages • Chronic plasmacytosis, hypergammaglobulinemia, splenomegaly, lymphadenopathy, vasculitis, glomerulonephritis, arteritis, anemia |
|
|
• Ubiquitous virus of swine
• Etiologic agent: SMEDI, reproductive losses • Host range: swine • Transmission: Oral-nasal, venereal, virus crosses the placenta |
Porcine Parvovirus
|
|
|
Porcine Parvovirus
Clinical Presentation |
• Infertility: < 30 days, fetal death and
resorption • Abortions: 30-70 days, fetal death, mummification, stillbirths • Weak pigs: > 70 days, stunting, immune response, immunotolerance |
|
|
Porcine Parvovirus Prevention and Control
|
• Persistent infection (immunotolerant)
occurs in pigs infected >70 days of gestation, chronic shedders • Durable in the environment up to 20 weeks, resistant to many disinfectants • Vaccination: inactivated vaccines, immunity 4-6 months • Natural infection: exposure of gilts, lifelong immunity |
|
|
• Aka the minute virus of dogs
• Disease: – interstitial pneumonia of neonatal puppies, mild enteritis – Fetal death, abortions, resorption • Approximately 50% of adult dogs have antibodies |
Canine Parvovirus-1
• Genetically distinct from CPV-2 |
|
|
• Mink enteritis virus
• Kilham virus of rats • H-1 virus of rats • Minute virus of mice |
Parvoviruses
|
|
|
Coronaviridae
• Enveloped +/-, size symmetry • _____________________________genome 30kb • Pathogens of __________ and ____________ species |
Coronaviridae
• Enveloped, large peplomers, helical symmetry • Single-stranded, positive-sense, linear RNA genome 30kb • Pathogens of avian and mammalian species |
|
|
Classification
• Coronavirus (genus) – Group I: – Group II: – Group III: – Unclassified: • Torovirus: |
Classification
• Coronavirus (genus) – Group I: (mammalian) TGE, FIP/FECV, CCV – Group II: (mammalian, avian) MHV, BECV, PHEV, TBC – Group III: (avian) IBV – Unclassified: rabbit • Torovirus: Breda, Berne |
|
|
Coronaviridae
Pathophysiologic Classification 1. Enteritis (diarrhea): 2. Respiratory: 3. Systemic: |
Pathophysiologic Classification
1. Enteritis (diarrhea): FECV, TGE, BECV 2. Respiratory: IBV, PHEV, BRCV 3. Systemic: FIPV, MHV |
|
Coronaviral Diseases
|
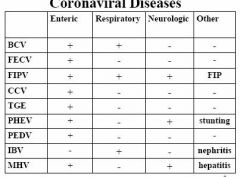
Coronaviral Diseases
|
|
|
Coronaviruses of Swine
1. 2. 3. 4. |
Coronaviruses of Swine
1. Transmissible gastroenteritis virus (TGEV) 2. Porcine respiratory coronavirus (PRC) 3. Vomiting and wasting disease, porcine hemagglutinating encephalomyelitis virus (PHEV) 4. Porcine epidemic diarrhea (PEDV) |
|
|
• Host range: Domestic swine
• Etiologic agent family: cornaviridae • Disease: Highly contagious diarrhea of piglets, occurs in explosive epidemics • Transmission: fecal-oral route • Infection is restricted to the gastrointestinal tract • Shedding in feces up to 2 weeks |
Transmissible Gastroenteritis
|
|
|
Coronaviruse
• Severe disease in piglets <3weeks of age • Vomiting, profuse diarrhea, dehydration • Weight loss, gaunt pigs • Death in 2 to 5 days • High mortality (up to 100%) in piglets <2 weeks of age • Older animals generally have shorter duration of diarrhea and low mortality |
Transmissible Gastroenteritis
(TGEV) cornaviridae |
|
|
Transmissible Gastroenteritis
(TGEV) cornaviridae Pathogenesis |
• Gastric pH is relatively neutral in
neonates and is buffered by milk • Virus replicates rapidly in mature enterocytes at the villous tips • Villi are blunted and replenished with immature enterocytes • Decreased digestive enzyme expression and absorption of nutrients leads to increased osmolarity of intestinal contents and efflux of water into the lumen |
|
|
Transmissible Gastroenteritis
(TGEV) cornaviridae Prevention and Control • _________ antibodies (________ immunity) play an important role in the severity of the disease • ___________(colostrum, milk) is protective • Systemic ________ is not protective • Maternal lymphocyte trafficking from the gut to mammary gland results in ________ secretion in colostrum/milk • Oral inoculation of sows with ____________ stimulates production of TGEV-specific ___ in milk |
Prevention and Control
• Maternal antibodies (lactogenic immunity) play an important role in the severity of the disease • Mucosal IgA (colostrum, milk) is protective • Systemic IgG is not protective • Maternal lymphocyte trafficking from the gut to mammary gland results in IgA secretion in colostrum/milk • Oral inoculation of sows with dead piglets stimulates production of TGEV-specific IgA in milk |
|
|
• Host range: Domestic swine
• Etiologic agent: Deletion mutant of TGEV lacking a single epitope of the E2 (Spike) protein • Transmission: respiratory secretions • Disease: subclinical respiratory infection |
Porcine respiratory coronavirus
|
|
|
• Host range: domestic swine
• Etiologic agent: __________ • Transmission: inhalation of aerosolized respiratory secretions • Diseases: acute vomiting and wasting of young pigs < 3weeks of age, neurologic disease, stunting in survivors |
Porcine hemagglutinating
encephalomyelitis hemagglutinating encephalomyelitis virus (HEV |
|
|
• Vomiting, wasting and acute
encephalomyelitis in young pigs • Anorexia, hyperesthesia, muscle tremors, paddling • Vomiting, depression, emaciation • Mortality: up to 100% • Must be distinguished from porcine polioencephalomyelitis virus, porcine circovirus-2 |
Porcine hemagglutinating
encephalomyelitis hemagglutinating encephalomyelitis virus (HEV coronaviridae |
|
|
• Etiologic agent: a porcine coronavirus
unrelated to TGEV • Geographic range: Europe and Asia • Clinical presentation: similar to TGE but usually less severe • Mortality = 50 to 90% |
Porcine epidemic diarrhea
|
|
|
• Ubiquitous infection of cattle
• Host range: domestic cattle, elk • Transmission: fecal-oral route • Disease: Malabsorptive diarrhea • Prevention/Control: maternal immunity |
Bovine coronavirus enteritis
Etiologic agent: bovine coronavirus |
|
|
• Major cause of diarrhea in calves
• Occurs 4 to 5 days post-ingestion • Affects calves 3 to 21 days of age with a peak age of 1 week • Profuse liquid diarrhea, hypoglycemia, lactic acidosis, hypovolemia, shock, death |
Bovine coronavirus enteritis
|
|
|
Winter dysentery
|
• Sporadic disease of adult cattle
• Etiologic agent: not determined, coronaviruses implicated, BVDVassociated with severe disease • Clinical Presentation: Profuse watery to bloody diarrhea, depression, anorexia, respiratory signs, decreased milk production |
|
|
• Clinical presentation: isolated from
“summer” pneumonia cases, epidemics of respiratory disease in feedlot calves • Other factors/agents involved in severe disease, e.g., BVDV |
Bovine respiratory coronavirus
• Etiologic agent: indistinguishable from bovine enteric coronaviruses |
|
|
• Host range: chickens
• Transmission: aerosolized respiratory secretions, highly contagious • Diseases: strain dependent, multiple serotypes – Acute respiratory disease – Nephritis |
• Etiologic agent: Infectious bronchitis
virus (IBV) |
|
|
• Infectious bronchitis:
– High morbidity, low mortality – Sneezing, coughing, oculonasal discharge, dyspnea • Decreased egg production: to <50%, egg & shell quality, egg yolk peritonitis, permanent damage to the oviduct • Nephritis: – High mortality – Pasty vents, urolithiasis, pale kidneys |
Avian Infectious Bronchitis
|
|
|
Avian Infectious Bronchitis
Prevention and Control |
• Shedding by carriers for months after
infection • IBV can persist up to 4 weeks outside the host • Vaccination with attenuated or inactivated virus |
|
|
• A sporadic, fatal disease of cats
• Host range: Felidae ubiquitous coronavirus • Transmission: Fecal-oral or respiratory • Age distribution: Disease of cats < 1 year old or >13 years • Target cell is the macrophage |
Feline infectious peritonitis
|
|
|
____________is a mutation of
an enteric feline coronavirus (FECV), a ubiquitous coronavirus, type I and II |
FIPV
|
|
|
Risks associated with FIP
• Age of initial infection with FECV: – ______________ – ______________ • High dose of ______ • Defects of ____ – _________________, e.g., FeLV, FIV or prior FPV infection – Genetic predisposition, e.g., Persian cats, cheetahs |
Risks associated with FIP
• Age of initial infection with FECV: – <16 weeks of age – >13 years of age • High dose of FECV • Defects of CMI – Immunosuppression, e.g., FeLV, FIV or prior FPV infection – Genetic predisposition, e.g., Persian cats, cheetahs |
|
|
FIP
Pathogenesis • Mutation in _____ yields ____ that replicates in macrophages • Systemic spread in _______ • Defective _____ leads to persistent FIPV replication • ________________ stimulation leads to production of binding Abs • FIPV + Ab = _____________ • Immune-mediated vasculitis (Type ________) |
Pathogenesis
• Mutation in FECV yields FIPV that replicates in macrophages • Systemic spread in macrophages • Defective CMI leads to persistent FIPV replication • Chronic antigenic stimulation leads to production of binding Abs • FIPV + Ab = immune-complex formation • Immune-mediated vasculitis (Type III/IV) |
|
|
FIP
Clinical Presentation 1. Wet or effusive form: __________________ – Abdominal or thoracic ______, with elevated ________ content, >35 g/dl – Hyper________ – Hypo_________ • Chronic fever of ______ origin • Anorexia, depression, anemia, weight loss • _________ with thoracic effusion |
Clinical Presentation
1. Wet or effusive form: severe vasculitis and leakage – Abdominal or thoracic effusion, with elevated protein content, >35 g/dl – Hypergammaglobulinemia – Hypoalbuminemia • Chronic fever of unknown origin • Anorexia, depression, anemia, weight loss • Dyspnea with thoracic effusion |
|
|
FIP
Clinical Presentation 2. Dry or non-effusive form: • ______ fever, wasting • Renal disease, ________ • Hepatic disease, _______ • Anterior _______ • Neurologic signs • Pathologic lesions: – _________________ – _________________ |
Clinical Presentation
2. Dry or non-effusive form: • Chronic fever, wasting • Renal disease, dehydration • Hepatic disease, icterus • Anterior uveitis • Neurologic signs • Pathologic lesions: – Moderate vasculitis – Granuloma formation |
|
|
FIP
Control and Prevention |
Control and Prevention
• FeCV infections usually occur very early in life; test new introductions • FeCV is transmitted from cat to cat; reinfection occurs; persistent shedders maintain the infection in multiple cat households • FIPV is not transmitted from cat to cat • FIP has an immunopathologic component • FIP vaccination may enhance the risk of disease |
|
|
• Host range: Canidae
•antigenically related to FECV • Transmission: fecal-oral • Disease: Acute diarrhea of dogs, more severe in puppies, subclinical infections • Most infections occur by 4 to 12 wks of age • Common infection of dogs, 70% seroprevalence |
Canine coronavirus enteritis
• Etiologic agent: Canine coronavirus |
|
|
dogs
• Nonfatal, self-limiting diarrhea • Frequently inapparent • Incubation period of 1 to 4 days • Acid-stable, remain infective following passage through gastric acid • Replicates in mature enterocytes of villous tips • Shedding occurs for up to 2 weeks • Recovery in 7 to 10 days |
Canine coronavirus enteritis
• Etiologic agent: Canine coronavirus |
|
|
• Host range: mice
multiple strains • Diseases: Enteritis, hepatitis, demyelinating encephalomyelitis, nephritis • Transmission: introduction of carriers, fomites, wild mice • Clinical: diarrhea with high mortality, jaundice, wasting and paralysis |
• Etiologic agent: mouse hepatitis virus
a coranavirus |
|
|
• Host range: rats
• Disease: inflammation of the salivary and nasolacrimal glands, self-limiting • Transmission: direct contact, fomites, aerosolized ocular secretions • Clinical: lacrimation, exophthalmus, squinting, corneal ulcers, swelling of eye • Age: severe disease in young rats |
• Etiologic agent: sialodacryadenitis virus
a coronavirus |
|
|
Toroviruses
• Host range: • Transmission: • Disease: • Replication: |
Toroviruses
• Host range: Horses, humans, cattle, pigs, turkeys • Transmission: presumed fecal-oral • Disease: diarrhea in humans, cattle, pigs and turkeys; inapparent infections in horses • Replication: difficult to culture or uncultivable |
|
|
• Distribution: >80% seroprevalence
• Mild to moderate diarrhea in calves up to 10 months of age • Infects infects the mid- and lower parts of intestinal villi, including the crypt cells, of small and large intestines • Diarrhea 1 to 3 days post-inoculation • Depression, anorexia • Dehydration and death in severe cases |
Torovirus diarrhea of cattle
|
|
|
• Emerged in Nov. 2002,
Guangdong, China • Reservoir host: unknown, civet cats, raccoon dogs • Clinical disease in humans: severe, acute interstitial pneumonia, pulmonary edema, hypoxia, respiratory failure |
Severe Acute Respiratory Syndrome
(SARS) coronavirus |
|
|
Innate Immunity
1. Anatomic: _________ 2. Physiologic: ____________ 3. ________ and _______ cells 4. Interferons and cytokines 5. Complement 6. Presence of receptors, enzymes, etc. to support virus replication |
Innate Immunity
1. Anatomic: skin, mucosa 2. Physiologic: gastric acid, lysozymes in tears 3. Natural Killer (NK) cells, and macrophages 4. Interferons and cytokines 5. Complement 6. Presence of receptors, enzymes, etc. to support virus replication |
|
|
Characteristics of Innate
Immunity |
• No antigen specificity
• No memory • No MHC class restriction • No antibody dependence |
|
|
Macrophages
• Target cells, infected by viruses, viral Ag presented in the context of ____ • Perform ______________ • Antigen presenting cells, viral Ag presented in the cleft of _________ • Release __________ involved in macrophage and T cell activation: __________ • Cause tissue injury:___________________________________ • Release _________ factors involved in angiogenesis, fibrosis, healing. |
Macrophages
• Target cells, infected by viruses, viral Ag presented in the context of MHC I • Perform phagocytosis • Antigen presenting cells, viral Ag presented in the cleft of MHC II • Release cytokines involved in macrophage and T cell activation: IL-1, IL-2, TNF, IFNs • Cause tissue injury: lysosomal enzymes, reactive oxygen, NO, prostaglandins, leukotrienes • Release growth factors involved in angiogenesis, fibrosis, healing. |
|
Interferons
|
Interferons
|
|
|
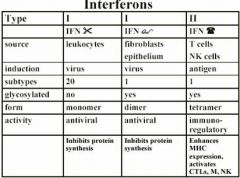
Interferons
• No _________ specificity • __________ specific • Produced ________ in the course of infection • Protect ___________ cells • Inhibit _________ spread of virus • Production/treatment correlates with recovery • Inhibition of production leads to _____________ |
Interferons
• No viral specificity • Species specific • Produced early in the course of infection • Protect neighboring cells • Inhibit early spread of virus • Production/treatment correlates with recovery • Inhibition of production leads to severe disease |
|
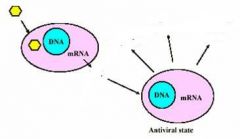
Antiviral activities of IFN
|
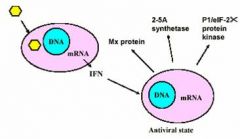
Antiviral activities of IFN
|
|
|
Complement
• ________ and _______ proteins • Activation results in amplification of products that: –______________________ (____________________) [C6,C7,C8,C9] –______________________ [C3a,C4a,C5a] –_______________ [C3b] –Solubilize immune complexes |
Complement
• Serum and membrane proteins • Activation results in amplification of products that: –Form pores in membranes (membrane attack complex) [C6,C7,C8,C9] –Mediate inflammation [C3a,C4a,C5a] –Act as opsonins [C3b] –Solubilize immune complexes |
|
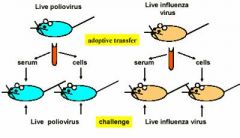
what happens to the mice
|
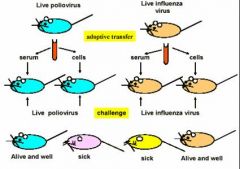
type of immunity
|
|
|
Acquired Immunity
|
• Inducible protection against
infection, disease or death • Requires time to develop • Specific for antigen • Responds to many antigens • Memory • Humoral vs cell-mediated components |
|
|
Anti-Viral Actions of Antibodies
1. 2. 3. 4. |
Y
Anti-Viral Actions of Antibodies 1. Neutralization – Prevents virus from binding to receptor – Prevents virus from uncoating 2. Opsonization 3. Antibody dependent cell-mediated cytoxicity – Macrophages, neutrophils, K cells 4. Complement activation |
|
|
Cell-Mediated Immunity
1. 2. 3. 4. |
Cell-Mediated Immunity
1.T helper cells (CD4+) a.Th1 (inflammatory) b.Th2 (helper) 2.T cytotoxic (CD8+) 3.T suppressor (CD8+) 4.T delayed type hypersensitivity (CD4+/CD8+) |
|
|
MHC
Endogenous Pathway • Proteins made in the cytosol are digested by __________ into ___________ • _____ transports the peptides to the ____ • _______ molecules associate with the peptides • MHC I + peptide complex is _______ to the cell’s ________ |
Endogenous Pathway
• Proteins made in the cytosol are digested by proteosomes into peptides • TAP transports the peptides to the ER • MHC I molecules associate with the peptides • MHC I + peptide complex is exported to the cell’s surface |
|
|
MHC
Exogenous Pathway • _________ of extracellular proteins • __________ enzyme digestion produces peptides • ______ molecules are transported to the endosomes and pick up the peptides • MHC II + peptide complex is ________ to the cell’s ________ |
Exogenous Pathway
• Endocytosis of extracellular proteins • Endosomal/lysosomal enzyme digestion produces peptides • MHC II molecules are transported to the endosomes and pick up the peptides • MHC II + peptide complex is exported to the cell’s surface |
|
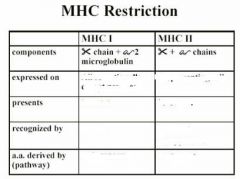
MHC Restriction
|

MHC Restriction
|
|
fill in the chart
|
out come of viral infection
|
|
|
Gell-Coombs Classification of
Hypersensitivity |
• Type I, Anaphylaxis
• Type II, Cytotoxic • Type III, Immune complex, Arthus reaction • Type IV, Cell-mediated, delayed hypersensitivity |
|
|
type of hypersensitivity?
• IgE + viral antigen release of granules from mast cells containing histamine, heparin, serotonin and plasma kinins • May account for erythema, congestion, swelling in upper respiratory tract post-RSV infections • Important in hypersensitivity due to proteins present in vaccines |
Type I, Anaphylaxis
|
|
|
Type of hypersensitivity?
• IgM/IgG + viral antigen on the cell surface results in cell destruction • May account for hemolysis during acute infection with EIAV leading to anemia • May account for liver necrosis in hepatitis B or yellow fever |
Type II, Cytotoxic
|
|
|
Type of hypersensitivity?
Y Y • IgG + viral antigen leads to immune complex formation • Lattice formation dependent on relative amounts of IgG and antigen • Dependent on Ig affinity, presence of complement, ability of the RE system to remove complexes by phagocytosis • Activation of complement • Activation of neutrophils |
Type III, Immune Complex
|
|
|
Type III, Immune Complex
• e.g., Aleutian disease of _____ • ____virus • _________ infection • Immune complex formation • Deposition in ________, __________, ______, ________ |
Type III, Immune Complex
• e.g., Aleutian disease of mink • Parvovirus • Persistent infection • Immune complex formation • Deposition in renal glomeruli, skin, synovium, choroid plexus |
|
|
Type of hypersensitivity?
• Virus-specific T lymphocytes + viral antigens presented in the context of MHC • Inflammation in an unfortunate location • E.g., CTLs and encephalitis in LCMV infection in adult mice, or Borna disease virus in rats |
Type IV, Cell-mediated
|
|
|
• An immune response directed
against normal host components which results in tissue injury • Can be primarily a humoral immune response • Can be primarily a cell-mediated immune response • Vaccine-induced suspect cases |
Autoimmunity
|
|
|
Viral Diagnostics
|
• Infectious virus
• Whole virions • Viral proteins • Viral RNA/DNA • Serology |
|
|
Virus Isolation
ADVANTAGES DISADVANTAGES |

Virus Isolation
|
|
|
Animal Inoculation
|
• Some viruses do
not replicate in cell culture and only grow by this method • Expensive • Slow • Cumbersome |
|
|
Inoculation of embryonated eggs
|
• Growth of
unknowns especially avian viruses & chlamydia • Sensitive • Expensive • Slow • Technical expertise and equipment needed |
|
|
Inoculation of cell culture
|
• Sensitive
• Allows identification of unknown agents • Allows recovery of viruses for further study • Expensive • Slow |
|
|
Cytopathology
|
• Cytoplasmic vacuolation
• Changes in cell shape • Cell death • Syncytia formation • Transformation |
|
|
Electron Microscopy viral detection
|
• Based on viral
morphology • Used to detect whole virions in feces • Sensitive • Rapid • Expensive, equipment and technical time • Technically difficult |
|
|
Polymerase Chain Reaction (PCR) for viral dection
|
Samples Samples MW MW
POS POS 1 2 3 1 2 3 •Sensitive •Specific •Applicable to bulk or pooled samples •Expensive •Technical considerations •Subject to false positives due to DNA contamination •Subject to false negatives due to inhibitors of reagents •Requires knowing the viral genetic sequence |
|
|
Radioimmunoprecipitation
|

type of viral test?
|
|
|
types of Serology for viral testing
|
• Immunological footprints
• Serum neutralization (SN, VN) • Enzyme-linked immunosorbent assay (ELISA) • Agar gel immunodiffusion (AGID) • Hemagglutination inhibition (HI) • Acute vs convalescent samples • IgM vs IgG |
|
|
Reoviridae
•envelope +/- shape:_______ • ____________________ genome • __________ and __________ pathogens |
Reoviridae
• Non-enveloped, double capsid, 80 nm icosahedral virus • Double-stranded, 10- 12 segmented RNA genome • Arthropod-borne and enteric pathogens |
|
|
reovirus
Classification 1. __________(genus): African horse sickness, bluetongue viruses, epizootic hemorrhagic disease of deer, Ibaraki virus 2. ____________: rotaviruses of many species 3. ____________: mammalian and avian reoviruses 4. ___________: Colorado tick fever virus 5. __________: viruses of fish and invertebrates |
Classification
1. Orbivirus (genus): African horse sickness, bluetongue viruses, epizootic hemorrhagic disease of deer, Ibaraki virus 2. Rotavirus: rotaviruses of many species 3. Orthoreovirus: mammalian and avian reoviruses 4. Coltivirus: Colorado tick fever virus 5. Aquareovirus: viruses of fish and invertebrates |
|
|
reovirus
• Arthropod-borne, Culicoides spp. • Tropism: reticuloendothelial cells, endothelial cells • Pathogenesis: vasculitis, vascular leakage • Pathologic lesions: edema, hemorrhage |
Orbiviruses
|
|
|
• Host range: Equidae, dogs, elephants,
zebras, camels, (humans) • Etiologic agent: (1-9 serotypes), multiple strains • Exotic to the U.S. • Disease: Highly lethal disease of horses, 90-95% mortality • Transmission: Culicoides spp. • Reservoir: unknown, zebras (?) |
African Horse Sickness
(Orbiviruses) |
|
|
African Horse Sickness (Orbiviruses)Clinical Presentation-Horses
|
1. Pulmonary form: fully susceptible horses
• Fever for 1 to 2 days • Dyspnea, coughing, frothy nasal discharge, peracute death • Mortality rate approaches 100% 2. Cardiac form: donkeys, vaccinated horses • Fever for 5 to 8 days • Edema of supraorbital fossae, head • Colic, subacute course • Mortality rate = 50-70% |
|
|
African Horse Sickness (Orbiviruses)Pathogenesis
|
• Culicoides bite
• Replication in local lymph nodes • Primary viremia • Infection of reticuloendothelial cells • Secondary viremia • Replication in endothelial cells • Vasculitis, damage to blood vessels • Pulmonary edema, petechial hemorrhages in pericardium |
|
|
Orbiviruses
• Host range: sheep, camelids, bighorn sheep, goats, cattle, deer, bison, elk, pronghorn, other wild ruminants, (dogs) •1-25 serotypes • Disease: clinical disease in sheep, subclinical infections in cattle & goats, hemorrhagic disease in deer and pronghorn • Transmission: Culicoides spp., • Target cells: hematopoietic cells, endothelial cells |
Bluetongue
|
|
|
• Fever, “wool break”
• Salivation, hyperemia of oral mucosa, oral ulcers, frothing of the mouth • Cyanosis of tongue • Nasal discharge, serous to mucopurulent • Edema of muzzle, ears, head, neck • Coronitis, lameness, recumbency • High morbidity, low mortality |
Bluetongue
|
|
|
bluetongue Reproductive effects - sheep
|
• Fetal infection
• Congenital defects: hydrocephalus, hydrancephaly, porencephaly, mummification, arthrogryposis • Abortion • Transient infertility in rams • Reduced lamb crop |
|
|
Culicoides varipennis and blue tongue virus
|
• “no-see-ums”, midges
• Late summer to fall • Breed in damp organic material • Virus is acquired in a blood meal from viremic animal • Replicates in midge • Shed in saliva during every blood meal |
|
|
bluetongue Prevention and Control
|
• Found throughout W. & S.E. U.S.
• Reservoirs: Maintained on RBCs of infected cattle and sheep for prolonged intervals • Non-tariff trade barrier: restriction to trade with New York, Canada and other countries • Control of Culicoides breeding habitat |
|
|
• Host range: WTD, mule deer,
pronghorn, reservoir host? • found in the western and S.E. U.S. • Disease: acute hemorrhagic fever • Transmission: Culicoides spp. |
Epizootic Hemorrhagic Disease
of Deer |
|
|
• Deer carcasses in creeks, late
summer/fall • Fever, anorexia, ataxia • Lethargy, weakness • Salivation, oral ulcers • Hyperemia of mucous membranes, skin • Edema of conjunctiva, head and neck • Lameness, hemorrhage of coronary band |
Epizootic Hemorrhagic Disease
of Deer |
|
|
Rotavirus
• shape:________ •______________________ genome • Stable in the environment • Stable at __ pH • Resistant to ________ solvents |
Rotavirus
• Double-shelled icosahedron (80 nm) • ds RNA, 10-12 segmented genome • Stable in the environment • Stable at low pH • Resistant to lipid solvents |
|
|
• Rotavirus diarrhea, “milk scours”
• Host range: cattle, sheep, pigs, horses, rabbits, rodents, humans • Transmission: fecal-oral route • Disease: malabsorptive diarrheas, dehydration, acid-base imbalance |
Rotavirus enteritis
|
|
|
Rotavirus Classification
• Based on genotype and group-specific viral antigen (VP6) • Group A: • Group B: • Group c: • Group D: • Group E: • Group F: |
• Based on genotype and group-specific viral
antigen (VP6) • Group A: humans, cattle, other species • Group B: humans • Group C: swine • Group D: fowl • Group E: swine • Group F: fowl |
|
|
Rotavirus enteritis Pathogenesis
|
• Infection and cytolysis of mature
enterocytes at the tips of the villi • Exposure of the basement membrane • Migration of immature enterocytes • Blunting & fusion of adjacent villi • Reduced absorptive surface/function • Transient malabsorption |
|
|
• Rotavirus infection specifically and
selectively decreases ________ expression on the ___________ • A functional __________ peptide secreted from rotavirusinfected cells. |
• Rotavirus infection specifically and
selectively decreases sucraseisomaltase expression on the apical brush border • A functional NSP4 enterotoxin peptide secreted from rotavirusinfected cells. |
|
|
Rotavirus enteritis
Factors influencing disease • Proteases:____________________________________________ • Milk:_____________________________ • Age:________________________________________ • Immunity:________________________________________ |
Factors influencing disease
• Proteases: VP4 cleavage is necessary for penetration of the host cell membrane • Milk: Osmotic pressure of proteins and saccharides • Age: Age-dependent expression of receptors on enterocytes • Immunity: colostrum and milk |
|
|
Rotavirus enteritis Clinical Presentation
|
• Neonatal animals: 1 to 8 weeks of age
• Incubation period: 12 hours to 6 days • Watery diarrhea, dehydration • Anorexia, depression, vomiting • Death is due to severe dehydration secondary to fluid loss |
|
|
Rotavirus enteritis Clinical considerations
|
• Shed in large amounts, 109 to 1012
virions/gram of feces • Virus shedding: up to 14 days p.i. • Spread on milk buckets, utensils, boots, hands • Not inactivated by chlorination |
|
|
Rotavirus enteritis Contributing factors
|
• Lack of maternal immunity, not
necessarily failure of passive transfer • Exacerbated by milk ingestion • Chilling, overcrowding • Lack of sanitation • Other pathogens: E. coli, coronavirus, cryptosporidia |
|
|
• Zoonotic disease
• Fever, myalgia, headache, meningoencephalitis, hemorrhagic fever • Tick-borne, D. andersonii • Virus is transmitted transtadially • Virus overwinters in nymphs and adults • Prolonged viremia in rodents |
Colorado Tick Fever
|
|
|
Bunyaviridae
• Envelope +/- Shape:__________ • genome:_________ • _________ and __________________ pathogens |
Bunyaviridae
• Enveloped, helical virus (80-120 nm) • Single-stranded, negative or ambisense, 3 RNA segments • Rodent- and arthropod-borne pathogens |
|
|
family Bunyaviridae
Classification (genera) 1. ________: Rift Valley fever virus 2. ________: Nairobi sheep disease, Crimean-Congo hemorrhagic fever 3. __________: Akabane, Cache Valley, La Crosse (California encephalitis) 4. ___________: Sin Nombre, Prospect Hill, etc. |
Classification (genera)
1. Phlebovirus: Rift Valley fever virus 2. Nairovirus: Nairobi sheep disease, Crimean-Congo hemorrhagic fever 3. Bunyavirus: Akabane, Cache Valley, La Crosse (California encephalitis) 4. Hantavirus: Sin Nombre, Prospect Hill, etc. |
|
|
• Host range: cattle, sheep, humans,
wildlife species • Diseases: hemorrhagic fever, hepatitis, abortion, encephalitis • Mortality: – 90% in lambs – 20-60% in adult sheep – 90-100% abortion rate in ewes, cows • Geographic range: Sub-Saharan Africa, Egypt |
Rift Valley Fever
|
|
|
Rift Valley Fever Transmission
|
• Mosquito-borne, Culex, Aedes spp.
– transmitted transovarially – eggs survive for extended periods • Ingestion of contaminated meat • Handling of contaminated carcasses |
|
|
• Host range: cattle, sheep, humans,
wildlife species • Entry via mosquito bite or oropharynx • Incubation of 30 to 72 hours during which virus replicates to high titers • Virus spreads to the liver and reticuloendothelial organs • Hepatic necrosis • Hemorrhages in GI tract and subQ tissues • Encephalitis, neuronal necrosis, perivascular cuffing |
Rift Valley Fever
|
|
|
• Host range: sheep and goats
• Vertebrate reservoir host: ? • Transmission: Rhipicephalus appendiculatus, transstadial/transovarial transmission, persistent infection of adult ticks • Disease: Fever, hemorrhagic enteritis, abortion • Mortality = 30-90% • Geographic range: east Africa, Nigeria, India 14 2. Crimean-Congo Hemorrhagic Fever • Host range: humans • Disease: severe hemorrhagic fever, necrotizing hepatitis, myocarditis, encephalitis • Reservoir hosts: – Small mammals and birds – Wild and domestic ruminants, sheep, goats, cattle |
Nairobi sheep disease
|
|
|
• Host range: humans
• Disease: severe hemorrhagic fever, necrotizing hepatitis, myocarditis, encephalitis • Reservoir hosts: – Small mammals and birds – Wild and domestic ruminants, sheep, goats, cattle |
Crimean-Congo Hemorrhagic
Fever |
|
|
Crimean-Congo Hemorrhagic
Fever Transmission |
• Tick-borne, Hyalomma spp.,
transovarial/transtadial • Direct contact with infected animals • Human to human contact, e.g., nosocomial infections |
|
|
• Host range: cattle, sheep, goats
• Diseases: developmental defects of the spine, joints, e.g., arthrogryposis, neurologic defects, e.g., hydranencephaly, abortions, fetal death • Transmission: mosquito- & Culicoidesborne virus • Geographic distribution: countries of eastern hemisphere |
• Etiologic agent: Akabane virus
|
|
|
• Host range: sheep, goats
• Disease: fetal defects, similar to Akabane, following maternal infection 30 to 60 days of gestation • Transmission: mosquito-borne • Reservoir host: deer • Geographic range: U.S. |
Cache Valley virus
|
|
|
• Host range: humans zoonotic disease
• Reservoir hosts: Apodemus agrarius, Peromyscus spp., voles, mice, rats • Persistent, inapparent infection in rodents • Transmission: – virus is shed in saliva, urine, feces (?) – infection of humans occurs via inhalation |
Hantaviruses
|
|
|
Hantavirus diseases
1. Hemorrhagic fever with renal syndrome: • • • 2. Hantavirus pulmonary syndrome: • • |
Hantavirus diseases
1. Hemorrhagic fever with renal syndrome: • Fever, headache • Hemorrhages in multiple organs • Acute renal failure with shock 2. Hantavirus pulmonary syndrome: • Acute respiratory distress syndrome • Fever, myalgia, headache • Nonproductive cough, shortness of breath |
|
|
Arenaviridae
• Pleomorphic, envelope +/- • 50-300 nm • _______________genome, ambisense • __________ reservoirs • Zoonoses |
Arenaviridae
• Pleomorphic, enveloped • 50-300 nm • Single-stranded RNA genome, ambisense • Rodent reservoirs • Zoonoses |
|
|
Family?
• Pleomorphic • Envelope containing glycoproteins • 2 single-stranded, ambisense RNA segments, L, S • Circularized nucleocapsids • Contains host cell ribosomes |
Arenaviridae
|
|
|
Arenaviridae
Classification • Lymphocytic choriomeningitis virus • ______________ – Lassa fever virus • _____________ – Junin, Machupo, Sabia, Guanarito, Tacaribe, etc. |
Classification
• Lymphocytic choriomeningitis virus • Old World – Lassa fever virus • New World – Junin, Machupo, Sabia, Guanarito, Tacaribe, etc. |
|
|
• Highly host-adapted
• Usually inapparent infection in rodents • Vertical transmission • Persistent infection • Shed virus in urine, saliva, feces • Important infection in laboratory rodents and “pocket pets” |
Arenavirus infections in rodents
|
|
|
• Host species: mice, hamsters
• Clinical presentation: – Usually asymptomatic – Runting, failure to thrive –Immunopathologic disease, glomerulonephritis |
Lymphocytic choriomeningitis
virus |
|
|
Lymphocytic choriomeningitis
virus Zoonosis • Clinical disease – – – – • At risk: – – |
• Clinical disease
– “flu-like” illness, fever, headache – Aseptic meningitis – Severe encephalomyelitis – Intrauterine infections leading to fetal death and congenital defects of brain • At risk: – children handling hamsters as pets – laboratory animal workers |
|
|
Birnaviridae
• envelope +/-, shape: stable at __ C for __ min and pH ______ • Genome: • Pathogens of ______, ____ and __________ |
Birnaviridae
• Non-enveloped, icosahedral virus (60 nm in diameter); stable at 60 C for 60 min and pH 3-9 • 2 linear, double-stranded, RNA segments, 6 kb in length • Pathogens of birds, fish and shellfish |
|
|
Birnaviridae Classification
1. _____________ (genus) – Infectious bursal disease virus 2. ______________ (genus) – Infectious pancreatic necrosis virus 3. _____________ (genus) |
Classification
1. Avibirnavirus (genus) – Infectious bursal disease virus 2. Aquabirnavirus (genus) – Infectious pancreatic necrosis virus 3. Entomobirnavirus (genus) |
|
|
• aka Gumboro disease
• Host range: variety of avian species, chickens, turkeys, penguins , 2 serotypes • Diseases: acute diarrheal disease of young chicks, immunosuppression in poultry • Geographic distribution: worldwide |
• Etiologic agent: Infectious bursal disease virus
|
|
|
Infectious bursal disease virus
Clinical Presentation |
• Morbidity up to 100%
• 3 to 6 weeks of age • Mortality from 0 to 30% • Vent picking, soiled vent feathers • Anorexia, depression • Diarrhea, dehydration • Trembling, prostration |
|
|
Infectious bursal disease virus Pathogenesis
• Following _______ inoculation, IBDV infects _________ and ______ cells in the intestines • Replicates in the ______ • Results in primary viremia and spread to the ___________________, • Lymphotropic for _____ cells • Secondary massive viremia and spread to other organs • Immunosuppression due to ___________________ responsiveness |
Pathogenesis
• Following oral inoculation, IBDV infects macrophages and lymphoid cells in the intestines • Replicates in the liver • Results in primary viremia and spread to the bursa of Fabricius, • Lymphotropic for B cells • Secondary massive viremia and spread to other organs • Immunosuppression due to reduced humoral immune responsiveness |
|
|
Infectious Bursal Disease Pathology
|
• Bursal edema, hemorrhages,
necrosis, atrophy • Watery to bloody diarrhea, dehydrated carcass • Splenomegaly; necrosis of lymphoid tissues including thymus, spleen, cecal tonsils, bone marrow; hemorrhages in the proventriculus and gizzard |
|
|
Infectious Bursal Disease Transmission
|
• Highly contagious viral infection
• Fecal-oral route • Virus persists in the environment up to 4 months • Harbored in meal worms for weeks • Resistant to most disinfectants |
|
|
Infectious Bursal Disease
prevention and control |
• Maternal immunity is important in
reducing susceptibility of chicks • Vaccination of breeders is the most effective method of preventing the disease in chicks • Vaccination of chicks • Sanitation is difficult • Good husbandry, e.g., environmental temperature |
|
|
Filoviridae
• ____________ symmetry, envelope +/- • _____________________ genome 19.1 kb • Pathogens of __________, _________________ |
Filoviridae
• Helical symmetry, enveloped, 80 x 800 to 1000 nm • Single-stranded, negative-sense, RNA genome 19.1 kb • Pathogens of primates, hemorrhagic fevers |
|
|
Filoviridae
Classification |
• Marburg-like virus (genus)
• Ebola-like virus – Zaire (subtype) –Sudan – Côte d’Ivoire – Reston |
|
|
Ebola Hemorrhagic Fever
• Host species: • Reservoir: • Etiologic agent: • Diseases: • Transmission: |
Ebola Hemorrhagic Fever
• Host species: primates • Reservoir: unknown • Etiologic agent: Ebola-like viruses • Diseases: hemorrhagic fever • Transmission: – Blood, secretions |
|
|
Bornaviridae
• Envelope +/- , shape: • 90 nm in diameter • _____________________ genome, 8.9kb • host range ________ |
Bornaviridae
• Enveloped, spherical virus • 90 nm in diameter • Single-stranded, negative-stranded RNA genome, 8.9kb • Wide host range |
|
|
Bornaviridae Classification
|
• Mononegavirales (order)
– Bornaviridae (family) • Bornavirus (genus) |
|
|
• Species affected: horses, sheep, cattle,
cats, rabbits, ostriches, dogs • Reservoir: birds?, rodents? • Disease: encephalomyelitis • Endemic: Germany, Iran, Israel, Japan, Sweden, Switzerland, U.K. • Exotic to the U.S.? |
• Etiologic agent: Borna disease virus
|
|
|
Borna disease virus
Clinical presentation-Equine • Prodromal phase: • Neurologic phase: – Limbic system: – Opthalmologic: |
Clinical presentation-Equine
• Prodromal phase: fatigue, colic, fever, coughing icterus • Neurologic phase: – Limbic system: excitability , somnolence, ataxia, hyperesthesia – Motor system: paresis, paralysis – Opthalmologic: nystagmus, pupillary reflex dysfunction, blindness |
|
|
• Mononegavirales (order) horses
• Usually fatal • Progression to death in 3 days to 3 weeks • Survivors have permanent neurologic deficits |
Borna Disease
|
|
|
Borna Disease Pathogenesis
|
• Acquired by intranasal route
• Passes into the CNS via olfactory nerves • Replicates in neurons, astrocytes and oligodendrocytes • Severe encephalomyelitis |
|
|
Borna Disease Pathology
|
• Lesions in the gray matter of the
olfactory bulb, basal cortex, caudate nucleus and hippocampus • Lymphocytic perivascular cuffing • Intranuclear, eosinophilic inclusions (Joest-Degen bodies) • Necrosis leading to hydrocephalus, cortical atrophy |
|
|
Borna Disease Immunity
|
• Cell-mediated immune response in
immunocompetent adults contributes to the immunopathology of disease • Humoral immune response is not protective • Vaccine is not available |
|
|
Arteriviridae
• Envelope+/- shape/size: • _________________ genome, 13-15 kb • Pathogens of _______, ______, ______, _________ • Macrophage, endotheliotropic |
Arteriviridae
• Enveloped, icosahedral virus, 50-70 nm in diameter • Single-stranded, positive-sense, RNA genome, 13-15 kb • Pathogens of swine, horses, primates, rodents • Macrophage, endotheliotropic |
|
|
Arteriviridae Classification
|
• Arterivirus (genus)
– Equine arteritis virus – Porcine respiratory and reproductive syndrome virus – Lactate dehydrogenase elevating virus – Simian hemorrhagic fever virus |
|
|
• Contagious viral disease of Equidae
• Etiologic agent: isolated from an outbreak of respiratory disease and abortions in Bucyrus, OH (1953) • Diseases: respiratory disease, abortions • Transmission: – Aerosolized respiratory secretions – Aborted fetuses, placenta, fluids – Venereal route via virus+ semen |
Equine viral arteritis
|
|
|
Equine viral arteritis
• Worldwide ______________ • Variation in seroprevalence between _______ and ______________ – 70 to 90% seroprevalence in _________________ – 2 to 3 % seroprevalence in _________________ • Increased prevalence due to increased __________ & __________; international trade in horses and semen |
• Worldwide distribution
• Variation in seroprevalence between breeds and geographic regions – 70 to 90% seroprevalence in Standardbreds – 2 to 3 % seroprevalence in Thoroughbreds • Increased prevalence due to increased surveillance & testing; international trade in horses and semen |
|
|
equine
• Usually sub-clinical and rarely fatal • Fever up to 41oC (106oF) 3 to 14 days post-exposure, lasts 2 to 9 days • Depression, anorexia • Leukopenia • Limb edema • Stiffness of gait |
Equine viral arteritis
|
|
|
equine
• Nasal and/or lacrimal discharge • Conjunctivitis and/or rhinitis • Periorbital/supraorbital edema • Edema of prepuce or mammary glands • Urticaria, papules & erosions on mucous membranes • Death in foals due to interstitial pneumonia or pneumoenteritis |
Equine viral arteritis
|
|
|
Equine viral arteritis
Pathogenesis, general • Virus replication in _____________ • Systemic spread via _______ • Infection of multiple __________ and _________ • Damage to endothelium leads to ________, ________ and _________ • Infarction of multiple organs following segmental necrosis of _____________ • ________, _________ shock |
Pathogenesis, general
• Virus replication in alveolar macrophages • Systemic spread via viremia • Infection of multiple organs and endothelial cells • Damage to endothelium leads to edema, congestion and hemorrhage • Infarction of multiple organs following segmental necrosis of small arteries • Hypovolemic, hypotensive shock |
|
|
Equine viral arteritis Clinical presentation, abortions
|
• Mares infected via respiratory tract
• Occurs with/without clinical signs in the mare • Abortions occur 10 to 30 days postinfection • Occurs 3 to 10 months of gestation • Abortion rates of 10 to 60% of mares |
|
|
Equine viral arteritis Pathogenesis, abortions
|
• Mares bred to infected stallions or
inseminated with infected semen do not experience any virus-related fertility problems • Pyrexia 6 to 8 days after venereal exposure • Spread of virus to the respiratory tract • Shed the virus to other horses via respiratory secretions up to 16 days • Systemic spread via viremia • Fetal infection • Endothelial infection and damage leads to edema, fluid accumulation in peritoneal and pleural cavities, and hemorrhages • Fetuses are frequently autolysed |
|
|
Equine viral arteritis Pathogenesis, stallions
|
• Spreads to seminiferous tubules
• Persistence of virus in the accessory sex glands and vas deferens of stallions • Chronic carrier state and shed of virus in semen of infected stallions • Only a transient effect on semen quality or fertility • Virus survives in chilled or frozen semen |
|
|
Equine viral arteritis Immunity
|
• Serum neutralizing antibodies detected
1 week post-infection • Long duration of immunity following infection • Colostral antibodies are protective for foals • Vaccination protects against disease and establishment of carrier state in stallions • Vaccination does not “cure” persistently infected stallions |
|
|
Equine viral arteritis prevention and control
|
1. Biosecurity
2. Isolation of broodmares from young stock and new introductions 3. Restrict movement of exposed horses 4. Quarantine infected horses 5. Serologic testing of stallions 6. Vaccination – inactivated or MLV vaccine to prevent persistent infections in stallions |
|
|
Equine viral arteritis Differential Diagnosis
|
• Equine influenza A viruses
• Equine herpesvirus-1, -4 • Equine infectious anemia virus • Exotic viruses: African horse sickness, Getah virus • Purpura hemorrhagica |
|
|
• Blue ear, mystery pig disease
• Host range: Domestic swine • Etiologic agent: – first recognized in the U.S. and Canada (1987) – North American and European genotypes |
Porcine reproductive and
respiratory syndrome (PRRS) |
|
|
Porcine reproductive and
respiratory syndrome (PRRS) Clinical Presentation 1. 2. 3. |
Clinical Presentation
1. Transient anorexia, fever in growers, cyanosis of ears, snout, tail, vulva 2. Respiratory disease in young pigs: dyspnea, “thumping”, increased bacterial pneumonia, pre-weaning mortality 3. Reproductive disease: delayed estrus cycles, agalactia, late term abortions, weak pigs, premature births, stillbirths, mummified fetuses |
|
|
Porcine reproductive and
respiratory syndrome (PRRS)Pathogenesis and Transmission • Tropism: ______________________________________ • Shed in ___________________________________________________ • Virus persists in ________ • Transmission: ____________________________________________________________________ |
Pathogenesis and Transmission
• Tropism: alveolar macrophages, antibody-enhanced uptake of virus • Shed in saliva, nasal secretions, urine, semen, mammary secretion, feces • Virus persists in tonsils • Transmission: direct contact, aerosolized respiratory secretions, AI with virus-positive semen |
|
|
• Host range: Mice, Mus musculus
• Transmission: via saliva, contact, biting • Disease: – Inapparent, persistent infections – Elevation of LDH levels – Influences immune response of mice to other agents – Target cell = macrophage |
Lactate dehydrogenaseelevating
virus (LDHV) |
|
|
• Host species: macaques, Macaca
fascicularis • Disease: acute, febrile hemorrhagic fever • Transmission: aerosolized respiratory secretions, direct contact • Mortality rate: approaches 100% in macaques • Reservoir hosts: African green monkeys, baboons |
Simian hemorrhagic fever
|

