![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
32 Cards in this Set
- Front
- Back
|
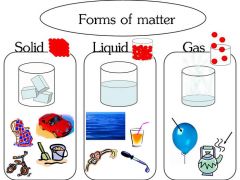
Solid |
has a definite shape and definite volume this fixed, closelty packed arrangement of particles in a solid causes it to have a definite shape and volume |
|
|
crystalline solid |
solids made up of crystals (Examples: Salt, Sugar and Snow) |
|
|
amorphous solid |
particles are not arranged in a regular pattern |
|
|
Liquid |
has a definite volume but no shape of its own Because its particles are free to move, a liquid has no definite shape. However , it does have a definite volume |
|
|
fluid |
a substance that flows |
|
|
surface tension |
the inward force, or pull, among molecules in a liquid that bring the molecues on the surface closer together. |
|
|
Viscosity |
liquid's resistance to flowing |
|
|
Gas |
has neither a definite shape or volume. As gas particles move, they spread apart, filling all the space available. Thus, a gas has neither definite shape nor definite volume. |
|
|
volume |
the amount of space that matter fills |
|
|
Pressure |
pressure of a gas is the force of its outward push divided by the area of the walls of the contianer |
|
|
Pressure Formula |
Pressure = Force divided by Area |
|
|
melting |
the change from a solid state to a liquid state |
|
|
melting point |
the specific temperature when melting occurs (in crystalline solids) |
|
|
melting point |
at a solids melting point, its particles vibrate so fast that they break free from their fixed positions |
|
|
freezing |
the change of state from a liquid to a solid |
|
|
freezing point |
at a liquid's freezing point, its particles are moving so slowly that they begin to take on fixed positions |
|
|
Vaporization |
the change of state from a liquid to a gas, |
|
|
Vaporization |
vaporization occurs when the particles in a liquid gain enough energy to move independently |
|
|
evaporation |
vaporization that takes place only in the surface if a liquid |
|
|
boiling |
vaporization that takes place both below and at the surface |
|
|
boiling point |
the temperature at which a liquid boils |
|
|
condensation |
the change in state from a gas to a liquid |
|
|
condensations |
condensation occurs when particles in a gas lose enough thermal energy ti firm a liquid |
|
|
sublimation |
the change in state from solid to gas |
|
|
Sublimation |
During sublimation, particles of a solid do not pass through the liquid state as they form a gas |
|
|
Pressure and Temperature |
When the temperature of a gas at a constant volume is increased, the pressure of the gas increases. When the temperature is decreased, the pressure of the gas decreases |
|
|
Charles's Law |
When the temperature of a gas at a constant pressure is increased, its volume increases. When the temperature of a gas at a constant temperature is decreased, its volume decreases. |
|
|
Charles Law was created by: |
French scientist Jacques Charles |
|
|
directly proportional |
when the graph of two variables is a straight line passing through the origin. |
|
|
Boyle's Law |
When the pressure of a gas at a constant temperature is increased, the volume of the gas decreases. When the pressure is decreased, the volume increases. |
|
|
inversely proportional |
When the product of two variables is constant |
|
|
SOLIDS LIQUID GASES |
|

