![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
30 Cards in this Set
- Front
- Back
|
Taking a Goody's™ aspirin powder works faster than an aspirin tablet. This the concept of increasing the reaction rate by....
|
increasing surface area
|
|
|
Increasing surface area increases reaction rate because it...
|
increases the number of places where collisions can occur
|
|
|
Increasing the pressure of a gas (by decreasing the volume) ____the reaction rate
|
increases
|
|
|
Increasing the pressure of a gas (by decreasing the volume) increases the reaction because it makes the particles ___ together.
|
closer
|
|
|
Making particles closer together (because of reduced gas volume) can increase the chance that ____ will occur.
|
collisions
|
|
|
Increasing pressure of a gas (by decreasing gas volume) has the same effect at increasing the ___ of the substance.
|
concentration
|
|
|
A piece of steel wool burns slowly in air (20% O₂) and brilliantly, quickly burns in 100% O₂. This method of increasing reaction rate is the concept of increasing....
|
concentration of reactants.
|
|
|
Increasing concentration of reactants speeds reaction because....
|
particles are closer together,
collisions are more likely to occur, there are more reaction sites |
|
|
Increasing temperature speeds reaction by ...
|
speeding up motion of the particles, making them collide more frequently and with more force.
|
|
|
Reaction speed doubles when it warms by 10ºC.This is the concept of speed increasing as ....
|
temperature increases.
|
|
|
A slow reaction is sped-up by addition of a chemical. This the concept of increasing reaction rate by use of a ....
|
catalyst
|
|
|
A ____ is a chemical that speeds up a reaction but itself is not consumed in the reaction.
|
catalyst
|
|
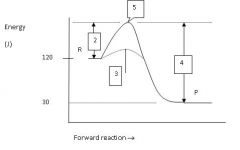
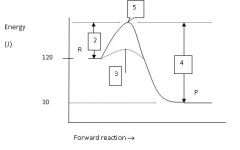
What is the energy level of the products?
|
30 J
|
|
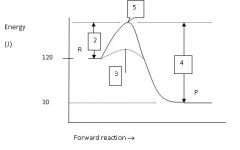
Which numbered region indicates the activation energy for the uncatalyzed reaction?
|
2
|
|
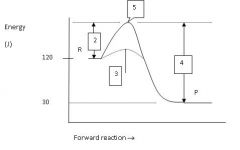
Which numbered region indicates the energy level of the activated complex?
|
5
|
|
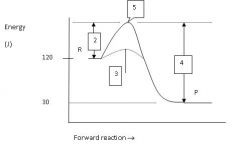
Which numbered region indicates the activation energy needed for the catalyzed reaction?
|
3
|
|
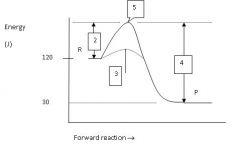
Calculate the enthalpy for this reaction.
|
ΔH = P - R = 30 - 120 = -90 J
|
|
Is this reaction endothermic or exothermic?
|
exothermic
|
|
Is this reaction exothermic or endothermic?
|
endothermic
|
|
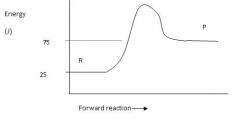
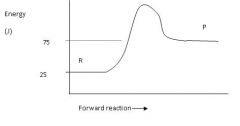
Calculate the enthalpy for this reaction.
|
ΔH = P - R = 75-25 = 50 J
|
|
|
Suppose ΔH = +130 J. Is it exothermic or endothermic?
|
endothermic
|
|
|
What effect does adding a catalyst have?
|
It reduces the activation energy?
|
|
|
What is the name of ΔH ?
|
Change in enthalpy
|
|
|
What does enthalpy mean?
|
energy of reaction
|
|
|
If reaction energy is higher than product energy then the reaction is considered to be...
|
exothermic
|
|
|
Suppose ΔH = +130 J. Is it exothermic or endothermic?
|
endothermic
|
|
|
What effect does adding a catalyst have?
|
It reduces the activation energy?
|
|
|
What is the name of ΔH ?
|
Change in enthalpy
|
|
|
What does enthalpy mean?
|
energy of reaction
|
|
|
If reaction energy is higher than product energy then the reaction is considered to be...
|
exothermic
|

