![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
75 Cards in this Set
- Front
- Back
|
In order to interpret an incoming signal, sensory receptor must be able to encode what information about the stimulus into APs: |
MILD |
|
|
Theory of labeled lines? |
informs on modality and location • Each receptor cell connects to a specific sensory area of the cortex through one afferent neuron pathway • Cortex interprets any signal coming from that receptor cell as having been caused by a particular type of stimulus in that particular location |
|
|
Sensory units: |
multiple sensory receptors that form synapses w/ a single afferent neuron.
In general, all of the sensory receptors associated w/ a single afferent neuron are of the same type, thus the theory of labeled- line perception can account for our ability to distinguish among different stimulus modalities. |
|
|
The fundamental assumption of the labeled-line theory |
THERE IS A DISCRETE PATHWAY FROM A SENSORY CELL TO THE INTEGRATING CENTER. |
|
|
A receptor sensitive to more than one sensory modality likely encodes info ... |
in the temporal pattern of its APs, so bursts of APs could convey a diff message than a continuous series |
|
|
How would relative firing patterns of several ADJACENT sensory cells carry info regarding stimulus modality? |
If first receptor very sensitive to stimulus A but less sensitive to stimulus B, while another receptor has the opp pattern, by comparing the relative intensity of the signal coming from the 2 receptors, an afferent neuron could code info regarding stimulus modality. |
|
|
receptive field: |
corresponds to the region of the skin that causes a response in that particular afferent neuron.
"the region in sensory space (e.g., the body surface, or a specialized structure such as the retina) within which a specific stimulus elicits the greatest action potential response" |
|
|
How does variation in size of a receptive field change affect detection of stimuli? |
- Large fields detect stimuli across larger area, - small receptive fields provide MORE PRECISE localization of the stimulus = greater acuity. |
|
|
Info from a single afferent neuron only signals whether stimulus occurred w/i receptive field, how do animals make this more precise? |
having afferent neurons w/ OVERLAPPING RECEPTIVE FIELDS. Stimulus causing neuron A and B to respond must have happened where their fields overlap |
|
|
Population coding: |
where information about the stimulus is encoded in the pattern of firing of multiple neurons. |
|
|
Lateral inhibition: |
Neurons on either side of highly stimulated neuron don't fire, they are inhibited. This increases the contrast b/w the signals from neurons at the center of the stimulus + neurons on the edge, allowing finer discrimination |
|
|
Lateral inhibition example: |
Weak touch across receptive fields of neurons A,B,C would cause each neuron to release small amount of NT onto its 2nd-order neuron, so all are stimulated. - a Strong prick in receptive field of neuron B releases lots of NT onto its 2nd order neuron B2. The skin bends slightly due to prick, weakly stimulating fields for A+C. Lateral interneurons that form synapses b/w the axon terminals of B and A+C receive the strong NT response B. These interneurons release an inhibitory NT that prevents release of NT from A+C, no response from them. |
|
|
Diagram of receptor location by lateral inhibition |
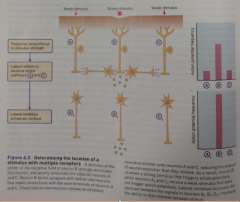
|
|
|
How do APs code INTENSITY? |
thru changes in FREQUENCY: strong stimuli trigger high-frequency APs |
|
|
dynamic range: |
relatively limited range of intensities that sensory receptor cells can encode stimuli over |
|
|
threshold of detection: |
weakest stimulus that produces a response in a receptor 50% of the time |
|
|
At the top of the dynamic range, the receptor cell is: |
is saturated + can't increase its response even if the signal strength increases.
if all available ion channels have opened or closed. The receptor will also reach the top of its dynamic range if the membrane potential reaches the equilibrium potential for the particular ion involved in the receptor or generator potential (because no net ion movement will occur beyond this point). The max rate of release of NT from the receptor cell, or the maximum frequency of APs in the afferent neuron can also set the top of the dynamic range. |
|
|
How can a receptor reach the top of its dynamic range? |
- if all of the available receptor proteins become saturated. - if all available ion channels have opened or closed. - if the membrane potential reaches the equilibrium potential for the particular ion involved in the receptor or generator potential (because no net ion movement will occur beyond this point). |
|
|
What can set the top of the dynamic range? |
- Any of the steps in sensory transduction:
- The max rate of release of NT from the receptor cell - the maximum frequency of APs in the afferent neuron |
|
|
Dynamic range graph |
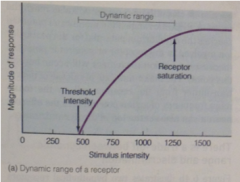
Response of receptor increases with increasing intensity |
|
|
Different dynamic ranges graph: |
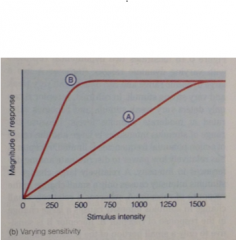
Rcpt A - large dynamic range + can detect both very weak + very strong stimuli. |
|
|
With a large range of stimulus intensities + the range of AP frequencies is limited, receptor A has ___ power for discriminating differences in intensity |
relatively low power to discriminate differences in intensity. A relatively large change in stimulus intensity causes only small change in response of receptor A (see slope), while a small change in intensity causes large change in response of receptor B. |
|
|
With a small range of intensities, B has ___ power for discriminating intensities |
Higher, B is sensitive to only a small portion of the possible range of stimulus intensities, but it has the ability to provide very fine discrimination w/i that range (as a small change in intensity causes large change in response) |
|
|
Range fractionation strategy: |
Use behaviours of populations of receptors to improve sensory discrimination: groups of receptors, each sensitive to a different range of stimulus intensities, can work together to provide fine discrimination across a wider range of intensities |
|
|
individual receptor cells are sensitive to only a small portion of possible range of intensities, so how does range fractionation work? |
Multiple receptors cover different parts of the range.
This system does intensity thru behaviour of populations of sensory receptors. |
|
|
Range fractionation graph: |
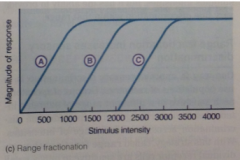
rcptrs work together to provide fine discrim across wide range of intensities |
|
|
upper limit of the frequency of APs is set by |
by refractory periods of the v-gated channels involved in the AP. |
|
|
How can a sensory receptor code for such a wide range of stimulus intensities (1-1.4 million) with such a small range of AP frequencies (1/sec to 1000/sec)? |
- Range fractionation can extend the dynamic range - BUT LOG ENCODING!! many receptors use this strategy |
|
|
Log encoding: |
|
|
|
Log encoding graph: |
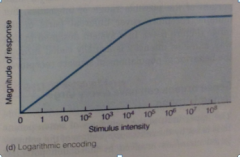
fine discrimination at low intensities + coarser discrimination at high intensities (limited change in response when large change in intensity) |
|
|
Weber-Fenchner relationship |
= log relationship b/w actual and perceived stimulus intensity Sensations that obey the Weber-Fenchner relationship: brightness, loudness, and weight. Day/night candle, book on sofa u carry |
|
|
Two functional classes of sensory receptors that code for stimulus DURATION: |
|
|
|
|
fire APs as long as stimulus continues, so can convey info about HOW LONG stimulus lasts. Most do not fire APs at same frequency throughout prolonged stimulus |
|
|
tonic receptor graph |
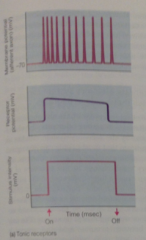
are depolarized throughout the duration of a stimulus, can show adaptation |
|
|
receptor adaptation |
Typically AP frequency often declines if stimulus intensity is maintained at a constant level |
|
|
phasic receptor |
Some receptors that adapt so quickly that they produce APs only when stimulus begins
-code changes in the stimulus but do not explicitly encode stimulus duration |
|
|
phasic receptor graph |
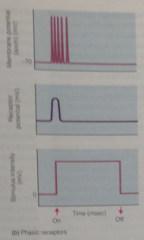
depolarize only at beginning, adapt rapidly |
|
|
|
= sense of smell, the detection of chemicals carried in the air. provides the ability to sense chemicals whose source is locate away some distance |
|
|
gustation |
= sense of taste, detect dissolved chemicals emitted from ingested food |
|
|
How are olfaction + gustation distinct from one another? |
based on structural criteria; - are performed by diff sense organs, - use diff Signal transduction mechanisms - separate integrating centers process the incoming info from the senses of taste + smell |
|
|
Odorant receptor proteins: |
GPCRs. Each odorant receptor cell expresses only a single kind of odorant receptor protein. |
|
|
Odorant pathway: |
Receptor change signals G(olf) to activate, which signals via adenylate cyclase, activating cAMP pathway that eventually -> depolarizing generator potential. |
|
|
bipolar olfactory neuron image: |
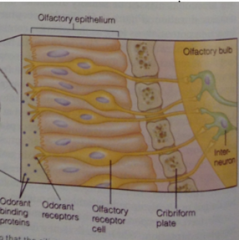
|
|
|
odorant pathway diagram: |
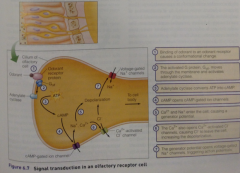
-cAMP opens cAMP-gated ion channels, allowing Ca2+/Na+ to enter (generator potential) |
|
|
Some odorant receptors are coupled to PLC cascade: |
PLC hydrolyzes PIP2 in the PM, producing IP3 and DAG, resulting in increase in intracellular Ca2+ just like in cAMP pathway
-This causes PM Cl- channels to open, ultimately depolarizing cell even more, triggering APs. |
|
|
How do you account for the fact that the total # of odors that an animal can distinguish is even larger than the amount of genes coding for odorant receptor proteins? |
1 olfactory neuron -> 1 odorant receptor gene, but each odorant receptor can recognize more than 1 odorant. Thus a given odorant excites multiple olfactory neurons. combinatorial code: Even if each odorant were coded by a combo of only 3 diff receptors, there would be approximately 1 bill potential combos. The code for each odor actually involves more than 3 receptors! |
|
|
Unlike olfactory, the gustatory system not able to discriminate among 1000s: grouped into 5 classes: |
|
|
|
What do the different tastes indicate? |
Sweet, umami, and salty tastes indicate nutritionally important carbs, proteins, and ions,
bitter + sour tastes indicate toxic substances |
|
|
In terrestrial vertebrates, Taste receptor cells found on: |
|
|
|
Taste buds: |
onion-shaped structures that contain multiple taste receptor cells (b/w 50 and 100), with a pore that opens out to surface of the body. |
|
|
Taste bud diagram: |
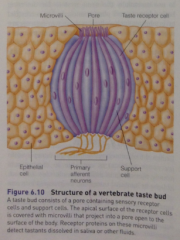
apical surface of the taste cell is folded into numerous microvilli, which contain the receptors + ion channels that mediate the transduction of the taste signal. |
|
|
tastants, |
= Dissolved chemicals from food,
enter thru this pore + contact taste receptor cell. |
|
|
What chemicals are flavours conveyed by: |
• SALTY: conveyed by Na+ ions in food |
|
|
Salty pathway: |
Salty substance receptor is actually not receptor but an Na+ ion channel, so it enters cell and so depolarizes it, triggering v-gated Ca2+ to release NTs
|
|
|
Salty pathway diagram: |
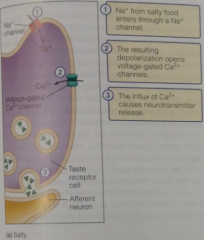
|
|
|
The Na+ channels involved in "salty" are also permeable to H+ ions, what does this indicate? |
may play role in perception of sour. Since Na+ and H+ compete for the channel, these channels are probably important for sourness perception only in species with relatively low Na+ levels in their saliva, use these channels to detect sourness. we have high saliva Na+ so can taste sourness thru other mechanisms. |
|
|
Example of a sour transduction mechanism:
(there are many) |
Apically localized K+ channel is blocked directly by H+ protons. Blocking these K+ channels leads to depolarization of the taste cells by decreasing K+ permeability and altering the resting membrane potential (Goldman eq). This depolarization opens v-gated Ca2+, causes NT release. |
|
|
Sour pathway diagram |
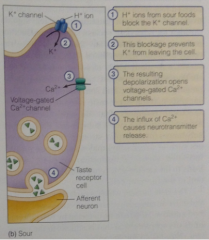
|
|
|
Sweet signal: |
Sugars bind to GPCRs at the apical cell surface, activating G protein gustducin, which signals thru an adenylate cyclase signal transduction pathway (with cAMP, protein kinase that closes K+ channels, depolarizing for v-gated Ca2+, releasing NT). |
|
|
Another pathway for sweet (artificial) |
IP3 instead of adenylate cyclase, but same closing of K+ channels, depolarizing etc |
|
|
Sweet pathway diagram |
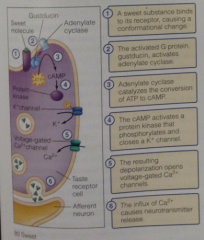
|
|
|
Sweetness receptors are sensitive to: |
Sweetness receptors are sensitive to: |
|
|
Umami detected by 2 different kinds of receptors: |
• 1 similar to sweet receptors -- Umami caused by L-glutamate and other AAs found in foods, as well as the food additive MSG, |
|
|
Umami pathway with glutamate receptors: |
When glutamate binds to this modified glutamate receptor, conformation changes, activating an associated G protein, which then activates a phosphodiesterase that degrades cAMP into AMP. ->The decreases in cAMP thought to trigger NT release, though precise pathways involved have not yet been ID'd. |
|
|
How is Bitter very complex? |
more specific than sweet, we have at least 25 genes coding for bitter-taste receptors, and each taste cell that is sensitive to "bitterness" expresses many of these genes. The way this complex pattern of expression is translated into perception of bitterness is unknown but we know the signal transduction mechanisms |
|
|
Bitter pathway: |
Activated G protein transducin activates PLC-> split into IP3 -> causes release of Ca2+ from intracellular stores-> NT release |
|
|
Bitter diagram: |
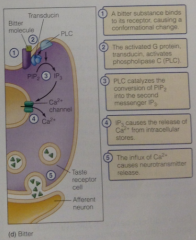
|
|
|
What has to happen for you to be able to take in sensory information from your surroundings? |
1. Sensory receptor detects signal 2. Transduce signals into a form that can be interpreted 3. Amplifies signals (maybe) 4. Signal sent to an integrating centre (e.g. brain) |
|
|
adequate stimulus: |
Most receptors are most sensitive to one specific type of stimulus |
|
|
polymodal receptors |
Certain receptors respond to several different stimuli with similar sensitivity |
|
|
example of a polymodal receptor in humans? |
eg: TRP channels
• TRPV1: heat, acidic pH, and capsaicin • TRPM8: cold and menthol |
|
|
Olfactory receptor neurons located in: |
olfactory epithelium. |
|
|
Olfactory bulb: |
where projection neurons are located. |
|
|
• Olfactory tract: |
projects to olfactory cortex via limbic |
|
|
Taste travels pathway: |
Taste buds -> cranial nerves -> brain stem - > thalamus and limbic system |

