![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
25 Cards in this Set
- Front
- Back
|
How many valence electrons are in group 3A?
|
three
|
|
|
What is the typical oxidation of most members of group 3A?
|
+3 (positive 3)
|
|
|
Another name for "outer level" electrons is ____ electrons.
|
Valence
|
|
|
Which 3 of the "A" families are mostly metals?
|
I A, II A, & III A
|
|
|
Are all the "B" elements metals?
|
Yes
|
|
|
What extra type of electrons do the B elements have?
|
sublevel d electrons
|
|
|
How many valence electrons are in group 4A?
|
four
|
|
|
How many valence electrons are in group 5A?
|
five
|
|
|
What oxidation number is typical of the nonmetallic members of group 5A?
|
-3 (negative 3)
|
|
|
How many valence electrons are in members of group 6A?
|
six
|
|
|
What oxidation number is typical of members of group 6A?
|
-2 (negative 2)
|
|
|
How many valence electrons are in members of group 7A?
|
seven
|
|
|
What oxidation number is typical of members of group 7A
|
-1 (negative 1)
|
|
|
How many valence electrons do the Noble Gases have?
|
eight
|
|
|
What is the oxidation number of the Noble Gases?
|
0
|
|
|
In which groups can nonmetals be found?
|
V A, VI A, VII A, VIII A
|
|
|
Metalloids have properties of both ______ and ______.
|
Metals, nonmetals
|
|
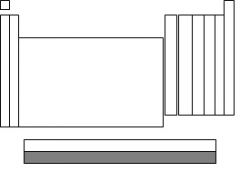
The highlighted section is called....
|
actinides
|
|
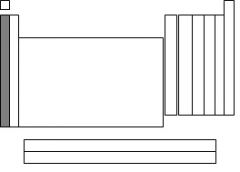
The highlighted section is called....
|
alkali metals
|
|
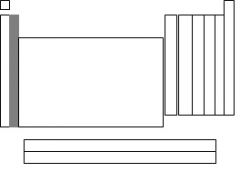
The highlighted section is called....
|
alkaline earth metals
|
|
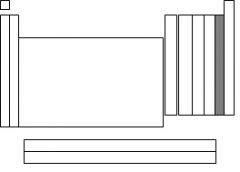
The highlighted section is called....
|
halogens
|
|
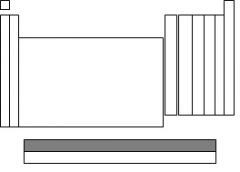
The highlighted section is called....
|
lanthanides
|
|
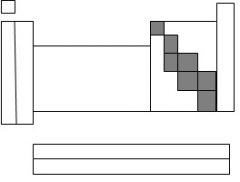
The highlighted section is called....
|
metalloids
|
|
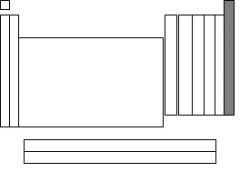
The highlighted section is called....
|
noble gases
|
|
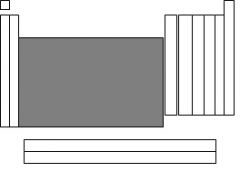
The highlighted section is called....
|
transition metals
|

