![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
45 Cards in this Set
- Front
- Back
|
Mechanism of tyrosine hydroxylase
|
hydroxylates tyrosine @ 3'
|
|
|
Mechanism of dopamine B-hydroxylase
|
put OH on carbon1 in B configuration
|
|
|
After NE binds to alpha/beta adrenergic receptor what happens
|
G-protein produces biochemical cascade
|
|
|
4 fates of NE once it is in the synapse
|
1)bind to alpha2 receptor
2)bind to alpha/beta receptor on effector cell 3)reuptake 4)diffusion/metabolism |
|
|
NE
|
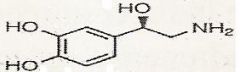
What is this?
|
|
|
tyrosine
|
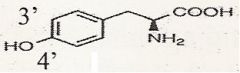
What is this?
|
|
|
L-dopa
|
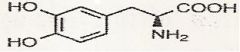
What is this?
|
|
|
dopamine
|
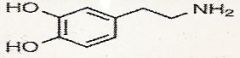
What is this?
|
|
|
Adrenergic Drug types (SAR's of the drugs) (4)
|
1)agonist/antagonists (act directly on receptor)
2)affect storage and release from vesicles 3)affect NE biosynthesis (enzyme inhibitors) 4)affect reuptake and catabolism (enzyme inhibitors) |
|
|
Bigger groups @ R1 = higher affinity at Beta receptors, the biggest groups give affinity to Beta2 receptors
|
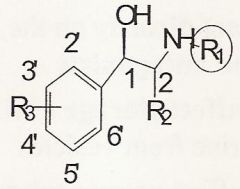
As the substituent at R1 increases in size what happens?
|
|
|
Beta receptor affinity is better than alpha receptor affinity
B/c it creates a chiral carbon that creates diastereomers w/ different activities |
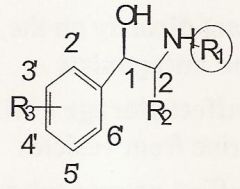
As the groups at R2 increase in size what happens? AND why is it bad to mess with R2?
|
|
|
If you do modify @ the R2 position which isomer has more activity and what is the name/activity of this compound?
|
1R,2S or 1S,2R (erythro)
ephedrine, partial agonist and indirect agonist for NE |
|
|
an OH in R configuration
|
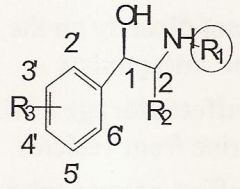
Maximal direct agonist activity is found when C-1 has what?
|
|
|
3'-5' dihydroxy b/c it is no longer a catecholamine and therefore resistant to COMT/first pass metabolism
|
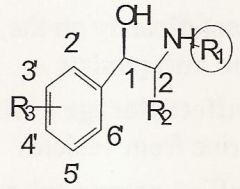
Which is better and why?
A 3'-4' dihydroxy or a 3'-5' dihydroxy? |
|
|
beta-2 receptors
|
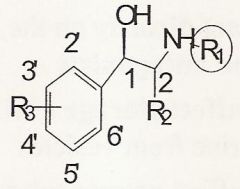
3'-5' dihydroxy is selective for...
|
|
|
just have one OH, at 3'-OH
|
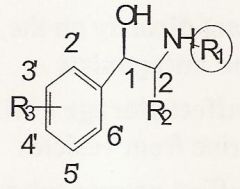
In order for an agonist to have just alpha receptor activity it must...
|
|
|
ONE OH at 4'
|
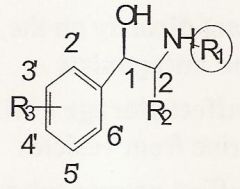
In order for an agonist to have just beta-receptor activity what must the substitution be @ R3
|
|
|
Have NO OH, a 2' OH or a 5' OH
|
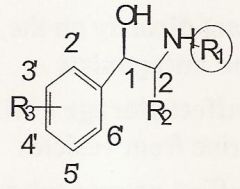
How do you get mixed activity for an agonist if you do an R3 substitution?
|
|
|
3 direct acting phenylethanolamine agonists
|
1)NE
2)E 3)phenylephrine |
|
|
4 indirect acting sympathomimetics (for phenylethanolamine)
|
1)ephedrine
2)pseudoephedrine 3)phenylpropanolamine 4)amphetamine |
|
|
What type of receiever accepts imitations more easily alpha or beta recievers?
|
BETA
|
|
|
Imidazoline agonists
a)for alpha1 b)for alpha2 |
a)naphazoline, tetrahydrozline
b)clonidine |
|
|
Alpha Adrenergic antagonists (3)
|
1)phenoxybenzamine
2)tolazoline (imidazoline drug) 3)phentolamine (imidazoline drug) |
|
|
SELECTIVE alpha1 antagonists
|
quinazoline derivatives
|
|
|
Quinazoline derivatives (characteristics) (3)
|
1)quinazoline ring system
2)piperazine ring system 3)rest of structure imparts solubility/half-life |
|
|
Naphazoline
|
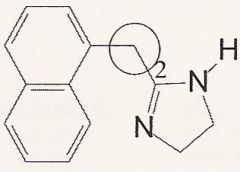
What is it?
|
|
|
Clonidine
|
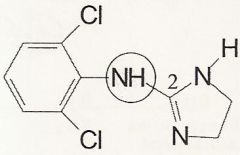
What is it?
|
|
|
Tolazoline
|
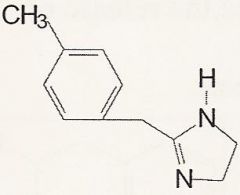
What is it?
|
|
|
Phentolamine
|
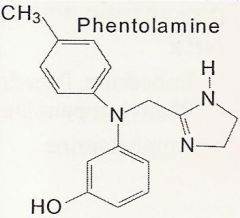
What is it?
|
|
|
Beta antagonists
a)1st one b)others |
a)aryloxypropanolamines (propanolol)
b)4-substituted phenyloxypropanolamines (practolol, metoprolol) |
|
|
Metoprolol is selective for ___ and why?
|
Beta1, b/c has group in para position
|
|
|
How do propanolamines act as beta antagonists?
|
have OCH2 added to ring to incr length of side chain making it so that it can NOT bind to Asp in AS
|
|
|
Quinazoline derivative, the piperazine ring (ring in middle w/ no double bonds and N's on its ends)
|
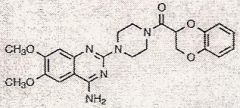
What category is this drug in and what is it's most special feature
|
|
|
3 quinazoline derivative drugs
|
1)prazosin
2)terazosin 3)doxazosin |
|
|
propanololamines
|
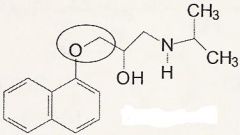
What category of drug is this in?
|
|
|
2 enzymes in the NE synthesis pathway that are not targeted by drugs to inhibit?
|
1)dopamine B-hydroxylase
2)phenylethanolamine N-methyltransferase |
|
|
Drugs affecting NE/E biosynthesis do what?
|
1)inhibit tyrosine hydroxylase
2)inhibit aromatic L-AA decarboxylase |
|
|
Drug that inhibits tyrosine hydroxylase and its use
|
Metyrosine, inhibits catecholamine production when tumors in adrenal medulla
|
|
|
Drug that inhibits aromatic L-AA decarboxylase and its use
|
Carbidopa, keeps L-dopa around to decrease parkinson's symptoms
|
|
|
Drugs affecting storage vesicles (3)
|
1)reserpine
2)bretylium 3)guanethidine |
|
|
Major structural classes in adrenergic agents and their overall fxn of 3 of em
|
1)Aryloxypropanolamines (beta-blockers)
2)guianidine derivatives 3)imidazoline derivatives (alpha agonists) 4)phenylethanolamine derivatives 5)phenylethylamine derivatives 6)quinazoline derivatives (alpha1 antagonists) |
|
|
If the 4' on a aryloxypropanolamines is substituted what does it do?
|
makes it selective for Beta1
|
|
|
If you see "oxy" in a structures name what category is it most likely?
|
BETA BLOCKER
|
|
|
Where is sympathetic tone dominant? (4) and effect of ganglionic blockade
|
1)arterioles (vasodilation)
2)veins (dilation, pooling, decr cardiac output) 3)sweat glands (anhidrosis) 4)genital tract (impotence) |
|
|
Where is parasympathetic tone dominant? (7) and effect of ganglionic blockade
|
1)heart (tachycardia)
2)iris (mydriasis) 3)ciliary muscle (cycloplegia) 4)GI (reduced tone and motility) 5)urinary bladder (urinary retention) 6)salivary glands (xerostomia) 7)genital tract (impotence) |

