![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
170 Cards in this Set
- Front
- Back
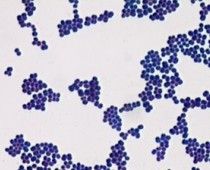
Who am I and what is my technique?
|
Staphylococcus aureus
Gram Positive Stain |
|
|
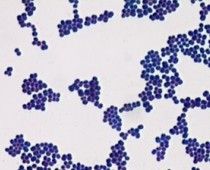
Who am I and how do you differentiate me from my close family member?
|
Staphylococcus aureus
Catalase + --> growth in clusters on gs |
|
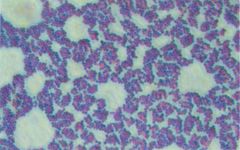
Who am I and how do you differentiate me from my family member?
|
Staphylococcus aureus
catalase + --> growth in clusters |
|
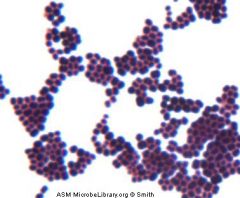
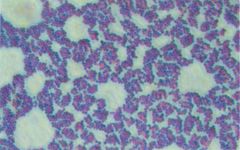
Who am I and how do you differentiate me from my family member?
|
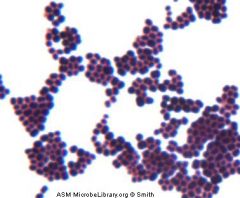
Staphylococcus aureus
Catalase + --> Coagulase + --> growth in clusters |
|
|
Staphylococcus aureus how do you differentiate me from epidermidis?
|
Staphylococcus aureus forms a fairly large yellow colony on rich medium; S. epidermidis has a relatively small white colony. S. aureus is often hemolytic on blood agar; S. epidermidis is non hemolytic. Staphylococci are facultative anaerobes that grow by aerobic respiration or by fermentation that yields principally lactic acid. The bacteria are catalase-positive and oxidase-negative. S. aureus can grow at a temperature range of 15 to 45 degrees and at NaCl concentrations as high as 15 percent. Nearly all strains of S. aureus produce the enzyme coagulase: nearly all strains of S. epidermidis lack this enzyme. S. aureus should always be considered a potential pathogen; most strains of S. epidermidis are nonpathogenic and may even play a protective role in humans as normal flora. Staphylococcus epidermidis may be a pathogen in the hospital environment.
|
|
|
Important phenotypic characteristics of Staphylococcus aureus
|
Gram-positive, cluster-forming coccus
nonmotile, nonsporeforming facultative anaerobe fermentation of glucose produces mainly lactic acid ferments mannitol (distinguishes from S. epidermidis) catalase positive coagulase positive golden yellow colony on agar normal flora of humans found on nasal passages, skin and mucous membranes pathogen of humans, causes a wide range of suppurative infections, as well as food poisoning and toxic shock syndrome |
|
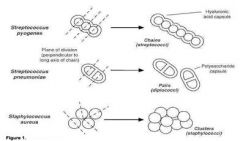
How to differentiate staph from strep
|
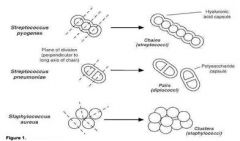
Both staphylococci and streptococci have round, spherical cell shape, but the arrangement of cells is different due to a different binary fission. Streptococci form a chain of round cells, because their division occurs in one linear direction, whereas staphylococci divide in various directions forming grape-like clusters.
|
|
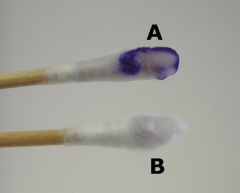
Oxidase test - which organisms need this test and what are the results?
|
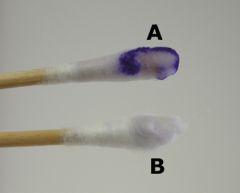
The oxidase test is mainly used to differentiate:
1) Oxidase positive Neisseria spp. from other gram-negative diplococci. 2) Oxidase positive Aeromonas hydrophila from Escherichia coli (gram-negative). 3) Oxidase positive Plesiomonas shigelloids from Shigella sonnei (gram-negative). |
|
|
What are the differences between gp and gn cell walls?
How does this effect staining? |
gp have a cell wall with a thick peptidoglycan layer comprising a majority of the wall.
gp stain violet (or purple for gp) gn havea thin peptidoglycan layer that comprises a small portion of the cell wall. gn have an outer lipopolysaccharide and protein layer - gp does not gn are more porous and stain pink-red |
|
|
what is the gram stain technique?
|
Gram stain procedure is as follows:
1.Place a slide with a bacterial smear on a staining rack. 2.primary stain: STAIN the slide with GRAM CRYSTAL VIOLET for 1 min. 3.Pour off the stain with COLD WATER. Note: fingers stain Gram-positive - use forceps! 4.Flood slide with a mordent: GRAM IODINE for 1 min. 5.Pour off the iodine with COLD WATER. 6.Decolourize by washing the slide briefly with GRAM DECOLORIZER until runs clear . 7.Wash slide thoroughly with COLD water 8.Flood slide with GRAM SAFRANIN counterstain for 30-60sec 9.Wash with COLD water. 10.Blot excess water and air dry. 11. use an oil immersioin lens. |
|
|
who created the gram stain?
|
christian gram 1884 - most commonly used bacterial stain
|
|
|
Gram stain helps differentiate bacteria into 2 groups based on wall structure - describe differences
|
GP (purple) thick peptidoglycan layer - majority of cell wall
GN - think peptidoglycan layer - small portion of cell wall, more porous (red/pink) |
|
Which one is GP and GN - describe
|
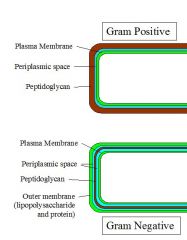
GRAM POSITIVE cell wall has thick peptidoglycan layer and teichoic acids, which are not found in gram negative cells.
GRAM NEGATIVE cell wall --thin peptidoglycan layer outside cytoplasmic membrane, containing a network of NAM and NAG with NAM's crosslinking the chains. NAM = n-acetylglutamic acid, and NAG = n-acetyle muramic acid. Enzyme known as transpeptidase does the cross linking and penicillin interferes with it so cell bursts. --There is a pleriplasmic space only in gram negative. --Gram-negative bacteria are those bacteria that do not retain crystal violet dye in the Gram staining protocol. |
|
|
Acid fast stains are used for what?
|
gram indeterminante/gram resistant bacteria
Mycobactera and Coccidian protozoa |
|
|
Why Mycobactera and Coccidian protozoa wont stain?
|
lipid rich cell wall the gs cannot penetrate
|
|
|
Why are Mycobactera classified as myco?
|
not for a fungus - but bc of the abundance of mycolic acid in the cell wall
|
|
|
Describe the bactera that don't stain with gs.
|
These Radicals May Microscopicaly Lack Color
Treponema - too thin - use dark field micro Rickettsia - small obligate intracelluar pathogens Mycobacteria - too small Legionella - intracellular, use silver stain Chlamydia - intracellular, thin cell lack peptioglycan |
|
|
2 primary shapes of bacteria
|
cocci and bacilli
others- vibrios - curved gn rods spircochetes gn helical coils |
|
|
diplo =
|
pairs
|
|
|
strep =
|
chains
|
|
|
staph =
|
clusters
|
|
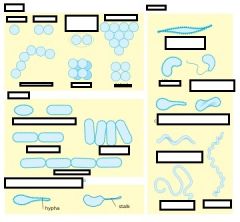
Name me
|
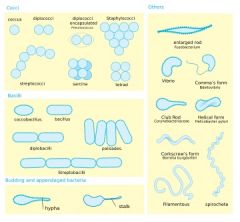
cocci- spheres
bacilli- rods others - vibrios, spirochetes diplo - pair strep-chain staph-cluster |
|
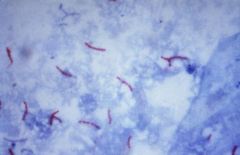
who am I and what stain?
|
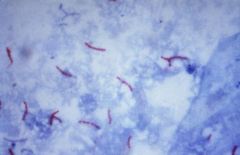
Mycobacteria tuberculosis
acid fast bacili |
|
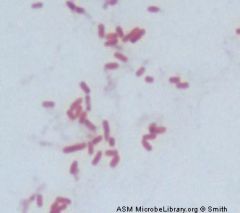
name me and stain
|
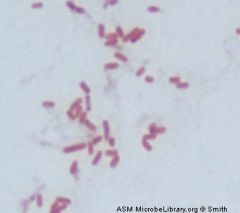
Gram negative rod
Escherichia coli |
|
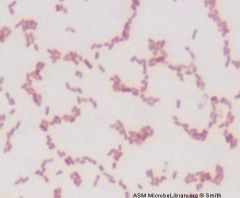
name me
|
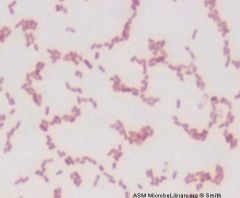
Gram Negative rod
Klebsiella pneumoniae |
|
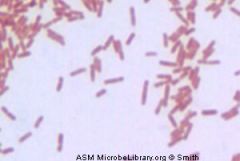
name me
|
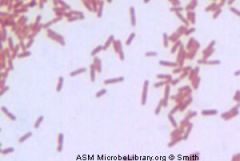
Gram Negative Rod
Pseudomona.aeruginosa |
|
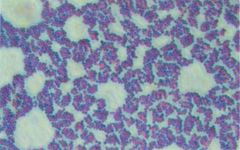
name me
|
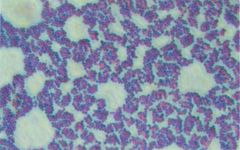
Gram Positive Cluster
Staphylococcus aureus |
|
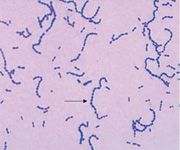
name me
|
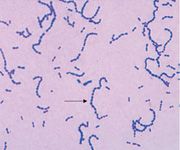
Gram-Positive Cocci chains > Streptococcus pyogenes
|
|
|
gram stain technique
What is mordent? |
iodine is added and forms a large complex with the crystal violet - called mordent.
|
|
|
Which cells can you get the mordent out and how?
|
the mordent washes out of the porous gram neg cells from the decolorizer
the thick gp cell walls hold the mordent hence purple gram neg need counterstain - red |
|
|
acid fast stain technique
|
The Kinyoun stain is a method of staining acid-fast microorganisms, specifically mycobacterium. The procedure for Kinyoun staining is similar to the Ziehl-Neelsen stain, but does not involve heating the slides being stained.1 The Kinyoun staining method uses carbolfuchsin as a primary stain, followed by decolorization with an acid-alcohol solution and methylene blue as a counterstain. Kinyoun carbolfuschsin has a greater concentration of phenol and basic fuchsin and does not require heating in order to stain properly.1 When viewed under a microscope, a Kinyoun stained slide will show acid-fast organisms as red and nonacid-fast organisms as blue.
Procedure 1.Flood slides with Kinyoun TB CARBOLFUCHSIN (KF) for 3-4 minutes. 2.Rinse gently with water until the water flows off clear. 3.Decolorize the slide by flooding with TB DECOLORIZER until runs clear. 4.Rinse gently with water until the water flows off clear. 5.Flood slides with TB BRILLIANT GREEN (or TB methylene blue) for 30 seconds. 6.Rinse gently with water until the water flows off clear. 7.Allow slides to air dry before viewing. |
|
|
acid fast stain examination
|
acid fast - dark red to pink over a green backgroun of non-acid fast substances
RED- (+) acid fast - coccidian parasites: Cryptosporidium, Clyclospora, Isospora, Mycobacteria Blue or Green: (-) gram pos or gn organisms |
|
|
How do you report acid fast?
|
Acid fast bacilli positive or AFB(+).
only one organism needed exact species cannot be determined by an acid fast stain alone acid fast stool can be identified based on staining and morph. |
|
|
Describe Basic culture media
|
simple preps - support growth of microorgs with no speical growth requirements - Tryptocase Soy agar
|
|
|
Describe Enriched culture media
|
preps that include additional nuturents beyond basic preps. Common: whole blood agar, lysed blood (choc agar), peptone (alkaline-peptone water), NAD'
|
|
|
Describe Selective culture media
|
have substances that INHIBIT growth of undesired orgs. bile salts (MacConkey), dyes, antibiotics.
also helps limit the growth of natural flora |
|
|
Describe Differential culture media
|
modified with dyes and other agents that change color in the precens of specific microorgs. usually combined with selective and enrichment additives - such as salmonell-shigell agar
|
|
|
Describe Transport culture media
|
sustain growth of microorgs when they cannot be cultured directly after specimen collection. Cary-Blair Transport Medium, Alkaline Peptone Water
|
|
|
Inoculation and Incubation
Asceptic Technique |
to prevent cross-contamination. flame sterized wire loops, straight wires and metal forcepts b4 use. do not allow the instrument to become red hot. cool wire loops by 'punching' the plated media before inoculation. A hot loop will kill organisms of interest.
|
|
|
Inoculation and Incubation
Inoculation |
'plating out' or 'looping out'
commonly used to isolate single colonies or to create undistrurbed confluent growth. |
|
|
Inoculation and Incubation
Confluent Growth |
tests require confluent growth rather than isolative growth. inocuation is prepared in a solution and plated with a swab rather than a loop.
Older method visually approximating turbidity - McFarland Trubidity Scale - a 0.5 McFarland turbidity --> approx 300 bacteria/ml. X&V factor and Kirby-Bauer Test |
|
|
general media prep
|
1. weight out dehydrated media according to instructions on media container
2. slowly mix into 1L distilled water 3. Heat to a biol without over boiling 4. autoclave 121C x 15min |
|
|
Blood Agar (BA) Prep
|
1. weight out dehydrated media according to instructions on media container
2. slowly mix into 1L distilled water 3. Heat to a biol without over boiling 4. autoclave 121C x 15min 5. allow to cool to 50C 6. add 50ml sterile defibrinated blood 7. pour into plates |
|
|
Chocolate Agar (CHOC) Prep
|
1. weight out dehydrated media according to instructions on media container
2. slowly mix into 1L distilled water 3. Heat to a biol without over boiling 4. autoclave 121C x 15min 5. cool to 80C 6. add 1% IsoVitaleX (NAD) 7. add 50ml sterile defibrinated blood 8. pour into plates |
|
|
MacConkey Agar (MAC) Prep
|
1. weight out dehydrated media according to instructions on media container
2. slowly mix into 1L distilled water 3. Heat to a biol without over boiling 4. autoclave 121C x 15min 5. allow to cool to 50C 6. pour into plates |
|
|
Tryptocase Soy Agar (TSA)
|
basic media, non-enriched
base for enriched BA or CHOC |
|
|
Blood Agar (BA)
|
most common enriched media: agar base and defibrinated blood.
Enrichment factor and a differential factor. used to detect hemolytic activity and differentiate the main groups of strep. |
|
|
Beta hemolysis (b)
|
Beta hemolysis (b) - clear is defined as complete or true lysis of red blood cells. A clear zone, approaching the color and transparency of the base medium, surrounds the colony. Many species of bacteria produce toxic by-products that are capable of destroying red blood cells.
GAS or GBS - proceed with Bacitracin Test |
|
|
Alpha hemolysis (a)
|
Alpha hemolysis (a) - green - is the reduction of the red blood cell hemoglobin to methemoglobin in the medium surrounding the colony.
coloration due to oxidized iron in cellular hemoglobin. Some text book authors refer to alpha as “partial hemolysis,” - S.pneumoniae/S.viridans - proceed with Optichin test |
|
|
Gamma hemolysis (g
|
Gamma hemolysis (g ) is somewhat self-contradictory. Gamma indicates the lack of hemolysis. There should be no reaction in the surrounding medium.
Group D Strep/Non-strep organisms |
|

What type of hemolysis am I?
What could I possibly be? What test would I do next? |

Beta hemolytic Streptococcus species,
Could be GBS or GAS Do Bacitracin test next. (FYI, Streptococcus pyogenes (transmitted light) (Lancefield group A). |
|

What type of hemolysis am I?
What could I possibly be? |

No hemolysis - Gamma
could be Group D strep or non-strep organism FYI - Streptococcus agalactiae (Lancefield group B) viewed with incident light: No obvious hemolysis. |
|
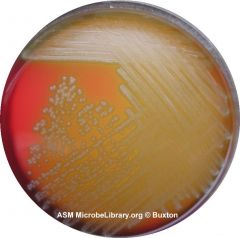
What type of hemolysis am I?
What could I possibly be? What test would I do next? |
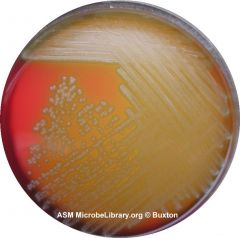
Alpha-hemolytic
Could be S. pneumo or S. viridans Proceed with Optichin test FYI - Streptococcus species “Viridans group” streptococci, including species such as the Streptococcus mutans, mitis, and salivarius groups display alpha hemolysis. |
|
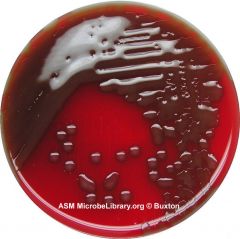
What type of hemolysis am I?
What could I possibly be? What test would I do next? |
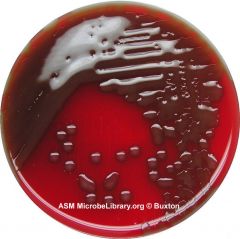
Alpha hemolysis
s. pnuemo/s.viridans do optichin test next. FYI - Streptococcus pneumoniae (Encapsulated strain). |
|

What type of hemolyis am I?
Who could I be? |

NO hemolysis
group d strep or non-strep organisms FYI = Listeria monocytogenes, removing colonies to see the subtle pink hemolysis directly beneath the colonies |
|
|
Chocolate Agar (CHOC) - what grows on it and why?
|
Chocolate agar (CHOC) - is a non-selective, enriched growth medium. It is a variant of the blood agar plate. It contains red blood cells, which have been lysed by heating very slowly to 80 °C. Chocolate agar is used for growing fastidious (fussy) respiratory bacteria, such as Haemophilus influenzae. These bacteria need growth factors, like NAD(factor V) (IsoVitaleX) and hematin (Factor X), which are inside red blood cells; thus, a prerequisite to growth is lysis of the red blood cells. The agar is named for the color and contains no actual chocolate.
supplies factors for H.influenza and Nisseria growth Since this medium supports many common bacteria, it is not useful for fecal cultures. for h.influenza to grow on it - must be incubated in an anaerobic (high CO2) environment. |
|
|
Chocolate Agar (CHOC) - grows only on it?
|
X&V factor test (+), gn coccobacilli --> H.influenza
gn, oxidase (+) cocci --> GC, N.meningitis |
|

what am I and what can be tested on me?
|
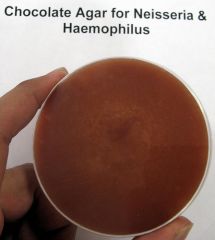
GC and H.influenza
|
|
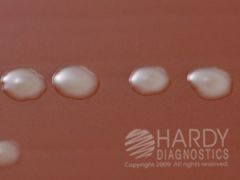
what agar am I and what could I be?
|
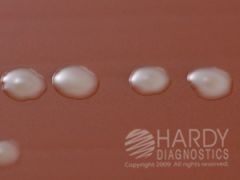
CHOC
GC, N.mening, H.flu FYI - Haemophilus influenzae (ATCC® 10211) colonies growing on Chocolate Agar (Cat. no. E14) Incubated in CO2 for 24 hours at 35 deg. C. |
|
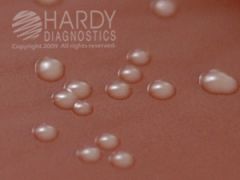
what agar am I and what is tested on me?
|
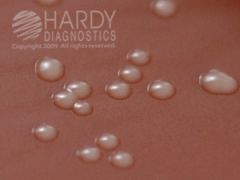
CHOC
GC Nmening, Hflu FYI - Neisseria gonorrhoeae (ATCC® 43069) colonies growing on Chocolate Agar (Cat. no. E14) Incubated in CO2 for 24 hours at 35 deg. C. |
|
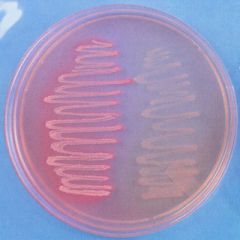
What agar am I?
What's special about me? |
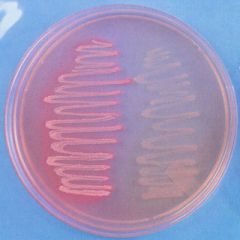
MacConkey agar is a culture medium designed to grow Gram-negative bacteria and stain them for lactose fermentation
- selective and differential media - bile salts inhibit gp growth - distingues lactose fermenters from non-ferm - color: light pink FYI: E. coli and a non-lactose fermentation - MacConkey agar inoculated with lactose-fermenting Escherichia coli (at left) and non-lactose-fermenting Proteus (at right). Lactose-fermenting bacteria appear bright pink, while non-lactose-fermenting bacteria appear colorless. This demonstrates the differential nature of MAC agar. |
|
|
MacConkey (MAC) agar -
|
used to grow enteric GN bacteria.
Agara contains bile salts and crystal violet dye which inhibit gram pos growth. non-lactoce ferms metabolize the peptone and produce byproducts that are basic. fermenters- hot pink nonferms - loose color become more clear. MacConkey agar is a culture medium designed to grow Gram-negative bacteria and stain them for lactose fermentation - selective and differential media - bile salts inhibit gp growth - distingues lactose fermenters from non-ferm - color: light pink |
|
|
MacConkey (MAC) agar
Pink : lactose (+) - what do you do now? - what orgs do you consider? |
proceed with oxidase and indole tests
e.coli klebsiellea enterobacter |
|
|
MacConkey (MAC) agar
clear: lactose (-), peptone (+) - what do you do now? - what orgs do you consider? |
proceed with motility tests and enteric growth plates
shigella salmonella proteus |
|
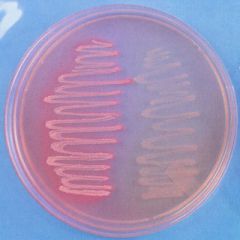
what media am I?
How do you know? |
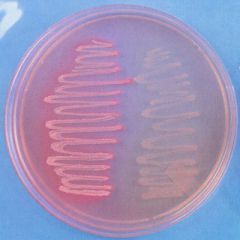
lactose fermenter- barbie pink
nonlactose fermenter - grandma pink |
|
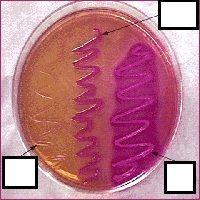
what agar am I?
|
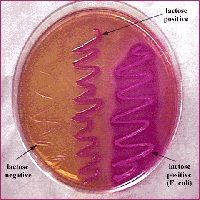
MAC
lactose fermenter - barbie pink lactose nonferm - grandma pink |
|
|
GN or GP - than what do you do?
-BA: no -CHoc: yes -Mac: no |
GN
Enteric or haemophilus - do X&V or Satelite test X&V (+) H.influenza x&V (-) Niseeria or Haemophilus parainfluenza |
|
|
GN or GP - than what do you do?
-BA: yes -CHoc: yes -Mac: yes |
gn
entric pahtogens: klebsiella, pseudomonas, e.coli do oxidase test (negative) lactose fermenter? Yes (MAC +) --do indole test: |
|
|
GN or GP - than what do you do?
Who am I? -BA: yes -CHoc: yes -Mac: no - catalase pos ?now what? |
GP cocci
staphylococcus do coagulase test to differentiate S. aureas (+) from coagulase (-) S. epi or S. saprophylticus |
|
|
GN or GP - than what do you do?
-BA: yes -CHoc: yes -Mac: no What do you do next? |
GP
Catalase test (staph vs. strep) |
|
|
What is X&V test for?
|
GN, - Choc
X&V (+) H.influenza x&V (-) Niseeria or Haemophilus parainfluenza |
|
|
Enteric Pathogen Medias
|
Salmonella-Shigella Agar (SS)
Xylose-lysine-deoxycholate Agar (XLD) Thiosulfate Citrate Bile Salts Sucrose Agar (TCBS) Campylobacter Agars (Campy) Hektoen Agar (HEK) |
|
|
Screening tests for fecal leukocytes
|
methylene blue staining
leuko-test assay |
|
|
what is the classic test to differentiate inflammatory vs noninflam diarrhea?
|
Methylene blue staining.
|
|
|
Advantages of Methylene blue staining
|
inexpensive, easy to perform, no refrigeration
|
|
|
Disadvantages Methylene blue staining
|
requires microscope and training
|
|
|
What test uses agglutination of latex beads?
|
Leuko-test assay
visible agglutination of latex beads coated wtih anitlactoferrin antibodies |
|
|
Disadvantages of leuko-test assay
|
cost, requires refrigeration, interferance by breast milk lactoferring, requires plastic tubes for dilution
|
|
|
advantages of leuko-test assay
|
easy to perform and read
sensitive |
|
|
Tests for Vibrio cholerae
|
1. co-agglutination (choleraScreen)- no longer in US
2. membrane antigen immunoassay (cholerasmart) 3. Cholera test strips and slides (smart strip cholera, smart check cholera) 4. culture |
|
|
1. co-agglutination (choleraScreen)- no longer in US
advantages and disadvantages |
easy to preform, adaptable to field situations
dis: cost, refrig |
|
|
2. membrane antigen immunoassay (cholerasmart)
concept, advan and disadvant |
monocolonal antibodies in conjugate bound to colloid gold particles
advant: no refrig, easy to perform, read, applicable to field dis: cost |
|
|
3. Cholera test strips and slides (smart strip cholera, smart check cholera)
concept, advan and disadvant |
dipstrip or slide method with colorimeteric changes, reliablitiy unknown, no refrig
|
|
|
culture
|
gold standadrd - plated on thiosulphate citrate bile salt agar (TCBS)
highly selective for vibrios based on sucrose fermentation: v.cholerae ferments sucrose and turns YELLOW V. parahemolyticus - sucrose negative |
|
|
BA (+)
MAC (?) What test do you do? |
Oxidase test - to see if enterobacteria or bacilli
|
|
|
BA(+)
MAC (?) Oxidase (-) what am I and what next? |
Enterobacteria
?lactose fermenter? -- yes - do indole test -- no - do XLD test |
|
|
Salmonella-Shigella Agar (SS)
|
selective media
ID of lactose and H2S differentiation of salmonella from shigella SS agar contains bile salts, lactose and peptone (like mac) Lac (+) Ferm - pink Lac (-) ferm- clear UNlike Mac: higher concentration of bile salts to detect H2S production -- H2S producers turn BLACK Pink: lactose fermenters colorless and black - salmonella colorless and clear colonies - shigella |
|
|
Xylose-lysine-deoxycholate agar (TCBS)
|
speical agar for growth and isolation of vibrios
often used in combo with transport media - alkaline-peptone water bile salts inhibit gp growth. enterobacteria inhibited by high concentration of thiosulfate and citrate Yellow: Sucrose (+) : V.cholerae - proceed with slide agglutination and serotyping blue-green: sucrose (-): v.parahemolyticus and other vibrios |
|
|
Camplobacter agars (Campy)
|
special for campy jejuni - brucella agar base (very enriched) and Skirrows or butlzler antibiotic mixture
camy agars are incubated for 2-3 days in low oxygen tension at 42C. Growth and oxidase (+) : gs and observe under microscope micoroscope - gn 'seagull'rods - campy jejuni |
|
|
Hektoen agar (HEK)
|
selective media
xld like expensive and difficult to read |
|
|
new method - Camplobacter (Campy) - membrane filtration culture
|
campy growth on agar is based on size not antibiotic resistance
|
|
|
What are the gram positive tests?
|
catalase
coagulase optochin bacitracin CAMP |
|
|
What are the gram negative tests?
|
oxidase
indole X&V Satellite |
|
|
What are the microbiotic sensitivity tests?
|
Kirby-Bauer
E test D test |
|
|
What does catalase test do?
|
differentatiates staph from step
|
|
|
What does coagulase test do?
|
differentiates staph aureus from other staph
|
|
|
What does optochin test do?
|
alpha hemolyltic strep differentiation
|
|
|
What does bacitracin test do?
|
beta hemolytic strep differentiation
|
|
|
What does CAMP test do?
|
poor man's bacitracin - beta hemolyic strep differentiation
|
|
|
What does oxidase test do?
|
differentatess the enterics from other gn species
|
|
|
What does indole test do?
|
differfentiates the enterics
|
|
|
What does X&V test do?
|
differentiates Haemophilus spp. from other fastidious gn
|
|
|
What does satellite test do?
|
poor man's x&V - differentiates Haemophius spp from other fastidious gn
|
|
|
What does Kirby-bauer test do?
|
disc diffusion antibiotic sensitivity
|
|
|
What does E test do?
|
concentaration gradient antibiotic sensitivity
|
|
|
What does D test do?
|
differentiates methicilliin sensitive S. auereus from methicillin resistant S. aureus
|
|

What test am I?
What do I do? |

Catalase is a common enzyme found in nearly all living organisms that are exposed to oxygen, where it functions to catalyze the decomposition of hydrogen peroxide to water and oxygen.
differentiate staph from strep |
|
|
catalase test
|
If bubbles or froth forms, the organism is said to be catalase-positive. Staphylococci[30] and Micrococci[31] are catalase-positive. Other catalase positive organisms include Listeria, Corynebacteria diphtheriae, Burkholderia cepacia, Nocardia, the Family Enterobacteriaceae (Citrobacter, E.Coli, Enterobacter, Klebsiella, Shigella, Yersinia, Proteus, Salmonella, Serratia, Pseudomonas), Mycobacterium Tuberulosis, Aspergillus, and Cryptococcus.
If not, the organism is catalase-negative. Streptococci[32] and Enterococci are catalase-negative. While the catalase test alone cannot identify a particular organism, combined with other tests such as antibiotic resistance, it can aid diagnosis. |
|

what test am I?
What do I do? |

The coagulase test is used to differentiate the potentially pathogenic species Staphylococcus aureus from the usually non-pathogenic species Staphylococcus epidermidis. The presence of coagulase results in the formation of a clot in a tube of citrated platelet-rich plasma (~ >150 x 106 platelets/cc plasma). The citrate is an anticoagulant that is added to avoid autoclotting.
|
|

what test am I?
What do I do? |

Coagulase test
Illustrated in this photograph is a positive latex slide coagulase test in the left frame, compared to a negative control in the right frame. Is the slide test more specific than the tube test in the identification of Staphylococcus aureus; or, again, are there other possibilities |
|

interpret results
|

Coagulase test
Positive (+) S. Aureus clearly visible clumping of the latex particles with clearing of the milky background. Almost instantaneous. Negative - S. epidermidis/saprohylticus - traces of granulartity may be seen in negative patterns due to isolate-reagent interactions. Increased granularity may be observed if the latex suspension is rotated for more than 20 seconds. |
|

what test am I?
What do i do? |
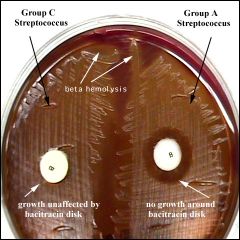
The bacitracin susceptibility test is used to distinguish Group A streptococci, which cause ninety percent of acute streptococcal infections in humans, from other streptococci. When grown on blood agar, Group A streptococci are sensitive to (killed by) the antibiotic bacitracin . A sterile disk impregnated with bacitracin is placed on the first sector of an isolation plate before incubation. A zone of inhibition (area with no growth) will be seen around the disk after incubation if the organism is a Group A beta-hemolytic Streptoccus. Other beta-hemolytic streptococci are resistant to (not killed by) bacitracin. Their colonies will thus grow right up to the disk of bacitracin
|
|
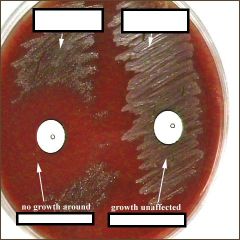
what test am I?
What do I do? |
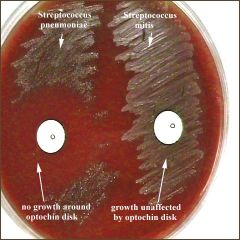
Optochin (P Disk)
Differentiates alpha hemolysis streps a quinine derivative, has a detergent-like action and causes selective lysis of pneumococci. A sterile disk impregnated with optochin is placed on the first sector of an isolation plate before incubation. A zone of inhibition (area with no growth) of 14 mm. or more in diameter will be seen around the disk after incubation if the organism is Streptococcus pneumoniae. Other alpha-hemolytic streptococci are resistant to (not killed by) optochin. Their colonies will thus grow right up to the disk of optochin or have zones of inhibition less than 14 mm. in diameter. |
|
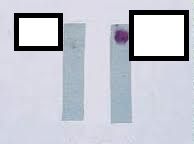
what test am I?
What do I do? |

Oxidase Test - Is it an Enteric pathogen?
Enteriobacteria are oxidase Negative Postive test - change purple within 30 seconds - pseudomonas, aeromonas, and others Negative - no color change or light yellow - enteric pathogen This test is used to determine the presence of the cytochrome oxidase. Cytochrome oxidase is an enzyme that transfers hydrogen in the electron transport chain. ( The electron transport chain is the final step in aerobic repiration or oxidative phosphorylation ) If bacteria are capable of transferring electrons the paper will turn purple. A blue- purple color produced is indicative of a positive test. |
|

what test am I?
What do I do? |

indole test
is it e.coli? Positive changes red or blue within 1-3min - Escherichia coli Neg- no cholor or light yellow - Klebsiella and others |
|

what test am I?
What do I do? |

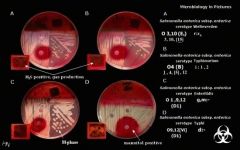
X, V, X+V Factor Test, top view
This test demonstrates Haemophilus influenzae's unique growth requirements. H.influenzae require NAD and heme factors (X and V) in order to grow. Growth is represented by the milky halo around the X+V factor disk (top center of plate). The factors do not diffuse throughout the entire quadrant. Growth only appears as far as the factors have diffused into the agar. |
|
|
X & V factor test - test basics
|
Species of the genus Haemophilus require either or both of two factors X and V for growth and can be
used to differentiate the species. Both factors are present in blood. X factor comprises protoporphyrin IX, haemin or other iron-containing porphyrins. These are required for growth because X-dependent strains are unable to convert d-aminolaevulinic acid to protoporphyrin. V factor comprises nicotinamide adenine dinucleotide (NAD) or nicotinamide adenine dinucleotide phosphate (NADP)2. The factors are incorporated in filter paper discs which are placed on a blood free medium previously inoculated with the organism under test. After incubation, the presence or absence of growth around the discs is recorded. The presence of growth around the disc but not elsewhere on the place indicates a requirement for that particular factor. |
|
|
X & V factor test - reslts
|
Positive: growth between factors X and V ONLY - H.influenza
Neg: growth around v - H.parainfluenza Factor H. influenzae H. parainfluenzae XV + + X - - V - + |
|
|
X &V test- method
|
1. saline suspension 0.5 McFarland turbidty. DO NOT transfer any choc agar.
2, using a SWAB, inoculate the supsension on a plate of TSA 3. place factor strips 10-20mm apart 4. incubate 24h in a 5-10% CO2 at 35C. |
|
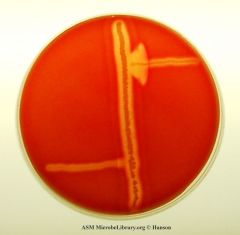
what test am I?
What do I do? |
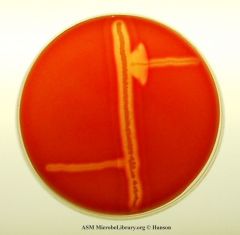
The CAMP test is a test to identify Group B β-streptococci based on their formation of a substance (CAMP factor) that enlarges the area of hemolysis formed by β-hemolysin from Staphylococcus aureus. It is frequently used to identify Listeria spp.
Although usually used to identify group "B", there is some evidence that the CAMP factor gene is present in several groups of streptococci, including group "A" CAMP test showing the arrow-shaped zone of enhanced hemolysis (positive result) of Streptococcus agalactiae (group B) and the negative result of Streptococcus pyogenes (group A) when tested against Staphylococcus aureus. (Anne Hanson, University of Maine) |
|
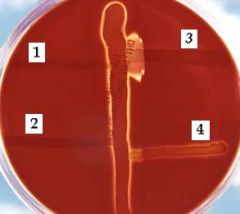
what test am I?
What do I do? |
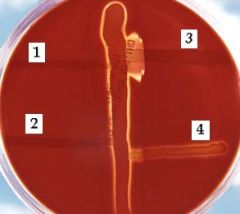
CAMP test - poor man's bacitracin test.
differentiating beta-hemoltyic strep from another. In this photo, a Blood Agar plate is shown after 24 hours of incubation at 37°C. The vertical streak is a beta-hemolysin-producing strain of Staphylococcus aureus, and at right angles to it are streaks of (1)Enterococcus faecalis, (2)Streptococcus salivarius, (3)S. agalactiae (GBS - enhanced hemolysis), and (4)E. durans. Note the large area of complete lysis where the extracellular compound of S. agalactiae encounters the beta-lysin of S. aureus. |
|
what test am I?
What do I do? |
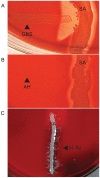
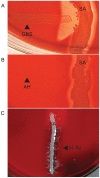
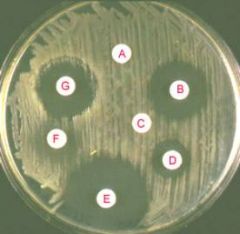
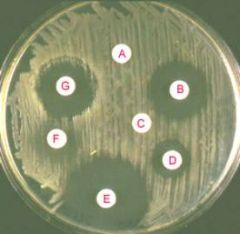
A) The CAMP test for identification of Group B streptococcus (horizontal streak, GBS) demonstrates an arrowhead shaped area of enhanced hemolysis when grown near S. aureus (vertical streak, SA). (B) The reverse CAMP test for identification of Arcanobacterium haemolyticum demonstrates an indentation in the linear area of hemolysis by S. aureus (vertical streak, SA) when it grows adjacent to A. haemolyticum (horizontal streak, AH). (C) The satellite test for identification of Haemophilus influenzae demonstrates growth of the organisms as small satellite colonies (arrowhead, H. flu) adjacent to S. aureus (vertical streak, SA). Similar satellite colony formation around S. aureus was seen for Abiotrophia defectiva. Similar results were seen on both defibrinated and citrated hair sheep blood agar; results are shown on citrated hair sheep blood agar plates.
|
|
what test am I?
What do I do? |
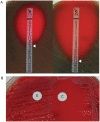
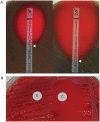
(A) Determination of MIC values for penicillin by E-test for a resistant (left) and a susceptible (right) strain of S. pneumoniae. (B) The D-test demonstrates inducible clindamycin resistance in a S. aureus strain carrying an inducible erm gene. A ?D?-shaped area of clearing is seen due to increased growth around the clindamycin disk (labeled ?C?) where it lies closest to the erythromycin disk (labeled ?E?). Similar results were seen on both defibrinated and citrated hair sheep blood agar; results are shown on citrated hair sheep blood agar plates.
|
|
what test am I?
What do I do? |
Kirby Bauer method using a Mueller-Hinton II agar plate
Procedure: Measure zones of inhibition → determine which antibiotics your organism is sensitive (S), resistant (R) or intermediate (I). This along with the rate of antibiotic diffusion are used to estimate the bacteria's sensitivity to that particular antibiotic. In general, larger zones correlate with smaller minimum inhibitory concentration (MIC) of antibiotic for that bacteria. A large zone of inhibition does NOT necessarily indicate a more effecive antibiotic than a small zone. |
|
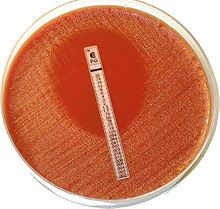
what test am I?
What do I do? |
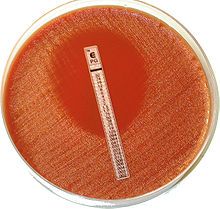
The Etest is basically an agar diffusion method.
The Etest utilises a rectangular strip that has been impregnated with the drug to be studied. A lawn of bacteria is spread and grown on an agar plate, and the Etest strip is laid on top; the drug diffuses out into the agar, producing an exponential gradient of the drug to be tested. There is an exponential scale printed on the strip. After 24 hours of incubation, an elliptical zone of inhibition is produced and the point at which the ellipse meets the strip gives a reading for the minimum inhibitory concentration (MIC) of the drug. |
|
What bug am I?
What tests do you use to diagnose me? |
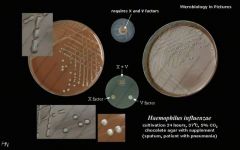
Haemophilus influenza
choc agar xv factor |
|
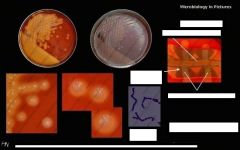
What bug am I?
What tests do you use to diagnose me? |
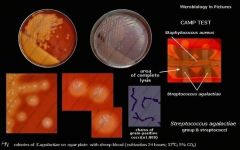
Streptococcus agalactiae (GBS)
BA camp chains |
|
What bug am I?
What tests do you use to diagnose me? |
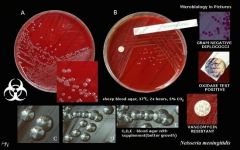
Neisseria meningitis
GN diplocicci BA - no hemolysis oxidase pos |
|
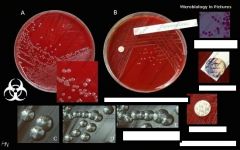
What bug am I?
What tests do you use to diagnose me? |
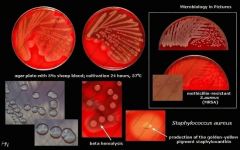
Staphylococcus aureus
BA - partial hemolysis |
|
What bug am I?
What tests do you use to diagnose me? |
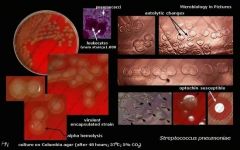
Streptococcus pneumoniae
alpah hemolysis autolytic changes optochin sensitive |
|
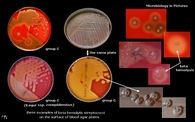
What bug am I?
What tests do you use to diagnose me? |
Beta-hemolytical streptococci
|
|
What bug am I?
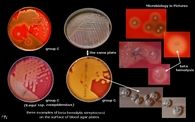
What tests do you use to diagnose me? |
salmonella
H2S production from XLD test |
|
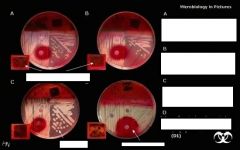
What bug am I?
What tests do you use to diagnose me? |
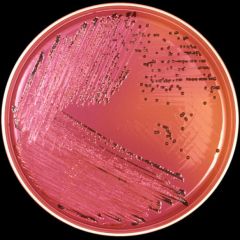
Salmonella sp. after 24 hours growth on XLD agar. Xylose Lysine (XL) agar is used when trying to culture and isolate Gram-negative enteric bacilli. When XL agar is supplemented with sodium thiosulfate, ferric ammonium citrate, and sodium deoxycholate, it is then termed XLD agar, and is then an even more selective medium than XL alone. The presence of any black colored area indicates the deposition of hydrogen sulfide, (H2S) under alkaline conditions. (CDC)
|
|
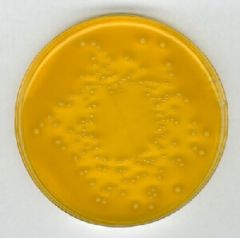
What bug am I?
What tests do you use to diagnose me? |
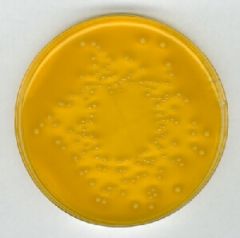
XLD lactose fermenter - klebsiella
|
|
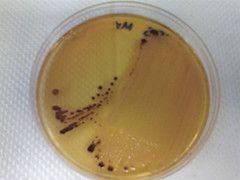
What bug am I?
What tests do you use to diagnose me? |
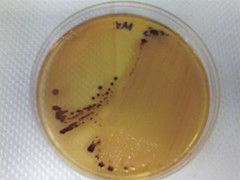
Salmonella-shigella agar
colorless with black colonies - salmonella |
|
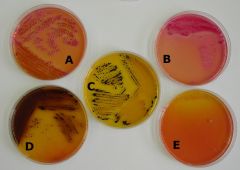
What bug am I?
What tests do you use to diagnose me? |
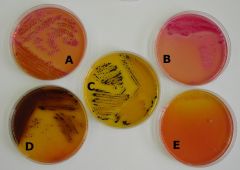
pearance of Colonies on Salmonella-Shigella Agar
A. Klebsiella pneumoniae B. Escherichia coli Klebsiella pneumoniae & Escherichia coli are positive for acid production from fermentation of the carbohydrate(s) present. C: Salmonella sp. D: Proteus mirabilis Both Salmonella sp. & Proteus mirabilis product hydrogen sulfide. E: Pseudomona aeruginosa The Pseudomonas colonies are nearly colorless. |
|
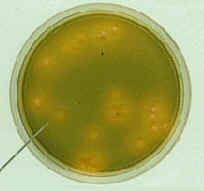
What bug am I?
What tests do you use to diagnose me? |
Thiosulfate Citrate Bile Salt Sucrose agar plate (TCBS)
Use : For the selective isolation of Vibrio cholerae and Vibrio parahaemolyticus from a variety of clinical specimens and in epidemiological investigations. Control organisms : Vibrio cholerae : Growth (yellow colonies) Vibrio parahaemolyticus : Growth (green colonies) Staphylococcus aureus : No growth Incubate : Aerobic 35° C, 24 hrs Shelf life : 4 weeks Price (Baht) : 15 (return plate) , 18 (disposable plate) |
|

What bug am I?
What tests do you use to diagnose me? |

Vibrio_cholerae_on_TCBS_agar.jpg
|
|
|
grows on BA and Choc, what do you do next?
|
microscopy - to see if gn or gp
|
|
|
grows on BA MAC AND CHOC
|
enteric pathogens
|
|
|
grows on choc only?
Next test? |
Haemophilus
X&V or Satellite test |
|
|
BA and CHOC only, Oxidase +, what do you do next?
|
microscopy - if bacilli - can be psuedomonas, vibrio, or campylobacter
if cocci - niesseria |
|
|
BA & CHOC only, Oxidase -, what do you do next?
|
enterobacteria,
- already know not lactose fermenter bc, MAC was neg - need XLD and motility test |
|
|
BA and CHOC +
MAC -, Oxidase - motililty - or H2S (XLD) - |
Shigella
|
|
|
BA and CHOC +
MAC -, Oxidase - motililty + or H2S (XLD) + |
Salmonella
|
|
|
BA, CHOC, MAC +
Oxidase - What test next? |
Indole
+ E.coli - Klebsiella (slimmy appearance) |
|
|
BA, CHOC, MAC +
Oxidase + Lactose - What am I? |
Psuedomonas
|
|
|
BA, CHOC, MAC +
Oxidase + Catalase + Seagull rods What am I? |
Campylobacter
|
|
|
BA, CHOC, MAC +
Oxidase + TCBS or Sucrose + Indole + What am I? |
Vibrio
|
|
|
CHOC, +
Oxidase + Fastidious intracellur 'kidney bean' diplococci on choc What am I? |
Neisseria (red very small v's)
|
|
|
What does SEcKS-P stand for?
|
Gram Negative mneumonic for Enterobacteria:
(sounds like Sexy-P) Salmonella E.coli Klebsiella Shigella Proteus |
|
|
What does the KEE of MacConKEE's Agar stand for?
|
Gram Negative mneumonic
Lactose fermenters use MacConKEE's agar: Klebsiella E.coli Enterobacter |
|
|
alpha-OVRPS (overpass)?
|
Gram Positive Mneumonic:
Alpha-hemolytic Optochin: Viridans is Resistant Pneumo is Sensitive |
|
|
Beta-BRAS:?
|
Gram Positive Mneumonic:
Beta-hemolytic Bacitracin group B is Resistant group A is Sensitive |
|
|
No StRESS?
|
Gram Positive Mneumonic:
Novobiocin: Saphro is Resistant Epi is Senstive |
|
|
growth on BA, Catalase -, No hemolysis on BA
|
Gamma:
Enteroccus or Peptostreptococcus |
|
|
growth on BA, Catalase -, Parial hemolysis on BA,
Optochin Resistant (-) |
alpha:
S. viridans |
|
|
growth on BA, Catalase -, Parial hemolysis on BA,
Optochin Sensitive (+) |
alpha:
S. pneumoniae |
|
|
growth on BA, Catalase -, Complete hemolysis on BA,
Bacitracin Sensitive (+) |
beta:
Strep pyrogens (GAS) |
|
|
growth on BA, Catalase -, Complete hemolysis on BA,
Bacitracin Resistant (-) |
beta:
Strep aglactiae (GBS) |
|
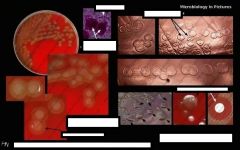
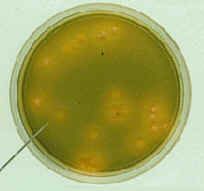
what is catalase negative, otochin sensitive, on microscopy has chains and lancet shapeed diplococci, has mucoid growth on BA, colonies with central umbilication, green hemolysis and is the same on CHOC?
|
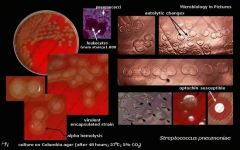
Streptococcus pneumoniae
|
|
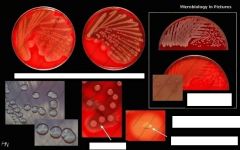
What is catalase positive, coagulase positive, growth in clusters on microscopy, has large, white-yellow, creamy colonies on BA (no hemolysis) and CHOC
|
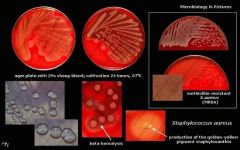
Staphococcus aureus
|
|
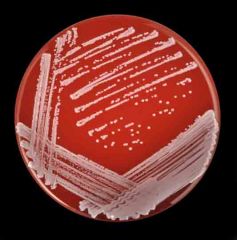
What is catalase positive, coagulase negative , growth in clusters, has white-grey, raised and creamy colonies on BA (no hemolysis) and CHOC
|
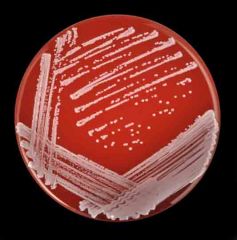
Staphylococcus epidermidis
|
|

what is catalase neg, alpha hemolyisis, Opticin resistant,
microscopy: pairs and chains, on BA has small to medium colonies, carmel odor, green hemolyis and same on CHOC? |

Streptococcus viridians
|
|
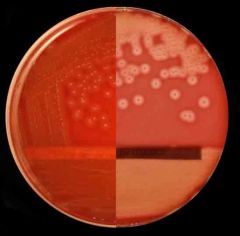
what is catalase neg, beta hemolysis, bacatracin sensitive,
microscopy growth in pairs and chains, on BA small to medium, translucent dull greay colonies, clear hemolysis, CHOC no hemolysis? |
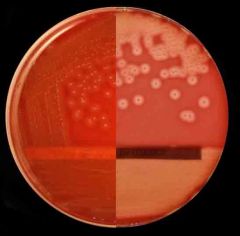
Streptococcus pyogens GAS
|
|
What is catalase negative, beta hemolysis, bacatracin resistant, microscopy growtth in pairs and chains, and on BA is small to medium, translucent dull grey colonies, clear hemolysis, and no hemolysis on CHOC?
|
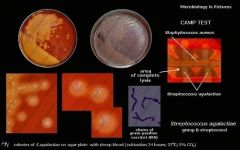
Streptococcus agalactae (GBS)
|
|
|
What are the fastidious growth bugs?
|
Haemophilus influenza
Haemophilus parainfluenza Neisseria gonrrhoeae Neisseria meningitis |
|
|
What are the fastidious growth that don't grow on MAC or BA?
|
Haemophilus influenza
Haemophilus parainfluenza Neisseria gonrrhoeae |
|
|
What does a positive satellite test mean?
|
Haemophilus influenza
|
|
|
What does a negative satellite test mean?
|
NOT Haemophilus influenza
Can be: Haemophilus parainfluenza Neisseria gonrrhoeae Neisseria meningitis |

