![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
143 Cards in this Set
- Front
- Back
|
electrochem is the study of relat betw chemical react and ?
|
electrical energy
|
|
|
oxidation is ______ of an electr
|
loss
|
|
|
reduction is ________ of a electron
|
gain
|
|
|
oxidat always occur with a?
|
reduct
|
|
|
oxidizing agen causes?
|
another atom in a redox react to undergo oxidat and is itself reduced
|
|
|
OIL RIG
is a way to remember that? |
oxidation is loss, Reduction is gain
|
|
|
why do we use oxidation numbers?
|
to keep track of how many electr are gained or lost in a react
|
|
|
oxidation of free elements is?
|
0
|
|
|
N2 , N has an oxidat number of?
|
0
|
|
|
He has an oxidat number of?
|
0
|
|
|
oxidation number for a monoatomic number is equal to?
|
the charge of the ion
|
|
|
Na+ has an ox n of?
|
+1
|
|
|
oxidation number of each group IA element in a compound is?
|
+1
|
|
|
oxidation of each gr IIA element in a compound is?
|
+2
|
|
|
oxidation number of group VIIA element in a compound is?
|
-1 except when combined with an element of higher electroneg
|
|
|
HCl. ox number of Cl is?
|
-1
|
|
|
HOCl, oxidat numb of Cl is?
|
+1
|
|
|
NaH, ox n of H is?
|
-1
|
|
|
CaH2, oxidat number of H?
|
-1
|
|
|
the ox n of H is _______ in comp with less electr elements than Hydrogen
|
-1
|
|
|
the more common ox n of H is?
|
+1
|
|
|
in most compd the oxidat number of O is?
|
-2
|
|
|
OF2, oxid number of O is?
|
+2. bec F is more electroneg than O
|
|
|
in peroxides like BaO2, the oxidat number of O is?
|
-1
|
|
|
sum of the oxidat numbers in all the atoms present in a neutral comp is?
|
zero
|
|
|
sum of the oxidat number of the atoms pres in a polyatomic ion is equal to?
|
the charge of the ion
|
|
|
SO42-, the sum of the oxidat numbers must be?
|
-2
|
|
|
SnCl2 + PbCl4 --> SnCl4 + PbCl2
which react is oxidized? |
Sn. it goes from +2 to +4
|
|
|
most common method for balanc redox equat is the?
|
half react method. also called the ion electron method
|
|
|
half reaction method is?
|
used to balance the redox react. equat is seperated into 2 half react. oxidat part and the reduct part
|
|
|
Mn04- + I- ---> I2 + Mn2+
what are the 2 half reactions? |
I- --> I2
Mn04- ---> Mn2+ |
|
|
what are the steps for balancing an oxidat reduct reaction?
|
1. seperate the 2 half react
2. balance the atoms in each half react. 3. |
|
|
when you are balanc the atoms in oxid reduct what atoms do you leave till last?
|
H and O
|
|
|
oxid reduct
in an acidic solut, add ________ to balance the O atoms and then add _____ to bal the H atoms |
H2O,
H+ |
|
|
oxid reduct
in a basic solut add _________ to balance the O's and H's |
OH-, and H20
|
|
|
MnO4- ---> Mn2+
balance the half reaction |
Mn04- --> Mn2+ + 4H20
Mn04- + 8 H+ --> Mn2+ + 4H20 |
|
|
balane the charges in each half reaction
|
2I- ---> I2+ 2e-
5 e- + 8 H+ + Mn04- --> Mn2+ + 4 H20 |
|
|
2I- ---> I2+ 2e-
5 e- + 8 H+ + Mn04- --> Mn2+ + 4 H20 after you bal the charges in each half react what do you need to do? |
make sure both half react have the same numb of electr so they will cancel.
|
|
|
2I- ---> I2+ 2e-
5 e- + 8 H+ + Mn04- --> Mn2+ + 4 H20 what do you do next? |
5(2I- --> I2 + 2e-)
2(5e- + 8H+ + Mn04- --> Mn2+ + 4 H20) |
|
|
5(2I- --> I2 + 2e-)
2(5e- + 8H+ + Mn04- --> Mn2+ + 4 H20) what do you do next? |
add the half reactions
10 I- --> 5I2 + 10 e- 16 H+ + 2 Mn04- 10 e- --> 2 Mn2+ + 8 H20 |
|
|
10 I- --> 5I2 + 10 e-
16 H+ + 2 Mn04- 10 e- --> 2 Mn2+ + 8 H20 what will you do to get the final equation? |
cancel out the electrons and
any H20s, H+s, or OH-s that appear on both sides of the equation. 10 I- + 16 H+ + 2 Mn04 ---> 5 I2 + 2 Mn2+ + 8 H20 |
|
|
AN OX and a RED CAT
stands for? |
anode is oxidation
cathode is where reduction occurs |
|
|
what are the 2 types of electrchem cells?
|
1. galvanic c or voltaic c
2. electrolytic cells |
|
|
spontan react occur in _______ cells?
|
galvanic or voltaic cells
|
|
|
galvanic cells are also called?
|
voltaic cells
|
|
|
nonspon react occur in ?
|
electrolytic cells
|
|
|
galvanic and electro cells have?
|
electrodes
|
|
|
redox in a galvanic cell has a _______ change in G
|
NEGATIVE
|
|
|
galvanic cells ________ energy
|
supply
|
|
|
galvanic cells
oxid and reduct react are put in seperate? |
half cells
|
|
|
batteries are galvanic or electrolytic cells?
|
galvanic
|
|
|
daniell c is an example of?
|
a galvanic c
|
|
|
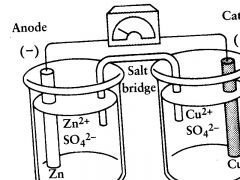
daniell c
zinc bar is put in a? |
ZnSO4 solution
|
|
|
danielle c
copper bar is put in ? |
CuSO4 solution
|
|
|
danielle cell
anode is? |
zinc bar where Zn is oxidized
|
|
|
daniell cell
cathode is? |
the copper bar. site of reduction of Cu2+ to Cu
|
|
|
daniell c
what happens at the cathode? |
Cu2+ is reduced to Cu
|
|
|
daniell cell
what would happen if the 2 half c were not seperated? |
the Cu2+ ions would react directly with the zinc bar and no useful electr work would be done
|
|
|
daniell c
why do you need a salt bridge and a wire to connect the half reactions? |
bec otherwise an excess negat ch would build up in solut sorrounding the cathode, and exc posit ch in solut sorrounding the anode
|
|
|
daniell c
salt bridge does what? |
permits the exch of cations and anions
|
|
|
daniell cell
the salt bridge usually has? |
an inert electrolyte like KCl or NH4NO3.
these ions wont react with the electrodes or with the ions in solution |
|
|
daniell cell
the cations of the salt bridge flow into the ____________ to bal out the charge of __________ |
CuSO4 solution
SO42- ions left in solution when the Cu2+ ions deposit as copper metal |
|
|
daniell cell
electrons flow from _______, toward the ____________ |
anode,
cathode |
|
|
daniell cell
electrons flow from the zince bar, which is the ________. |
anode
|
|
|
daniell cell
what does the flow of electrons do to the cell? |
it depletes the salt bridge and uses up the Cu2+
|
|
|
cell diagram
what are the rules for writing it? |
1. react and prod are listed from left to right
anode I anode solut II cathode sol I cathode 2. single vertical line repres a phase boundary 3. double vertical line repres the salt bridge or |
|
|
cell diagram
how are the reactants and products written? |
anode I anode solution II cathode sol I cathode
|
|
|
cell diagram
single vertical line is a? |
phase boundary
|
|
|
cell diagram
double vertical line is? |
presence of a salt bridge or some other barrier
|
|
|
electrolytic c has a ________ G
|
positive
|
|
|
electrolytic cell is spontan or nonspont?
|
nonspontaneous
|
|
|
electrolysis
the half reactions are placed? |
in the same container
|
|
|
Zn is oxid to Zn2+
|
what happens at the anode?
|
|
|
electrolytic c
michael faraday theory the amount of chem change is directly proportional to? |
number of moles of electr that are exch during a redox react
|
|
|
electr c
number of moles exch can be determ from? |
balanced half reaction
M n+ + ne- --> M (s) |
|
|
M n+ + ne- --> M (s)
one mole of M will be prod in an electr c if ________ moles of electr are supplied |
n
|
|
|
electr cell
How do you calculate the charge of a mole of electrons? |
(1.6 X 10-19 C)( 6.022 X 1023 mole-1e-1)= 96487 C/mol e-
|
|
|
electr cell
what is the charge of a mole of electrons? |
96,487 C/mol e-
|
|
|
faradays constant is?
|
1 F=96,487 C or J/V
|
|
|
t or f
1 f=96,487 c |
t
|
|
|
reminder from physics
J=? |
J=VC
|
|
|
in an electrolytic cell the anode is __________ and the cathode is __________
|
anode is positive
cathode is negative |
|
|
in a galvanic cell, the __________ is negative and the cathode is __________
|
anode is negative,
cathode is positive |
|
|
in an electrolytic cell
reduction occurs at the _______ and the __________ is posit and the _______ is negat |
reduct occurs at cathode
anode is positiv cath is negative |
|
|
galvanic cell
_________ occurs at cathode ____ is neg _____ is positv |
galv cell
reduct occurs at cathode anode is negat cath is positiv |
|
|
electrolytic cell
molten NaCl is electrolyzed to form? |
Cl2(g) and Na (l)
|
|
|
oxidation
|
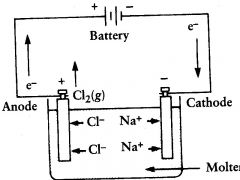
what occurs at the anode?
|
|
|
cathode
|

Na+ migrates toward the?
|
|
|
it floats at the top and can be removed
|
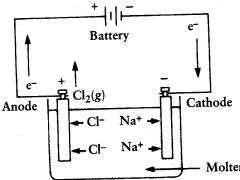
where does the liquid sodium end up?
|
|
|
anode. there they are oxid to Cl2
|
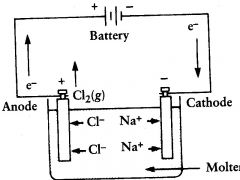
Cl- ions migrate towards the?
|
|
|
anode of an electr cell is considered to be ?
|
positive
|
|
|
why is the anode of an electrolytic cell considered pos?
|
its attach to posit pole of battery and attracts anions from the solut
|
|
|
in an electrolyt cell
Is it the anode or the cathode that attracts anions from the solution? |
anode
|
|
|
why is the anode of the galvanic cell considered negative?
|
bec the spont oxidat react that occurs there is the source of the cells negative charge.
|
|
|
oxidation occurs at the _____ in galvanic and electrolytic cells
|
anode
|
|
|
CAThode atrracts the CATions
so in a daniel cell, in the copper half cell, what moves towards the cathode? |
the copper II cations
|
|
|
CAThode attracts the CATions
in a daniel cell, in the __________ half cell, the copper II cations move towards the cathode |
copper
|
|
|
CAThode attracts the CATions
in electrophoresis, the ____________ charged Amino acids will migrate toward the cathode |
positively charged
|
|
|
CAThode attracts the CATions
electrophoresis negativ ch AA will move towards the? |
anode
|
|
|
electrophoresis
_________ move towar cathode _________ move toward the anode |
positiv ch aa - cathode
negat ch aa- anode |
|
|
sometimes when electrolysis is done in aq solut, __________ and not the solute might be oxid or reduc
|
water
|
|
|
in aq NaCl. if it is electrolyzed, H2O migth be reduced at the cathode to prod?
|
H2 gas and OH- ions
|
|
|
aq NaCl water be reduced to H2 and OH- instead of?
|
Na+ being reduced to Na (s)
|
|
|
What can you use to determine the species in a reaction that will be oxidized or reduced?
|
reduction potential
|
|
|
reduction potential is?
|
tendency of a species to acquire electrons and be reduced
|
|
|
reduction potential
the more positive the potential the greater? |
the species tendency to be reduced
|
|
|
reduct pot
the more ____________, the greater ___________ |
more posit the potent,
greater tend to be reduced |
|
|
reduct potential
___________ the reduct potent, ____________ tendency to be reduced |
more posit the reduct potent,
greater the tendency to be reduced |
|
|
reduct potential
what is the mnemonic? |
A positive potential means more likely to succeed.
|
|
|
a reduct potent is measured in?
|
volts (V)
|
|
|
a reduct pot is defined relative to?
|
a standard hydrogen electrode
(SHE) |
|
|
SHE is?
|
standard hydrogen electrode
|
|
|
SHE has a potent of?
|
0.00 volts
|
|
|
Standard reduction potential Eo is measured under?
|
standard condit
|
|
|
what are standard conditions?
|
25 C
1 M conc for each particip ion, part press of 1 ATM for each gas and metals in pure state |
|
|
higer Eo mean?
|
greater tendenc for reduct to occur
|
|
|
lower Eo means?
|
a greater tend for oxidat to occur
|
|
|
a ___________ Eo means a greater tend for reduct to occur
|
higher
|
|
|
Ag+ + e -> Ag (s) E° = +0.80 V
Tl+ + e- -> Tl(s) E°= -0.34 V which species will be oxid and which will be reduced? |
Ag+ will be red to Ag cause it has the higher Eo
|
|
|
to get the oxidat potential of a given half reaction you have to?
|
1.the reduct half react has to be reversed.
2. sign of reduct potent has to be reversed |
|
|
standard electromotive force is?
|
EMF.
the differ in potential betw the 2 half cells |
|
|
how do you figure out the EMF?
|
add the stand reduct pot of the reduc species and the stand oxidat potent of the oxidized species
|
|
|
t or f
when adding stand potent do not multiply by the number of moles oxid or reduc |
t
|
|
|
EMF= Eo red + ?
|
EMF= Eo red + Eo ox
|
|
|
a galvanic cell has a ___________ EMF
|
positive
|
|
|
an electrolytic cell has a ____________ EMF
|
negative
|
|
|
standard reduction potentials for Sm3+ and
[RhCI6]3- are -2.41 V and +0.44 V respectively, EMF of reaction: Sm3- + Rh + 6 Cl- -> [RhCI6]3- + Sm who is getting reduced? |
Sm3+ is getting reduced
|
|
|
standard reduction potentials for Sm3+ and
[RhCI6]3- are -2.41 V and +0.44 V respectively, EMF of reaction: Sm3- + Rh + 6 Cl- -> [RhCI6]3- + Sm who is getting oxidized? |
Rh is oxidized to Rh3+
|
|
|
standard reduction potentials for Sm3+ and
[RhCI6]3- are -2.41 V and +0.44 V respectively, EMF of reaction: Sm3- + Rh + 6 Cl- -> [RhCI6]3- + Sm whats the EMF? |
-2.41 V + -.44 V
=-2.85 |
|
|
standard reduction potentials for Sm3+ and
[RhCI6]3- are -2.41 V and +0.44 V respectively, EMF of reaction: Sm3- + Rh + 6 Cl- -> [RhCI6]3- + Sm if EMF is - 2.85, what kind of cell is this? |
electrolytic
|
|
|
standard reduction potentials for Sm3+ and
[RhCI6]3- are -2.41 V and +0.44 V respectively, EMF of reaction: Sm3- + Rh + 6 Cl- -> [RhCI6]3- + Sm if EMF is -2.85. in which direct will the react be spontaneous? |
to the left
|
|
|
a positive ch in G or Gibbs free energy is spont or non spont?
|
it is not spontant
|
|
|
a ch in G that is negat is spont or non spont?
|
spont
|
|
|
How do you determ the spontaneity of a reaction?
|
use ch in G
|
|
|
the maximum amount of useful work produced by a chemical reaction is
|
ch in G
or Gibbs free energy |
|
|
electrochem cell the work done is dependent on?
|
number of coulombs and the energy available
|
|
|
electrochem cell
ch in G= |
ch G= -nFEcell
|
|
|
-nFEcell=?
|
ch in G
|
|
|
∆G= -nFEcell
what is F |
faraday's constant
|
|
|
∆G= -nFEcell
in terms of units, what do you have to watch out for? |
that F and G are both expressed in J. You cant have one in J and the other in KJ
|
|
|
∆Go= -nFEocell
this means that the react? |
took place under standard conditions
|
|
|
∆G= -nFEcell
∆G is negative only if? |
EMF is positive
|
|
|
∆G is ________, when __________ is positive
|
negative,
EMF is positive |

