![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
31 Cards in this Set
- Front
- Back
|
1) Which of the following is an incorrect description of benzene?
A) The CCC bond angles are all equal to 120°. B) The molecule is planar. C) The molecule is a 6-membered ring which contains alternating single and double carbon-carbon bonds. D) The molecule is aromatic. E) The molecule can be drawn as a resonance hybrid of two Kekule structures. |
C) The molecule is a 6-membered ring which contains alternating single and double carbon-carbon bonds.
|
|
|
2) What is the bond order of the carbon-carbon bonds in benzene?
A) 0.5 B) 1 C) 1.5 D) 2 E) 3 |
C) 1.5
|
|
|
3) In the molecular orbital representation of benzene, how many π molecular orbitals are present?
A) 1 B) 2 C) 4 D) 6 E) 8 |
D) 6
|
|
|
4) How many distinct nodal planes which are perpendicular to the molecular plane are present in the π4* orbital of benzene?
A) 0 B) 1 C) 2 D) 4 E) 5 |
C) 2
|
|
|
5) How many pairs of degenerate π molecular orbitals are found in benzene?
A) 6 B) 5 C) 4 D) 3 E) 2 |
E) 2
|
|
|
6) Which of the following undergoes SN2 reaction with sodium methoxide most rapidly?
A) PhCH2Br B) Ph3CBr C) PhCH2CH2Br D) PhBr E) PhCH2CH2CH2Br |
A) PhCH2Br
|
|
|
7) Which of the following undergoes solvolysis in methanol most rapidly?
A) PhCH2Br B) Ph3CBr C) PhCH2CH2Br D) PhBr E) PhCH2CH2CH2Br |
B) Ph3CBr
|
|
|
8) Aromatic molecules contain __________ π electrons.
A) no B) 4n + 1 (with n an integer) C) 4n + 2 (with n an integer) D) 4n (with n an integer) E) unpaired |
C) 4n + 2 (with n an integer)
|
|
|
9) Which of the following is another name for cyclobutadiene?
A) [2]annulene B) [4]annulene C) [6]annulene D) Dewar benzene E) antibenzene |
B) [4]annulene
|
|
|
11) Which of the following is the same as the tropylium ion?
A) cycloheptatrienyl cation B) cycloheptatrienyl anion C) cyclopentadienyl cation D) cyclopentadienyl anion E) cyclopropenyl anion |
A) cycloheptatrienyl cation
|
|
|
16) Nitrogen's lone pair electrons occupy what type of orbital in pyridine?
A) s B) sp C) sp2 D) sp3 E) p |
C) sp2
|
|
|
17) Which of the following is not a fused-ring heterocycle?
A) purine B) pyrimidine C) benzofuran D) indole E) quinoline |
B) pyrimidine
|
|
|
18) How many distinct trichlorobenzene isomers are possible?
A) 2 B) 3 C) 4 D) 5 E) 6 |
B) 3
|
|
|
19) Which of the following is another accepted name for methyl phenyl ketone?
A) anisole B) cresol C) acetophenone D) benzophenone E) cresone |
C) acetophenone
|
|
|
20) Which of the following is also an acceptable name for 3-nitrophenol?
A) o-nitrophenol B) m-nitrophenol C) p-nitrophenol D) 3-cresol E) hydroquinone |
B) m-nitrophenol
|
|
|
21) Which of the following is also an acceptable name for 1,3,5-trimethylbenzene?
A) styrene B) mesitylene C) cumene D) cymene E) 1,3,5-xylene |
B) mesitylene
|
|
|
22) Which of the following compounds has the lowest boiling point?
A) 1,2,3-trichlorobenzene B) 1,2,4-tirchlorobenzene C) p-dichlorobenzene D) m-dichlorobenzene E) o-dichlorobenzene |
C) p-dichlorobenzene
|
|
|
23) Which of the following compound has the highest melting point?
A) benzene B) toluene C) o-dichlorobenzene D) m-dichlorobenzene E) p-dichlorobenzene |
E) p-dichlorobenzene
|
|
|
24) In the UV-visible spectra of the following compounds, in which does lambda max appear at the highest wavelength?
A) 3-phenylpropene B) 1-phenylpropene C) benzene D) n-octylbenzene E) isopropylbenzene |
B) 1-phenylpropene
|
|
|
25) In the proton NMR, in what region of the spectrum does one typically observe hydrogens bound to the aromatic ring?
A) 1.0-1.5 ppm B) 2.0-3.0 ppm C) 4.5-5.5 ppm D) 7.0-8.0 ppm E) 9.0-10.0 ppm |
D) 7.0-8.0 ppm
|
|
|
26) In the carbon NMR, in what region of the spectrum does one typically observe carbons which are part of the aromatic ring?
A) -10-0 ppm B) 40-60 ppm C) 80-100 ppm D) 120-150 ppm E) 200-220 ppm |
D) 120-150 ppm
|
|
|
27) Which of the following compounds has the most signals in the noise-decoupled 13C NMR spectrum?
A) o-dibromobenzene B) m-dibromobenzene C) p-dibromobenzene D) 1,3,5-tribromobenzene E) 1,2,3,4-tetrabromobenzene |
B) m-dibromobenzene
|
|
|
28) In the mass spectrum of isobutylbenzene, the base peak occurs at m/z:
A) 134 B) 133 C) 119 D) 105 E) 91 |
E) 91
|
|
|
29) How many peaks are in the proton spin decoupled 13C NMR spectrum of p-dichlorobenzene?
A) 2 B) 3 C) 4 D) 5 E) 6 |
A) 2
|
|
|
30) How many peaks are in the proton spin decoupled 13C NMR spectrum of 1,3,5-trinitrobenzene?
A) 1 B) 2 C) 3 D) 4 E) 6 |
B) 2
|
|
|
31) How many peaks are in the proton spin decoupled 13C NMR spectrum of 1,2,3-trichlorobenzene?
A) 1 B) 2 C) 3 D) 4 E) 6 |
D) 4
|
|
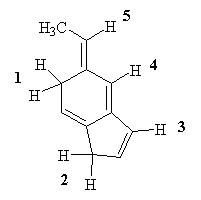
13) Which of the labeled H atoms (1 -5) in the following molecule would be predicted to be the most acidic?
A) 1 B) 2 C) 3 D) 4 E) 5 |
A) 1
|
|
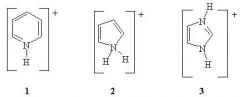
14) Rank the following in order of increasing pKa (from lowest to highest pKa)
A) 1 < 2 < 3 B) 3 < 2 < 1 C) 2 < 1 < 3 D) 3 < 1 < 2 E) 2 < 3 < 1 |
C) 2 < 1 < 3
|
|
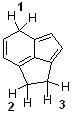
15) Which sequence ranks the indicated protons in order of increasing acidity?
A) 1 < 2 < 3 B) 2 < 3 < 1 C) 3 < 1 < 2 D) 3 < 2 < 1 |
D) 3 < 2 < 1
|
|
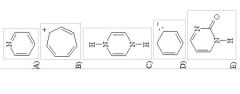
10) Which of the following structures, if flat, would be classified as antiaromatic?
|
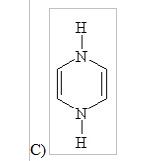
C
|
|
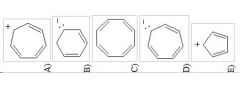
12) Which of the following structures is aromatic?
|
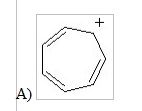
A
|

