![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
16 Cards in this Set
- Front
- Back
|
What do enzyme-catalysed reactions in vitro usually need extremes of?
|
Temperature
Pressure pH Concentration |
|
|
Do enzymes affect the free energy of a reaction?
|
No, they enable the reaction to overcome a kinetic barrier
|
|
|
Describe 3 ways in which enzymes promote a reaction to occur
|
Orientation - may hold two substrates together in the appropriate position for the reaction
Reactive group - enzyme binds with substrate transiently during reaction Proton donors - some enzymes act as proton donors |
|
|
Describe 2 ways in which enzymes are specific
|
Structure of substrates
Types of reaction they catalyse |
|
|
Name 5 different factors that may change the rate of an enzyme-catalysed reaction
|
Temperature
pH Concentration of substrate Concentration of enzyme Time course of reaction (conc. of product) |
|
|
How does temperature affect the rate of reaction?
|
Approx. x2 per 10 degree rise
until denaturing at too high temperatures |
|
|
How does pH affect an enzyme-controlled reaction?
|
Acts by protonating or deprotonating an ionisable group on the enzyme or the substrate
|
|
|
What is Vmax?
|
The enzyme's maximum rate
(enzyme becomes saturated with substrate) |
|
|
What is Km?
|
The Michaelis constant - the concentration at which the rate of the enzyme reaction is half Vmax
|
|
|
What are the units of Km?
|
Substrate concentration units
|
|
|
Why is it difficult to estimate Vmax and thus Km?
|
Vmax never reached
Hard to align a curve to a scatter of points Some enzymes inhibited by high [S], so readings at high [S] unreliable |
|
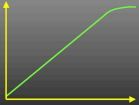
What does this curve represent?
|
Rate/[E]
|
|
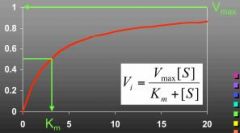
What does this curve represent?
|
Rate/[S]
|
|
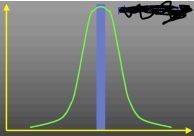
What does this curve represent?
|
Rate/pH
|
|
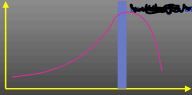
What does this curve represent?
|
Rate/temperature
|
|
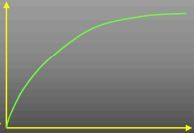
What does this curve represent?
|
Rate/time course of reaction ([P])
|

