![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
140 Cards in this Set
- Front
- Back
|
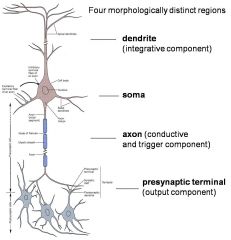
name the 4 morphologically distinct regions of the neuron
|
|
|
|
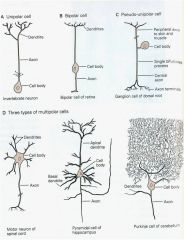
Neurons can be classified as _____,____,____
draw them out! |
|
|
|
is the metabolic center of the neuron cell
Contains the nucleus, endoplasmic reticulum |
soma
|
|
|
Main conducting unit for carrying signals to other neurons - excitable
Usually a single _____ per neuron |
axon
|
|
|
Initial segment of the axon= ______ is the location for generation of an action potential
Terminates in presynaptic terminal(s) – i.e. one axon can have more than one presynaptic terminal (en passant – Schaffer collaterals CA3-CA1) |
axon hillock
|
|
|
Principle of __________ signals within a neuron flow only in one direction from the dendrite to the soma to the axon if threshold is achieved
|
dynamic polarization
|
|
|
Principle of _________– neurons do not connect indiscriminately with one another
|
connection specificity
|
|
|
name the 3 types of glia?
|
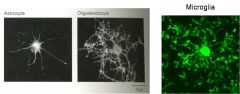
remember that oligodendrocytes are in the CNS, and Schwann cells are in the PNS
|
|
|
___________ form the blood brain barrier – an impermeable lining within the brains capillaries and venules which prevents toxic substances from entering the brain
|
astrocytes
|
|
|
astrocytes have long processes some of which terminate in ________ on neurons – postulated to provide nutrients to neurons
End feet within blood vessels which cause the endothelial cells to form tight junctions creating the protective |
end feet
blood brain barrier (BBB) |
|
|
which glia does this?
Maintain the potassium ion concentration in the extracellular space Absorb neurotransmitter Control brain vascular tone |
astrocytes
|
|
|
Schwann cells can envelop how many internodes of an axon?
|
one
the femoral nerve is approx 0.5m long, the internode distance is 1-1.5mm, thus approximately 300-500 internodes occur meaning up to 500 Schwann cells |
|
|
__________ is partly responsible for the prevention of axonal regrowth in the CNS
|
glial scarring
|
|
|
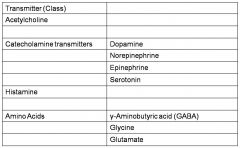
name the classes of neurotransmitters.
and the specific types in the classes |
|
|
|
Formed when 2 hemi-channels come together
Each hemi-channel represents the assembly of six _______ protein subunits – which are tetraspanning membrane proteins that share 3 conserved extracellular cysteine residues which are crucial for docking |
gap junctions
connexin |
|
|
t/f
gap junctions can be modulated by pH, Ca2+, or by second messenger systems activated by neurotransmitters. |
true
|
|
|
t/f
Low pH or elevated Ca2+ frequently open gap junction channels (conditions that exist when cells are damaged). |
false
Low pH or elevated Ca2+ frequently close gap junction channels (conditions that exist when cells are damaged). |
|
|
The core of the membrane fusion machinery is formed by a conserved set of vesicle membrane proteins— called ________—that assemble together into highly stable complexes.
|
SNAREs
|
|
|
Neuronal exocytosis is mediated by the vesicular v-SNARE,_________ and the target membrane t-SNAREs, ______and _______.
|
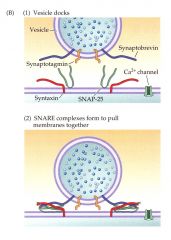
synaptobrevin
syntaxin SNAP-25 |
|
|
say the steps from ACTION POTENTIAL IN THE PRE-SYNAPTIC TERMINAL to neurotransmitter release in the dendritic spine
|
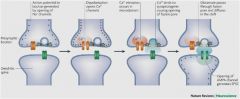
|
|
|
what's the 3 ways that a neurochemical transmission can be terminated
|
BREAKDOWN - Acetylcholine is degraded by acetylcholinesterase which sits in the neuromuscular cleft.
UPTAKE - Monoamines such as noradrenaline and serotonin are taken up by specific transporters (serotonin transporter, noradrenaline transporter) that are located in the plasma membrane of the presynaptic membrane DIFFUSION - Peptide transmitters such as substance P or enkephalin diffuse away and are degraded slowly by proteases in the exctracellular space |
|
|
t/f
ion channels open and close in response to specific electrical, mechanical, or chemical signals (gating) |
t
|
|
|
what are 2 classes of post-synaptic receptors
|
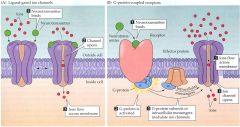
Ionotropic – the receptor is coupled to an ion channel. i.e. this is a chemically gated channel. Response to transmitter binding is fast (ms).
Metabotropic - Neurotransmitters can also activate receptors that are not directly linked to ion channels but rather activate second messenger machinery inside the post-synaptic neurons. Response is slow (up to min). Usually modulatory in nature |
|
|
what are the 2 types of summation?
|
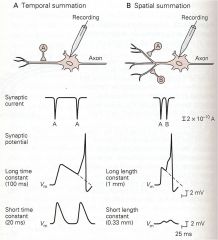
temporal + spatial
|
|
|
t/f
A neurotransmitter (one chemical compound) can have multiple effects depending on the receptor it binds. |
true
i.e. Glutamate can bind to AMPA, NMDA, and metabotropic receptors – these receptors as distinguished by more specific agonists |
|
|
Metabotropic receptors are coupled to 2nd messenger systems (GTP binding proteins) – the response is ____
|
slow
|
|
|
Ionotropic receptors contain both the NT recognition site as well as the ion channel – the response is ____
|
fast
|
|
|
In neuroscience, ___________is the ability of the connection, or synapse, between two neurons to change in strength. There are several underlying mechanisms that cooperate to achieve synaptic plasticity, including changes in the quantity of neurotransmitters released into a synapse and changes in how effectively cells respond to those neurotransmitters. Since memories are postulated to be represented by vastly interconnected networks of synapses in the brain, synaptic plasticity is one of the important neurochemical foundations of learning and memory
|
synaptic plasticity
|
|
|
The NMDA receptor (NMDAR) is a specific type of ionotropic glutamate receptor. NMDA (N-methyl D-aspartate) is the name of a selective agonist that binds to NMDA receptors but not to other glutamate receptors. Activation of NMDA receptors results in the opening of an ion channel that is nonselective to cations. This allows flow of Na+ and small amounts of Ca2+ ions into the cell and K+ out of the cell.
Calcium flux through NMDARs is thought to play a critical role in synaptic plasticity, a cellular mechanism for learning and memory. The NMDA receptor is distinct in that it is both ligand-gated and voltage-dependent. |
yea
|
|
|
t/f
equilibrium potential = reversal potential = Nernst potential. |
true
|
|
|
the transmembrane potential or the transmembrane potential gradient--- Simply, it is a property that 'belongs' to the membrane: the electrical potential difference (which is voltage) that exists across the membrane. However, the equilibrium potential you can think of conceptually as 'belonging' to a particular ____
|
ion
For a given ion, there is a certain membrane voltage at which the distribution across the membrane of a particular ion is in equilibrium. As you will recall, equilibrium is the state at which the NET flow of ions of zero, so, while actual movement is occurring, it is happening such that no measurable build-up of ion is occurring on either side of the membrane relative to the other. |
|
|
t/f
APs can be generated only at the nodes of Ranvier, where there are 'breaks' in the insulation. |
true
|
|
|
t/f
Decreasing nerve diameter would decrease internal resistance and therefore slow the conduction velocity. |
false
Decreasing nerve diameter would increase internal resistance and therefore slow the conduction velocity. |
|
|
name the TWO MAIN PARAMETERS THAT INCREASE CONDUCTION VELOCITY
|
1. increasing fiber size (increasing diameter of the nerve results in decreased internal resistance; thus conduction velocity along the nerve is faster). 2. myelination (as above, saltatory conduction can occur).
|
|
|
take me through the steps of the action potential
|
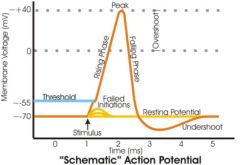
1) The cell is at resting membrane potential (rather near to the reversal potential for K+, but slightly more positive).
2) Depolarization occurs, causing rapid opening of the voltage-sensitive activation gates of the Na+ channel, and the Na+ conductance of the membrane promptly increases. The upstroke in membrane voltage occurs, caused by the inward Na+ current. At the peak, the voltage approaches that of the reversal potential for Na+, but, due to concurrent K+ flow (voltage-gated K+ channels open slightly more slowly than voltage-gated Na+ channels), the membrane potential does not reach the Na+ reversal potential voltage. The brief portion during which the AP is actually positive (and on its way to the reversal potential for sodium) is called the overshoot (not a label above). 3) The repolarization (heading back to the resting membrane potential), occurs because the DEpolarization eventually CLOSES the inactivation gates of the Na+ channel, and Na+ conductance again approaches zero (just like at rest). The voltage-gated K+ channels that lag the Na+ channels act to counterbalance the depolarization due to Na+ flow, and once the Na+ channels inactivate the K+ channels bring the membrane potential rapidly back down to normal resting values (REpolarization). However, the K+ conductance remains high for some time after closure of the Na+ channels (because the voltage-gated K+ channels also close slowly), so the membrane potential actually 'overshoots' the resting potential, becomes very negative (MORE negative than at rest) and gets really, really close to the reversal potential for K+. This is the hyperpolarizing afterpotential, or the 'undershoot' |
|
|
If AMPA-type glutamate receptors are activated then ___ will flow depolarizing the cell - producing an Excitatory Post-Synaptic Potential (EPSP).
If GABA receptors are activated,___will flow, which prevents action potentials and produces an Inhibitory Post-Synaptic Potential (IPSP). Note that action potentials are not always generated. Most EPSP remain subthreshold, so most intercellular communication does not result in a post-synaptic action potential. Typically, APs are generated when several converging inputs to a cell sum to bring its membrane potential to threshold (spatial summation) or when multiple sequential EPSPs from the same input sum to bring the post-synaptic potential above threshold (temporal summation). |
Na+
Cl- |
|
|
____ is primarily involved in the NMDA receptor - it acts as block on the channel. When a post synaptic terminal (where the NMDA channel sits) is depolarized, the ___ is dislodged. So the NMDA receptor requires two things to activate it: glutamate (it is a glutamate-activated channel) and post-synaptic depolarization (to release the ____ block).
|
Mg2+
|
|
|
the release of the vesicles from the cytoskeleton anchor was managed by a second-messenger system mediated via ____
|
synapsins
Current studies suggest the following hypothesis for the role of synapsin: synapsins bind synaptic vesicles to components of the cytoskeleton which prevents them from migrating to the presynaptic membrane and releasing transmitter. During an action potential, synapsins are phosphorylated by Ca2+/calmodulin-dependent protein kinase II, releasing the synaptic vesicles and allowing them to move to the membrane and release their neurotransmitter. |
|
|
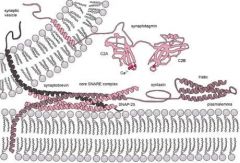
There are two groups of SNAREs:
v-SNARE: synaptobrevin/VAMP t- SNAREs: syntaxin, SNAP-25 (the classification of synaptotagmin is debatable sometimes it is lumped in with v-SNAREs) These complexes are responsible for allowing a vesicle to dock at the terminal presynaptic membrane. We already discussed that the release of the vesicles from the cytoskeleton anchor was managed by a second-messenger system mediated via synapsins. Once the vesicle can move to the active zone, SNAREs come into play. SNAP and NSF complex bind the SNARE complexes. Therefore, SNAP is NOT a SNARE (but SNAP-25 is a SNARE). This is rather unfortunate naming. |
|
|
|
time constant, tau = (Rm)(Cm) Rm is resistance across membrane and Cm is capacitance of membrane. * A long time constant means a post-synaptic potential will stick around for a long time, so it is likely to affect a subsequent potential. Two sequential EPSPs therefore have a good chance to _____
|
reach threshold
A short time constant means that a post-synaptic potential will dissipate quickly, so subsequent post-synaptic potentials are "on their own." As a result, it is less likely the cell will fire an action potential in response to two excitatory inputs that are separated in time by more than a couple of tau. |
|
|
length constant, lambda = root(Rm/Ra) Rm is the resistance across membrane and Ra is axial resistance (resistance due to cytoplasm). *
t/f A long length constant will permit a post-synaptic potential to be propagated a long distance without degradation; this allows for multiple stimulus inputs that come spatially far apart sites to sum together effectively at the soma. |
A neuron with a short length constant has a depolarization that degrades over short distances. As a result, a large post-synaptic potential at the dendrite is tiny by the time it reaches the cell soma. This means that inputs at distance dendrites (distal) affect the spiking of the cell much less than do inputs to close by dendrites (proximal) contribute.
|
|
|
what's the deal with quantal transmission?
|
For a given content of NT within a vesicle, a particular response at the postsynaptic cell will occur. The response increases incrementally with the addition of more vesicles, but in a predictable fashion given the ability of any vesicle to change the response in a particular way. In short, increases in the response occur ONLY by increasing the number of quanta (and these quanta are nothing more than vesicles) or increasing the content of the vesicles.
|
|
|
name 2 inhibitor NT's
|
glycine + GABA
|
|
|
name some excitatory NT's?
|
ACh, NE, Epi, glutamate and serotonin (5-HT)
|
|
|
t/f
dopamine is an excitatory NT |
f
Dopamine (DA) can be either an excitatory or inhibitory NT, depending on its post-synaptic receptor. While this is technically also true for glutamate, it acts as an excitatory NT the vast majority of the time. |
|
|
EPSPs are caused by the opening of channels permeable to ____ (eg. glutamate channels).
IPSPs are inputs that hyperpolarize the postsynaptic cell and therefore move it farther away from threshold and LESS able to fire an AP. These are caused by opening of channels permeable to ______ |
Na+
Cl- |
|
|
what are the 3 types of glutamate receptors?
|
There are 3 different types of glutamate receptors:
1. AMPA/Kainate (aka non-NMDA receptor) 2. NMDA (N-methyl-D-aspartate) receptor 3. Metabotrophic glutamate receptor The non-NMDA receptor is the most common glutamate receptor and considered the fast receptor channel; it functions largely in the way in which you might expect. Metabotrophic receptors are linked to 2nd messenger cascades via G-proteins and not to ion channels directly (although they can indirectly affect ion channels through G-protein second-messenger systems). The NMDA receptor is different and should be considered more closely. If non-NMDA receptor is the fast receptor channel, NMDA receptor is the slow-receptor channel. It also does not function on an all-or-nothing idea merely requiring substrate binding and opening of the channel to ions. Instead, when glutamate binds, it is 'not enough' and the receptors ALSO require sufficient depolarization. Near the resting potential (-65mV), the channels are blocked by Mg2+ present in the extracellular space. The channels are thus non-conductive even though they have bound agonist (glutamate). As the cell is depolarized (this occurs via the activation of OTHER excitatory receptors), the voltage change forces magnesium out of the NMDA receptor channel and it then becomes conductive to Na+, K+ and Ca2+ (that is, it is now permeable to those ions). |
|
|
NMDA receptor channels require what 2 events?
|
BOTH ligand-binding (glutamate) and voltage change (via OTHER channels, and therefore the Mg2+ is removed and the receptor can undergo a conformational change) to open effectively. The requirement for both glutamate binding (glutamate in the cleft means the presynaptic neuron is active) and membrane depolarization (meaning the post-synaptic neuron is also active) is central to how memories are formed. Essential, the NMDA channel is the coincidence detector (Hebb's rule).
|
|
|
Acetylcholine (ACh) is degraded via ________ which is located in the neuromuscular cleft itself.
|
acetylcholinesterase
So, as soon as ACh is released from the presynaptic cell, and while it is diffusing over to that post-synaptic cell, it is being degraded. A disease called myasthenia gravis (MG) is an autoimmune disorder in which the post-synaptic ACh receptors are attacked by the body itself and destroyed. These patients therefore do not get a strong signal from the presynaptic cell, not because anything is wrong on the presynaptic side, but because as the ACh diffuses over to the post-synaptic cell, it does not encounter as many receptors and that post-cell does NOT get a normal signal. The patient is very weak, especially after periods of activity. For instance, a hallmark of the disease is lid-lag; after a day of having their eyelids elevated in order to see/look around, the muscle in the eyelid do not receive the appropriate signal and become weak, and the patient's eyelids will droop (called ptosis). Why is this disease relevant to this discussion? In MG, the treatment of choice is to use an anti-acetylcholinesterase (or AChesterase inhibitor), such as neostigmine. Such a drug will block the AChesterase from degrading ACh. Then, there will be more ACh around in the cleft and you increase the likelihood of signal transmission to the post-synaptic cell. So while this does NOT directly salvage the post-synaptic ACh receptors (attempts to achieve this are immunosuppressant therapies), it does keep ACh around and alleviate some of the symptomatology. |
|
|
choose the method of NT action termination:
1. breakdown of NT 2. Uptake of NT 3. Diffusion of NT away from cleft and eventual depredation Peptide transmitters, such as enkephalin and substance P |
FIRST diffuse away from the cleft, and are then degraded by proteases located in the extracellular space.
|
|
|
choose the method of NT action termination:
1. breakdown of NT 2. Uptake of NT 3. Diffusion of NT away from cleft and eventual degredation Monoamines, such as NE, serotonin, epinephrine |
are taken up by specific transporters located in the plasma membrane of the PREsynaptic cell. The monoamines are then degraded in the cytoplasm of the presynaptic cell. This process is called, in the medical literature and discourse, 'reuptake'. Have you heard of SSRIs? Or, perhaps you have a friend/relative taking or heard a television commercial for Prozac, Zoloft, Paxil or Celexa? These are SSRIs SSRI stands for Selective Serotonin Reuptake Inhibitor. They do what their name implies: they block the reuptake of Serotonin (5-HT) selectively (only serotonin, not the other monoamines) at the level of the presynaptic cell.
It is believed that low amounts of the monoamines can contribute to various mood disorders (depression). So, if there is a way to 'keep serotonin around' in the synaptic cleft, then the effect of the neurotransmitter should be increased, since there is, put simply, more of it around. Of note, reuptake does NOT occur at the level of the post-synaptic cell. |
|
|
is an anesthetic agent. It is used to paralyze the patient undergoing anesthesia and surgery. It blocks ACh receptors on the motor end plate, causing a prolonged depolarization and therefore paralysis. It is a DEPOLARIZING agent,
|
succinylcholine
|
|
|
is a topical anesthetic agent that works by blocking the fast transient sodium channels, thereby preventing depolarization, the AP, and subsequent signaling. Put simply, it blocks nerves so you don't feel pain.
|
lidocaine
Tetrodotoxin too |
|
|
is the toxin produced by the bacteria Clostridium botulinum. It produces total blockade of ACh release from the presynaptic cell.
|
Botulinus toxin, or Botox
|
|
|
works in much the same way at Botox, and is a toxin created by Clostridium tetani. It produces total blockade of glycine and GABA release from the presynaptic cell.
|
tetanus toxin
|
|
|
a drug used in the treatment of Myasthenia gravis; works to increase the amount of ACh in the cleft by BLOCKING the enzyme that would degrade this ACh; it is therefore an anti-cholinesterase.
|
neostigmine
|
|
|
causes IRreversible blockade of nicotinic ACh receptors. On the other hand, _____ causes reversible blockade of nicotinic ACh receptors.
|
alpha-bungratoxin
curare |
|
|
are illegal substances that work as NMDA receptor antagonists
|
PCP (phencyclidine) and Ketamine
|
|
|
what inhibits the machinery critical to maintaining the membrane potential, and, therefore, absolutely necessary for an action potential and any signaling to occur: the Na/K pump (Na/K ATPase)?
|
digitalis or ouabain.
|
|
|
what's the babinksi sign? what is it a sign of?
|
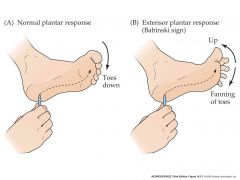
upper motor neuron syndrom
|
|
|
name all the descending motor tracts originating in CORTEX?
|
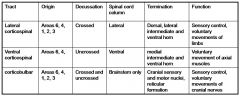
|
|
|
t/f
the ventral/anterior corticospinal tract decussates |
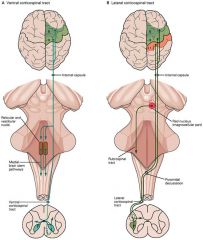
false
|
|
|
t/f
the corticobulbar tract descends to the spinal cord |
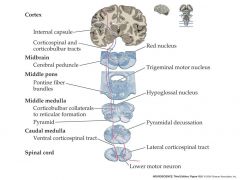
f
stays in brainstem |
|
|
UMN or LMN lesion:
facial weakness on one side of the face? |
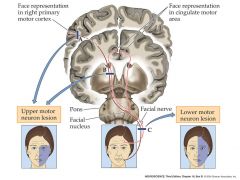
LMN
|
|
|
LMN or UMN lesion:
weakness of bottom left side of face? which side is lesion on? |
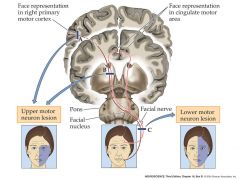
right UMN
|
|
|
name all the descending motor pathways originating in BRAINSTEM?
(6) |
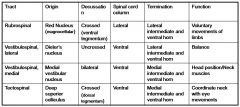
also, reticulospinal (pontine), and reticulospinal (medullary)
|
|
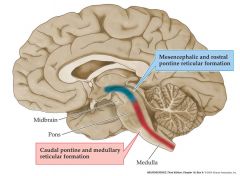
what's the function of the reticulospinal pathway?
|
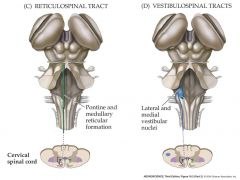
automatic movements axial and limb muscles
|
|
|
what's the difference between automatic and voluntary movement?
|
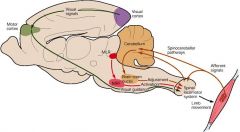
find out more
|
|
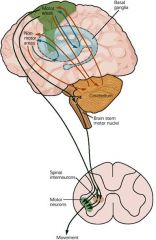
|
motor systems
|
|
|
how do you characterize the topography of weakness for CNS, PNS?
|
CNS- hemiparesis (one-side)(cortex to spinal cord), paraparesis with sensory level (spinal cord) etc.
PNS- distal, proximal, or Sxs following a specific nerve root or peripheral nerve distribution |
|
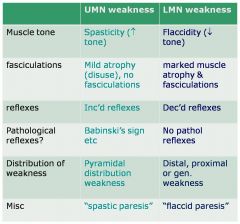
How do UMN and LMN weakness differ?
Fill in this chart!! |
|
|
|
what's fasciculation?:
|
involuntary twitching of muscle fibers innervated by the same lower motor neuron.
|
|
|
what's muscle tone? how can we characterize muscle tone?
|
Muscle tone: resistance to passive movement (e.g., the examiner moves the leg or arm back and forth with the pt relaxed).
Normal, spastic (clasp-knife phenomenon), rigid, flaccid |
|
|
what's the clasp-knife response?
|
Upper motor neuron lesions are characterized by spasticity. Spasticity is increased muscle tone leading to resistance of the limbs to passive manipulation. This spasticity classically results in the clasp-knife response. The clasp-knife response is when the spastic limb is passively moved with great resistance, when suddenly the limb "gives", becoming very easy to move. The clasp knife response is most prominent in the muscle groups least affected by the upper motor lesion, e.g., flexors in the upper extremities or extensors in the lower extremities.
|
|
|
what's pyramidal distribution weakness?
characteristic of what kind of lesion (UMN or LMN) |

UMN
Extensors are weaker than flexors in UE Flexors are weaker than extensors in LE Distinction between weakness of central and peripheral origin by means of the distribution of that weakness is a possible means of differentiation, although not as reliable as muscle tone, reflexes, and fasciculations. Central lesions are thought to affect extensors more than flexors in the upper limb and flexors more than extensors in the lower limb. Such a classic distribution of a central lesion is often referred to as a "pyramidal" pattern. seems like this guy is doing what's easier for him--flexing UE and extending LE |
|
|
what's paresis? what are the types?
|
Paresis is a condition typified by partial loss of movement, or impaired movement
paresis: for weakness plegia:paralysis * Monoparesis -- One leg or one arm * Paraparesis -- Both legs * Hemiparesis -- One arm and one leg on either side of the body * Tetraparesis -- All four limbs |
|
|
what's proximal vs distal weakness?
|

distal weakness?
Footdrop, weakness of hand grip, cannot open jars Proximal weakness? - difficulty climbing chairs, reaching for objects high up **footdrop is shown |
|
|
tell me about the stretch reflex (Ia reflex): i.e. Patellar reflex
|
stretch reflex (Ia reflex): i.e. Patellar reflex
-muscle spindles in quads---- Ia afferents to DRG----- a. homonymous motoneurons–-- quads contract b. Ia inhibitory interneuron---antagonist motoneuron--- hamstrings relax |
|
|
Inverse stretch reflex (Ib reflex):
|
contraction of quads– activate Golgi tendon organ---Ib afferents----Ib interneurons----inhibit homo motoneurons ---inhibits quads and activate hamstrings
|
|
|
A 74 y/o woman woke up with a facial droop and weakness of her R side. Exam showed that she had paralysis of her R arm & leg, and a left facial droop resulting in weak eye closure, unable to raise her L eyebrow, and asymmetric smile. She also noted double vision when looking to the L. Tone and DTRs were inc’d on the right, with (+) Babinski sign.
Q1. Is this UMN or LMN weakness? Q2. Is this likely to be brain or spinal cord? Q3. Are there associated symptoms or signs to help us localize further? |
Q1. Is this UMN or LMN weakness?
UMN Q2. Is this likely to be brain or spinal cord? hemiparesis/hemiplegia: brain > spinal cord Paraparesis/quadriparesis: spinal cord or lower brainstem > brain Q3. Are there associated symptoms or signs to help us localize further? L peripheral facial weakness, L 6th nerve findings: L pons Diagnosis was: left pontine infarct |
|
|
A 54 y/o male has been having weakness and tingling of both feet for 1 yr. Then 2 mo ago, his legs gave out while walking. He also started to have hand weakness”
Exam: weakness of both legs and hand muscles; sensory loss in his feet; tone was dec’d; absent DTRs at the ankles; negative Babinski. Q1. Is this UMN or LMN weakness? Q2. Which part of the PNS is affected? Q3. Are there associated symptoms or signs to help us localize further? |
Q1. Is this UMN or LMN weakness?
LMN Q2. Which part of the PNS is affected? Pure motor: anywhere from alpha motoneurons to nerve, NMJ or muscle Motor + sensory: nerve roots or peripheral nerves Q3. Are there associated symptoms or signs to help us localize further? no |
|
|
A 40 y/o female with 2 yr history of progressive hand and feet weakness, preceded by muscle spasm and cramps involving her arms. She complains of difficulty buttoning clothes and cutting food. She also notices involuntary muscle twitching, and more recently difficulty with articulation.
Exam: CN: normal, except for tongue fasciculations. Motor: fasciculations in the arms, increased tone in the lower extremities, atrophy of hand muscles. Deltoids, biceps, triceps (4/5), hand muscles (3/5). The rest of the muscles were normal. Sensory: normal Reflexes: increased at the biceps, triceps, patella (3), and ankles (4) with clonus. Plantar responses were downgoing. Gait: spastic |
Q1. Is this UMN or LMN weakness?
Both Q2. Is this likely to be brain or spinal cord? both Q3. Are there associated symptoms or signs to help us localize further? lack of sensory findings diagnosis: ALS Amyotrophic Lateral Sclerosis - a form of motor neurone disease is a progressive, fatal, neurodegenerative disease caused by the degeneration of motor neurons, the nerve cells in the central nervous system that control voluntary muscle movement. The disorder causes muscle weakness and atrophy throughout the body as both the upper and lower motor neurons degenerate, ceasing to send messages to muscles. Unable to function, the muscles gradually weaken, develop fasciculations (twitches) because of denervation, and eventually atrophy because of that denervation |
|
|
tests to help localize UMN, LMN lesions?
|
Tests to help localize UMN lesions: brain MRI /MRA; spine MRI, myelogram; evoked potentials
Tests to help localize LMN lesions: Nerve conduction test/electromyography Nerve & muscle biopsy |
|
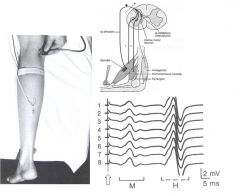
what's the H-reflex?
|
H-reflex is analogous to the mechanically induced spinal stretch reflex (for example, knee jerk reflex) because in both cases muscle-spindle innervating fibers are activated. Although stretch reflex gives just qualitative information about muscle spindles and relfex arch activity; if the purpose of the test to compare performances from different subjects, H-reflex should be used. In that case, in fact, latencies (ms) and amplitudes (mV) of H-wave can be compared
The H-reflex is a reflectory reaction of muscles after electrical stimulation of sensory fibers (Ia afferents stemming from muscle spindles) in their innervating nerves (for example, those located behind the knee). The H-reflex test is performed using an electric stimulator, which gives usually a square-wave current of short duration and small amplitude (higher stimulations might involve alpha fibers, causing a M-wave, compromising the results), and an EMG set, to record the muscle response. That response is usually a clear wave, called H-wave, 50 ms after the stimulus, not to be confused with M-wave at 25-30 ms, which might appear in too intense stimulations. |
|
|
what's electromyography? (EMG)
|

There are two kinds of EMG in widespread use: surface EMG and needle (intramuscular) EMG. To perform intramuscular EMG, a needle electrode is inserted through the skin into the muscle tissue. the electrical activity is observed while inserting the electrode. The insertional activity provides valuable information about the state of the muscle and its innervating nerve. Normal muscles at rest make certain, normal electrical sounds when the needle is inserted into them. Then the electrical activity when the muscle is at rest is studied. Abnormal spontaneous activity might indicate some nerve and/or muscle damage. Then the patient is asked to contract the muscle smoothly. The shape, size and frequency of the resulting motor unit potentials is judged. Then the electrode is retracted a few millimeters, and again the activity is analyzed until at least 10-20 units have been collected. Each electrode track gives only a very local picture of the activity of the whole muscle.
A motor unit is defined as one motor neuron and all of the muscle fibers it innervates. When a motor unit fires, the impulse (called an action potential) is carried down the motor neuron to the muscle. The area where the nerve contacts the muscle is called the neuromuscular junction, or the motor end plate. After the action potential is transmitted across the neuromuscular junction, an action potential is elicited in all of the innervated muscle fibers of that particular motor unit. The sum of all this electrical activity is known as a motor unit action potential (MUAP). This electrophysiologic activity from multiple motor units is the signal typically evaluated during an EMG. The composition of the motor unit, the number of muscle fibres per motor unit, the metabolic type of muscle fibres and many other factors affect the shape of the motor unit potentials in the myogram. |
|
|
The H-reflex is evoked by _______ of the afferent nerve, rather than a mechanical
stretch of the muscle spindle, that results in monosynaptic excitation of alpha-motoneurons. Hence, the H-reflex bypasses the muscle spindle and the fusimotor activity that may influence the sensitivity of the Ia afferents to engage a ‘simple’ reflex circuit. |
electrical stimulation
|
|
|
The H-reflex and M-wave do not recruit the same alpha-
motoneurons. Alpha motoneurons are recruited in an orderly fashion from ______ (more excitable with large Ia excitatory postsynaptic potentials; EPSP) to _____ (less excitable smaller EPSP) Thus, smallmotoneurons innervating slow motor units are recruited first in the H-reflex, while electrical stimulation that elicits the M-wave activates larger diameter axons that innervate fast motor units. |
smallest
largest |
|
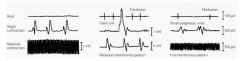
label each of the pix with:
--normal muscle --denervated muscle --myopathy |
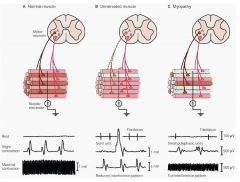
|
|
|
what's a motor unit?
|
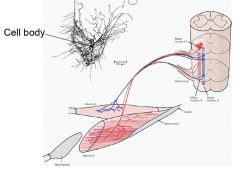
A motor neuron and all muscle fibers that it innervates
a motor unit is the quantum of the motor system, that is, the smallest reducible unit that still functions/can be engaged. It is made of a single alpha-motor neuron (be it a slow MN, FR MN or a FF MN) and all the muscle fibers that the single motor neuron innervates. |
|
|
Total force produced by a muscle is a function of ____ and ____
|
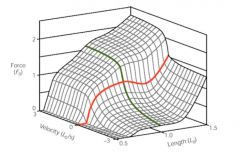
Total force is a function of length and velocity
|
|
|
name the 3 types of muscle fibers
|
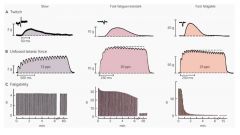
slow
fast-fatigable fast fatigue-resistant |
|
|
t/f most muscles have one type of muscle fibers
|
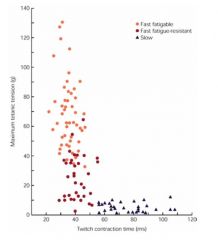
f Most muscles have all three kinds of motor units
|
|
|
Upper motor neurons which innervate the muscles of the face and head are located near the lateral fissure of the brain. Their axons coalesce to form the corticobulbar tract. These axons then descend within the Genu of the internal capsule to the medial part of the cerebral peduncle. The upper motor neuron axons then synapse on ________ of the cranial nerve nuclei which are located in midbrain, pons and medulla.
|
lower motor neurons
|
|
|
what are four main categories of movement?
|
1. Passive: occurs without any neural input and produced by an external force moving a part of the body. If the external force encounters resistance to such movement, it is either rigidity (lesion of basal ganglia) or spasticity (lesion of CST).
2. Reflexive: involuntary movement and include movements to withdrawal from painful stimulus. Involve a 'reflex arc'; certain reflexes exist at different periods in life or as indicative of underlying pathology. 3. semi-automatic movement: this is stereotyped and include rhythmic behavior such as walking or chewing, but also non-rhythmic ones like vomiting and micturition. These movements are under control of central pattern generators, whose circuitry can be found in the brainstem and spinal cord. 4. Self-generated movement: a heterogeneous group of movements, such as emotional and voluntary movements to consciously directed movements (willful and purposeful). |
|
|
take me through the steps of catching a ball?
|
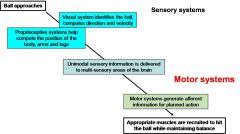
|
|
|
Motor functions are performed by:
Primary motor cortex: Brodmann’s area __ Premotor cortex: Brodmann’s area __ Somatosensory cortex: Brodmann’s area __, __, and ___ |
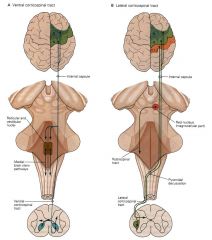
4
6 3,2,1 |
|
|
which tracts are part of the medial descending pathway (in brainstem)?
lateral descending pathway (in brainstem? |
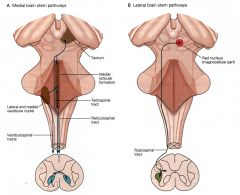
medial descending pathway:
VESTIBULOSPINAL TECTOSPINAL RETICULOSPINAL Provides input to interneurons in the ventral medial part of the spinal cord. These interneurons in turn control motor neurons in the ventromedial motor nucleus. lateral: RUBROSPINAL tract Provides input to interneurons in the dorsal lateral part of the ventral spinal cord. These interneurons in turn control motor neurons in the dorsolateral motor nucleus. |
|
|
the motor neurons coming from the
Dorsolateral motor nucleus go to ____ Ventromedial motor nucleus go to ____ |
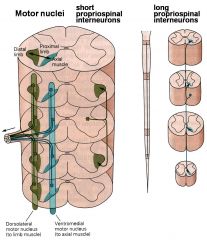
leg and arm muscles
to axial muscles |
|
|
what's part of the PNS? the CNS?
|
|
|
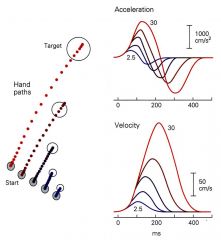
what' significant about this?
|
the brain plans a higher acceleration when you have to reach a longer distance
Extent of movement is planned before movement is initiated The representation of the plan for movement: MOTOR PROGRAM |
|
|
What's motor equivalence?
|

In the brain, movement is represented in an abstract form rather than as a series of joint motions or muscle contractions
(there are many ways to do the same task...The brain represents the outcome of motor actions independently of the specific effector used or the specific way the action is achieved.) |
|
|
what's a reflex?
|
Automatic, stereotyped movement in response to stimulation of peripheral receptors.
|
|
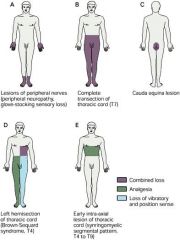
|
lesions
|
|
|
what causes an abnormal reflex?
|
|
|
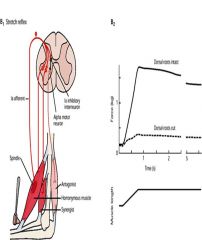
|
Spinal reflexes involve coordinated contraction of numerous muscles in the limbs
|
|
|
what's the crossed-extension reflex?
|
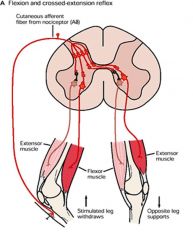
this is the reflex that allows you to pull your hand away from a burning stove.
contralateral flexors are relaxed and extensors contracted....it's said that this would help with balance/posture for the person pulling their hand away |
|
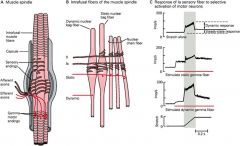
what are the 2 types of intrafusal fibers in muscle spindles?
|
nuclear bag fibers
nuclear chain fibers both innervated by Ia and II afferents both innervated by gamma-motoneurons nuclear bag fibers (detect the rate of change in muscle length--fast, dynamic changes) nuclear chain fibers (detect static changes in muscle length) |
|
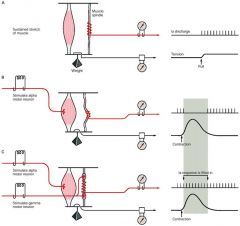
what does this show?
|
gamma-motoneurons:
innervate intrafusal muscle fibers adjust sensitivity of the muscle spindle (so it will respond appropiately during muscle contraction) ***alpha-motoneurons and gamma-motoneurons are coactivated so that muscle spindles remain sensitive to changes in muscle lenght during contraction |
|
|
what' convergence and divergence?
|
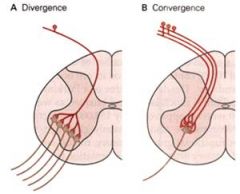
|
|
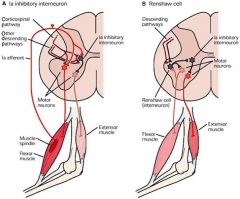
what's recurrent inhibition?
|
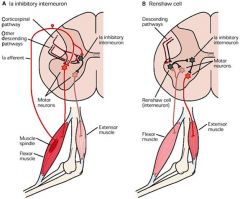
(renshaw cells)
-renshaw cells are inhibitory cells in the ventral horn of the spinal cord -they receive input from collateral axons of motoneurons and, when stimulated, negatively feedback (inhibit) on the motoneurons |
|
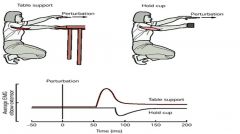
what does this say?
|
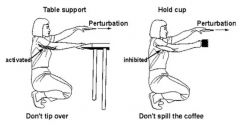
It is important to understand that reflexes are adaptable and control movements in a
purposeful manner. For example, a perturbation (a disturbance of motion, course, arrangement, or state of equilibrium) of the left arm can cause contraction of the opposite elbow extensor in one situation (opposite arm used to prevent the body from being pulled forward) but not the other, where the opposite arm holds a cup and the perturbation causes an inhibition of opposite elbow extensor. Also, don’t forget that spinal reflexes are functionally efficient because they enable adjustments of movements to be made at the level of the spinal cord, without having to involve (and wait for) decisions from higher levels of the motor systems. |
|
|
what's the golgi tendon organ?
|
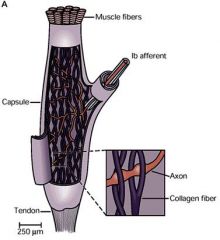
are arranged in series with extrafusal muscle fibers and detect muscle tension
(afferent fibers of golgi tendon organs are Ib) The Golgi organ (also called Golgi tendon organ, neurotendinous organ or neurotendinous spindle), is a proprioceptive sensory receptor organ that is located at the insertion of skeletal muscle fibers into the tendons of skeletal muscle. The body of the organ is made up of strands of collagen that are connected at one end to the muscle fibers and at the other merge into the tendon proper. Each tendon organ is innervated by a single afferent type Ib sensory fiber that branches and terminates as spiral endings around the collagen strands. |
|
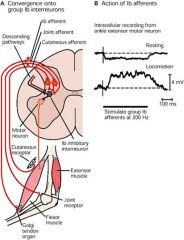
what does this say?
|
Action of Ib afferents changes with behavior
|
|
|
what's the long-loop reflex pathway?
|
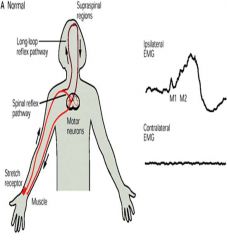
A stretch reflex with a latency of 50-80 ms, modified by instructions mediated by a higher centre of the brain.
|
|
|
klippel-feil syndrome
|
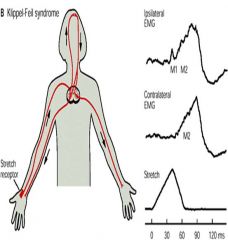
|
|
|
what are microglia?
what are they derived from? |
1 of 3 types of glia (along with oligodendrocytes/schwann cells and astrocytes) Are phagocytes that mobilize after injury infection or disease onset
Physiologically and embryologically unrelated the other cells in the nervous system mesoderm |
|
|
cool
|
|
|
2 forms of intercellular communication?
|
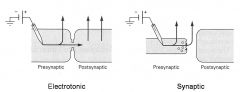
|
|
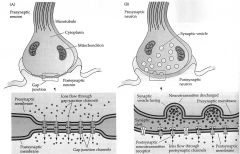
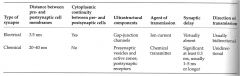
what's the difference between electrical and chemical synapses?
|
|
|
|
Vesicle release is ___ dependent
|
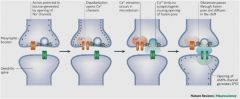
Ca2+
|
|
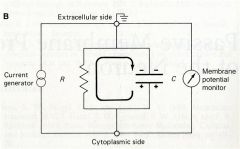
analogy with cell:
what would be the capacitor? the current generator? |
The membrane = capacitor (separation of charge and storage of charge)
The Na+/K+ pump is the current generator in this example |
|
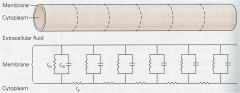
the speed of the action potential is determined by ___, ___, and ____
|
Ra, Cm, Rm
***Speed of conduction is inversely related to the product of Cm*Ra Cm: The larger the capacitance the more charge must be supplied to change voltage V=Q/C C is inversely proportional to the thickness of the insulating material (myelin) so rate = 1/capacitance= insulating material (myelin) so the more myelin, the higher the speed of conduction! |
|
|
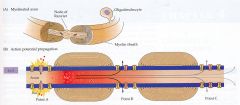
what's saltatory conduction?
|
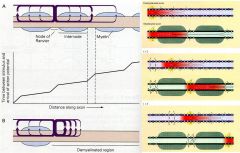
|
|
|
what's the most common neurological disability in young adults
|
MS (multiple sclerosis)
Called multiple because patients have multiple attacks separated by time and space Abrupt onsets of neurological deficits that persist for days or weeks and then remit/recover. Particularly common are monocular blindness (lesions in optic nerve), motor weakness or paralysis (lesions in corticospinal tracts), abnormal somatic sensations (lesions of somatic sensory pathways), double vision (lesions of medial longitudinal fasciculus) and dizziness (lesions of vestibular pathways). Eventually there is a cumulative acquisition of neurological defects which do not remit Cause is unknown but MS attacks myelinated axons – denuding them of myelin or causing them to degenerate |
|
|
To be recognized by a protein an ion must first shed at least part of its ______
For an ion to bind favorably the free energy cost for dehydration must be compensated by interactions gained at the binding site – snug fit mechanism Ion selectivity is determined in part by Coulomb’s Law – “The magnitude of the electrostatic force between two point electric charges is directly proportional to the product of the magnitudes of each charge and inversely proportional to the square of the distance between the charges.” |
hydration shell
|
|
|
what's the Nernst eqn?
|

|
|
|
At rest permeability for___ is very high so resting membrane potential is near the equilibrium potential of ___
|
K+
|
|

|

a motor unit is the quantum of the motor system, that is, the smallest reducible unit that still functions/can be engaged. It is made of a single alpha-motor neuron (be it a slow MN, FR MN or a FF MN) and all the muscle fibers that the single motor neuron innervates.
|
|
|
the somatotopy of the internal capsule: Remember, it can be recalled by saying 'FATL' (face-arm-trunk-leg), and this organization is largely preserved throughout the motor system. area
anterior limb of IC; mostly ascending connections from thalamus, not relevant for this question. genu of IC; where the____ is represented, so this is corticobulbar fibers. posterior limb of IC; where ___is represented, so ant and lat CST fibers. The patient has a lesion in area 2, given the isolation of facial weakness on history/physical. |
'F' (face)
'ATL' |
|
|
ion channels have 2 or more stable conformational states
_____ is the transition between these two states and involves a conformational change Such a change requires energy – voltage, chemical, mechanical Most channels transition from the open to an inactive state before they can open again – refractory |
gating
|
|
|
After a neuron fires, there is an absolute refractory period for the voltage gated sodium channels during which it is impossible to trigger another action potential. This period is ~ 1ms. Thus the maximum firing rate of a neuron is ~ _____ action potentials per second.
|
1000
|
|
|
cm is inversely proportional to thickness of myelin (more myelin=smaller cm)
Speed of conduction is inversely related to ra and cm (small cm means faster/slower conduction) |
faster
|
|
|
steps of synaptic transmission
|
Action potential travels down axon of pre-synaptic neuron
When the action potential reaches the pre-synaptic terminal and depolarizes it, calcium channels open Calcium enters the pre-synaptic terminal and binds to calcium receptors on vesicle fusion protein (synaptotagmin) Vesicles containing neurotransmitter fuse with the pre-synaptic membrane and release their contents into the synaptic cleft Neurotransmitters diffuse across the synaptic cleft and bind to receptors on the post-synaptic membrane The binding of neurotransmitter activates the receptor (different receptors have different effects on the post-synaptic neuron) -Note that the post-synaptic receptors, NOT the neurotransmitter, determine the post-synaptic response |
|
|
Summation of post-synaptic potentials (PSPs) occurs in the dendrites and soma.
Excitatory inputs (EPSPs) are typical on dendritic spines while inhibitory inputs (IPSPs) usually occur on the _____or ____ |
dendritic shaft or soma
|
|
|
Interneurons:
(a) Classification based on their axon projections ____ interneurons: axons in the same or adjacent spinal segment ______ neurons: axon project to distant spinal segments ______ neurons: axons ascend to higher brain centers |
Local
Propriospinal projection |
|
|
Motor neurons to small force units are ____ too
|
small
|
|
|
Origin of anterior corticospinal tract:
The other cortico-tracts (lateral CS + CB) = |
6, 4
6, 4 and 1, 2, 3, |
|

fill this out!!
|
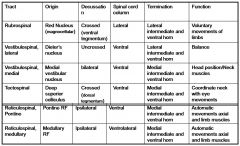
|
|
|
difference bw REM and non-REM sleep?
|

REM sleep
“Dreaming and paralyzed” – EEG, eye movements, voltage are reminiscent of wakefulness BUT: low muscle tone Non-REM sleep Think: “stereotypical sleep” |
|
|
what are the 2 mechanisms regulating sleep?
|
Homeostatic
Based on when you slept last and how much The longer since you slept and the less you slept, the stronger the signal to encourage sleep Seeks balance of sleep and wakefulness Circadian Based on a regular, daily rhythm Rhythm maintained by suprachiasmatic nucleus in hypothalamus Influenced by external cues |

