![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
129 Cards in this Set
- Front
- Back
|
Atoms
|
Atoms can combine to form molecules
in which two or more atoms are joined together |
|
|
Classifications of matter
|
solid, liquid or gas
element, compound, or mixture. |
|
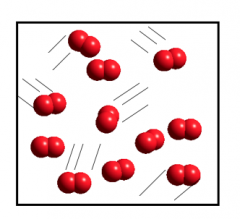
Gas
|
Gas: Has no fixed volume or shape. Expands to fill its container.
|
|
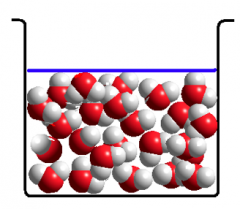
Liquid
|
Liquid. Has a fixed volume but no fixed shape – takes on the shape of that part of
water molecules in liquid water are loosely associated |
|
|
Solid
|
Solid. Has a definite shape and a definite volume, e.g. diamond
|
|
|
Pure Substances
|
- Most forms of matter are not chemically pure, e.g. air is a mixture.
- Distinct properties and composition that do not vary from sample to sample. - Water and table salt are pure substances. - All pure substances are either elements or compounds. |
|
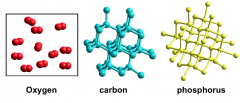
ELEMENTS
|
- Cannot be decomposed into simpler substances.
- Like Lego models. - Elements contain only one kind of atom. Some elements are: |
|
|
Compounds
|
Compounds are substances composed of two or more elements.
-Sodium chloride (table salt) - butanol (brake fluid) - octanol (gasoline) - glucose (sugar) |
|
|
Mixtures
|
Mixtures are combinations of two or more substances in which each substance retains its chemical identity.
|
|
|
Heterogeneous mixtures
|
One can see that these differ from point to point in the mixture.
Heterogeneous mixture is seen under microscope to be composed of several different types of materials - Granite - vinaigrette - Things like wood or rocks or sand. |
|
|
Homogeneous mixtures
|
Homogeneous mixtures are also called solutions.
- Air is a homogeneous mixture of oxygen and nitrogen - A solution of NaCl in water is a homogeneous mixture - Brass an alloy - Coffee |
|
|
Compounds
|
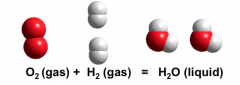
Most elements can interact with other elements to form compounds
|
|
|
Law of constant composition or constant proportions
|
The elemental composition of a compound is always the same. e.g. water is always 11% H and 89% O. ‘Natural” water?
|
|
|
Physical properties
|
can be measured without changing the identity and composition of the substance.
- color - odor - density - melting point - boiling point - hardness. |
|
|
Chemical properties
|
describe how a substance can change, or react, to form other substances.
- flammability. |
|
|
Intensive properties
|
Do not depend on the amount of the substance
- melting point - temperature - density. |
|
|
Extensive properties
|
Depend on amount
- weight - volume |
|
|
Physical change
|
Substance changes its appearance, but not its composition.
- ice to water |
|
|
Chemical change
|
A substance is transformed into a chemically different substance.
- Burning wood |
|
|
Physical or Chemical Property?
Zinc (Zn) (1) Silver-grey metal (2) Melting point 420 ºC (3) Reacts with O2 (or air) to form ZnO (4) Density = 7.13 g/cm3 (5) Dissolves in H2SO4 to give H2 gas |
(1) physical property
(2) physical property (3) chemical property (4) physical property (5) chemical property |
|
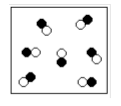
(1) Phase State
(2) Molecule or atoms (3) Compound (Y?N) (4) Mixture (Y?N), what kind |
(1) gas
(2) molecules (3) yes (4) no |
|

(1) Phase State
(2) Molecule or atoms (3) Compound (Y?N) (4) Mixture (Y?N), what kind |
(1) solid and gas
(2) atoms (3) no (4) yes, heterogenous |
|
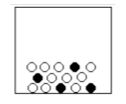
(1) Phase State
(2) Molecule or atoms (3) Compound (Y?N) (4) Mixture (Y?N), what kind |
(1) solid
(2) atoms (3) no (4) yes, homogeneous |
|

(1) Phase State
(2) Molecule or atoms (3) Compound (Y?N) (4) Mixture (Y?N), what kind |
(1) gas
(2) molecules and atoms (3) no (4) yes, homogeneous |
|
|
Separation of mictures
|
Mixtures can be separated
- filtration - distillation - chromatography |
|
|
Base Units
(1) Mass (2) Length (3) Time (4) Temperature (5) Amount of Substance |
(1) kilogram, kg
(2) meter, m (3) second, s or sec (4) Kelvin, K (5) mole, mol |
|
|
giga
G |
10^9
gigabyte |
|
|
mega
M |
10^6
megaton |
|
|
kilo
K |
10^3
1 kilometer (km) 1 kilogram (kg) |
|
|
deci
d |
10^ -1
1 deciliter (dL) |
|
|
centi
c |
10^-2
1 centiliter (cL) |
|
|
milli
m |
10^ -3
1 milligram (mg) 1 milliliter (mL) 1 millimole (mmol) |
|
|
micro
μ |
10^ -6
1 microgram (μg) |
|
|
nano
n |
10^ -9
1 nanometer (nm) |
|
|
pico
p |
10^-12
1 picometer (pm) |
|
|
femto
f |
10^ -15
1 femtosecond |
|
|
Density
|
Density is mass/volume
Commonly expressed as gm/ml or gm/cc Air 0.0001 g/cc Ethanol 0.79 Water 1.00 Table salt 2.16 Iron 7.9 Gold 19.32 |
|
|
Exact numbers
|
Are usually defined numbers.
- # of inches in a foot - # of people in the lecture theater |
|
|
Inexact Numbers
|
These are measured numbers, which always have a degree of uncertainty.
|
|
|
Precision
|
Precision is how closely a set of measurements agree with each other.
|
|
|
Accuracy
|
Accuracy is how closely the set of measurements agree with the correct or true value.
|
|
|
Significant Figures in Calculations
For multiplication and division |

The result contains the same number of significant figures as the measurement with the FEWEST SIGNIFICANT FIGURES
|
|
|
Significant Figures in Calculations
For addition and subtraction |

The result contains the same number of decimal places as the result with the FEWEST DECIMAL PLACES
|
|
|
Dimensional Analysis
Converting inches into cm |

|
|
|
Dimensional Analysis
Converting m/min into m/s: |
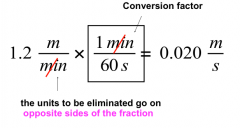
|
|
|
The Atomic Theory of Matter
Dalton 4 statements |
(1) Elements are made of tiny particles called atoms
(2) All atoms of one element are identical (3) Atoms are not created nor destroyed in chemical processes (4) Atoms of different elements can combine to form compounds |
|
|
The Atomic Theory of Matter
The Law of Multiple Proportions |
12 g carbon + 16 g oxygen => CO
12 g carbon + 32 g oxygen => CO2 32 g : 16 g = 2 : 1 |
|
|
J.J. Thomson
|
- Discovery of the electron through Cathode Ray
- Atoms of all elements contain electrons - Electron: negatively charged particles of very small mass - beams repelled by –ve charge, attracted by a +ve charge, and deflected by a magnetic field. - He was able only to calculate a charge to mass ratio from his experiments. |
|
|
Cathode ray tube
|
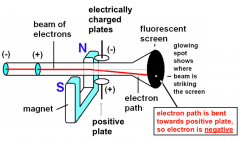
|
|
|
J.J. Thomson's Model
|
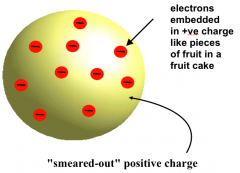
“Plum Pudding Model”
|
|
|
Robert Millikan
|
- Determination of mass of the electron
- Oil-drop experiment - Measured the charge on the electron, from which its mass could then be calculated |
|
|
MASS OF ELECTRON
|
MASS OF ELECTRON = 9.10 * 10^-28 g
|
|
|
Henri Becquerel
|
discovered that a Uranium compound spontaneously emitted high-energy radiation = radioactivity
|
|
|
Rutherford
|
- Discovery of the nucleus
- 3 types of radiation emitted, α, β, and γ particles -Experiments showed that with electrically charged plates (1) α rays were deflected towards the -ve plate, (2) γ rays were not affected (3) β rays were attracted towards the +ve plate. |
|
|
α particles are positive He nuclei
β particles are –ve electrons γ particles are neutral photons. |
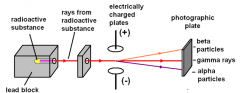
Rutherford analyzed particles emitted by radioactive substances
|
|
|
examined beams of α particles emitted by radioactive substances.
Nearly all of the beams passed straight through a sheet of gold foil a few thousand atoms thick- a few were scattered at large angles. From this Rutherford proposed the nuclear model of the atom. |
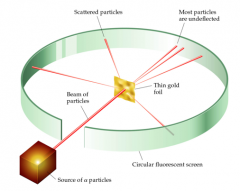
Rutherford’s gold foil experiment
|
|
|
The Modern View of Atomic Structure
|
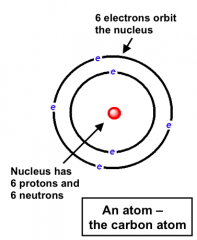
|
|
|
The charge on
(1) Electron (2) Proton (3) Neutron |
The charge on:
(1) Electron is -1.602 x 10-19 C (Coulombs) (2) Proton is +1.602 x 10-19 C (3) The neutron is neutral The chemical properties of the elements are controlled by the # of electrons and protons. |
|
|
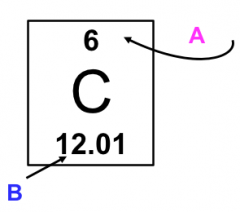
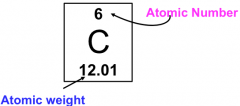
Atomic Number (Z)
|
Every atom has an equal number of protons and electrons
protons = electrons |
|
|
Isotopes
|
These have same Atomic Number (same number of protons) but different numbers of neutrons in the nucleus.
|
|
|
Mass Number
|
The Mass Number = # protons + # neutrons
|
|
|
Mass Number of Carbon
|
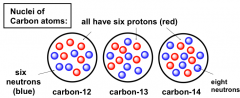
C (carbon) always has Z = 6, i.e. it has 6 protons
BUT can have different numbers of neutrons. |
|
|
Isotopes of Carbon
|
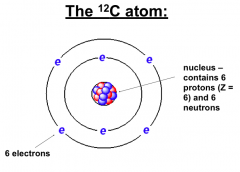
12-C and 14-C both have 6 protons (Atomic Number 6)
BUT 12-C has 6 neutrons, while 14C has 8. |
|
|
Isotopes of Hydrogen
|
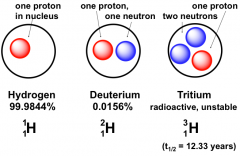
|
|
|
Atomic weights
|
- average masses
- expressed in amu |
|
|
Atomic Weight of "naturally occurring C"
12-C= 98.93% 13-C= 1.07% |
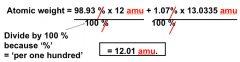
|
|
|
Average Atomic Mass
|
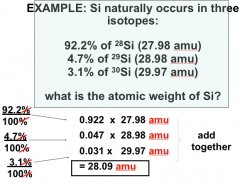
|
|
|
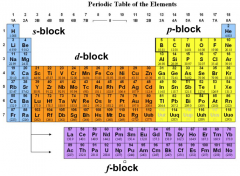
Mendeleev
|
The Periodic Table
|
|
|
s - block
d - block f - block p - block |
|
|
|
|
|
|
Groups in the periodic table
|
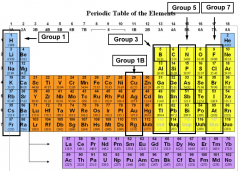
The vertical columns in the periodic table are called GROUPS.
group 1: Alkali metals group 8: Noble gases group 7: Halogens |
|
|
Periodic Table
Group 1 |
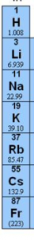
Alkali metals: are all soft silvery reactive metals that form basic hydroxides.
H, Li, Na, K, Rb, Cs, Fr |
|
|
Periodic Table
Group 8 |
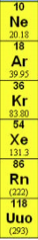
Noble gases: are all chemically unreactive gases.
Ne, Ar, Kr, Xe, Rn, Uuo |
|
|
Periodic Table
Group 7 |
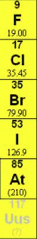
Halogens: all exist as X2 molecules, and form salts where they are the anion X-.
F, Cl, Br, I, Ar, Uus |
|
|
Periodic Table
Group 1B |

the coinage metals: corrosion-resistant metals
- They are all excellent conductors of electricity. - They all can form chlorides of the composition MCl (M = metal), such as CuCl, AgCl, and AuCl. |
|
|
Rows or Periods in the Periodic Table:
|
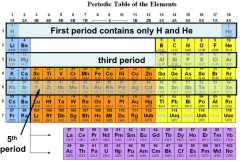
|
|
|
Elements exist as diatomic molecules at room temp
|
H, N, O, F, Cl, Br, I
(Diatomic molecules are molecules composed only of two atoms) |
|
|
Ions are formed when a neutral atom
|
- gains electrons or
- loses electrons |
|
|
(1) cation atom
(1) anion atom |
(1) An atom can LOSE one or more electrons to become a positively charged CATION
(2) An atom can GAIN electrons to become a negatively charged ANION |
|
|
The Empirical Formula
|
Shows the ratio of elements present in the compound. The ratios are denoted by subscripts next to the element symbols.
AKA: simplest formula Ex: Glucose has a molecular formula of C6H12O6. It contains 2 moles of hydrogen for every mole of carbon and oxygen. The empirical formula for glucose is CH2O. |
|
|
Molecular Formulas
|
An expression which states the number and type of atoms present in a molecule of a substance.
Ex: There are 6 C atoms and 14 H atoms in a hexane molecule, which has a molecular formula of C6H14. |
|
|
The Empirical Formula versus Molecular Formulas
|
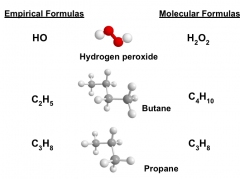
|
|
|
Empirical and Molecular Formulae of glucose
|
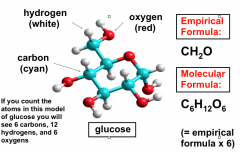
|
|
|
Structural Formula
Representation |
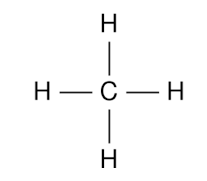
|
|
|
Ball- and- Stick Model
Representation |
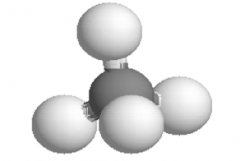
|
|
|
Perspective Drawing
Representation |
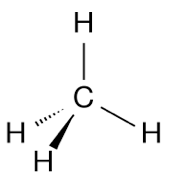
|
|
|
Space- Filling Model
Representation |
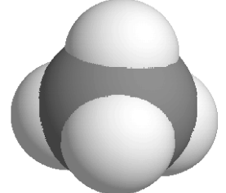
|
|
|
Ions are formed when a neutral atom
|
- gains electrons or
- loses electrons |
|
|
Ionic Compounds
|
Cations and Anions can combine to form Ionic Compounds
EX: (Na+) +(Cl-)= NaCl |
|
|
Empirical Formulas for Ionic Compounds:
|
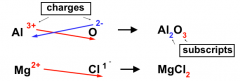
(A) determine charge on ions formed
(B) add ions so that compound is neutral overall A simple approach: simply swap the charge on one ion to be the subscript on the other |
|
|
Organic substances
|
Organic substances are compounds of carbon and hydrogen (plus other elements such as N or S) associated with living things.
EX: CH3CH2OH Ethanol |
|
|
Inorganic substances
|
Inorganic substances are substances not associated mainly with living things.
EX: AlCl3 aluminum chloride |
|
|
Combination reaction
|
Two or more substances react to form one product
|
|
|
Decomposition reaction
|
one substance breaks down into two or more
|
|
|
Combustion reactions
|
Combustion reactions are rapid reactions in air that produce a flame.
|
|
|
Hydrocarbons
|
Hydrocarbons consist only of C and H.
|
|
|
Formula weight
|
The formula weight (F. Wt.) is the sum of the atomic weights of each atom in its chemical formula.
If the chemical formula is that of a molecule, then the formula weight is referred to as a molecular weight (M. Wt.). F. Wt = (amu*quantity subscript) + ... |
|
|
Molecular weight
|
If the chemical formula is that of a molecule, then the formula weight is referred to as a molecular weight (M. Wt.).
M. Wt = (amu*quantity subscript) + ... |
|
M. Wt. of glucose
|
C= 12.0 amu
H= 1.0 amu O= 16.0 amu So F. Wt. = (6*12.0)+(12*1) +(6*16) = 180 amu |
|
|
Percentage composition from formula
|
% element =
(no. atoms of element) x (At. Wt. element) x 100 ______________________________________________________ F. Wt. of compound C/12 H/22 O/11 (sucrose): F. Wt. = 342.0 amu. % C = (12 x 12.0) x 100/342 = 42.1% % H = (22 x 1.0) x 100/342 = 6.4% % O = (11 x 16.0) x 100/342 = 51.5% check: 42.1 + 6.4 + 51.5 = 100.0%. |
|
|
Mole
|
F. Wt. or M. Wt. expressed in grams
EX: Water has a formula weight of 18.0 amu, so 1 mole of water weighs 18.0 grams. |
|
|
Avogadro’s number
|
6.022 * 10^23
|
|
|
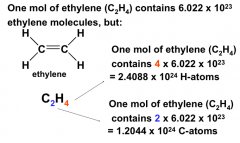
Numbers of atoms in molecules
|
|
|
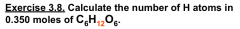
|

|
|
|
Quantitative Information from Balanced Equations
|
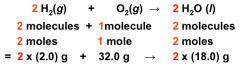
The coefficients in a balanced equation represent
(1) relative numbers of molecules involved in the reaction (2) AND THE RELATIVE NUMBERS OF MOLES, and therefore the relative masses |
|
|
Weights of products and reactants in a balanced equation
|

|
|
|
Limiting Reagent
|
If we have exactly 2 mol of H2 and 1 mol of O2, then we can make 2 mol of water. But what if we have 4 mol of H2 and 1 mol of O2. Now we can make only 2 mol H2O with 2 mol H2 left over. In this case the O2 is the limiting reagent.
|
|
|
Solve limiting reagent problems
|
Step 1. Convert to moles
Step 2. Guess limiting reagent (it doesn’t matter if you guess wrong) Step 3. Multiply all the moles in the equation by the factor: Step 4. Compare required moles of Na3PO4 with moles we previously calculated Step 5. Convert moles to grams |
|
|
Percent Yield
|
The quantity of product that forms if all of the limiting reagent reacts is called the theoretical yield. Usually, we obtain less than this, which is known as the actual yield.
Percent yield = actual yield (g) x 100% __________________________ theoretical yield (g) |
|
|
Gases
|
(1) expand to fill their containers
(2) volume decreases with increasing pressure (3) Gases always form homogeneous mixtures with each other, even gasoline vapor and water vapor, which do not mix as liquids |
|
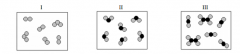
Which of the following diagrams represents a pure substance?
(a) I and II (b) I, II, and III (c)I only (d) II only |
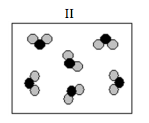
Which of the following diagrams represents a pure substance?
(d) II only |
|
|
Silicon (Si) is an element found in group 4A of the periodic table. Which of the following is a chemical property of silicon?
(a) The electrical resistance of crystalline silicon changes with mechanical stress (b) Silicon reacts with oxygen to form silicates (c) Silicon is a grey solid at room temperature (d) The melting point of silicon is 1414 |
Silicon (Si) is an element found in group 4A of the periodic table. Which of the following is a chemical property of silicon?
(b) Silicon reacts with oxygen to form silicates |
|
|
Silicon has a tabulated density of 2.329 g/cm3. A laboratory technician has determined the density of silicon experimentally. The values she determined are listed below. Which value is the most precise?
(a) 2.33 ± 0.52 (b) 2.24 ± 0.02 (c) 2.29 ± 0.22 (d) 2.34 ± 0.17 |
Silicon has a tabulated density of 2.329 g/cm3. A laboratory technician has determined the density of silicon experimentally. The values she determined are listed below. Which value is the most precise?
(b) 2.24 ± 0.02 |
|
|
The density of an unknown solid was determined as 0.178 lb/in3. Which substance is it?
(a) Iodine, d=4.93 g/cm3 (b) Arsenic, d=5.83 g/cm3 (c) Germanium, d=5.32 g/cm3 (d) Aluminum, d=2.70 g/cm3 |
The density of an unknown solid was determined as 0.178 lb/in3. Which substance is it?
(a) Iodine, d=4.93 g/cm3 llb=435.6g 0.178lb/1inch * 456.6g/1lb = 80.74g/inch^3 80.74g/inch^3 * in^3/76.387m^3= 3 - |
|
|
The discovery of the ______________ by ______________ led to the proposal of the first atomic models in the early 20th century.
(a) proton, E. Rutherford (b) electron, R. Millikan (c) electron, J.J. Thompson (d) proton, J.J. Thompson |
The discovery of the ______________ by ______________ led to the proposal of the first atomic models in the early 20th century.
(c) electron, J.J. Thompson |
|
|
Which atom has the largest number of neutrons?
(a) 31 P (b) 37 Cl (c) 39 K (d) 40 Ar |
Which atom has the largest number of neutrons?
(d) 40 Ar |
|
|
Vanadium has two naturally occurring isotopes, 50
V with an atomic mass of 49.9472 amu and 51 V with an atomic mass of 50.9440 amu. The atomic weight of vanadium is 50.9415 amu. The percent natural abundances of the vanadium isotopes are ________% 50 V and ________ % 51V. (a) 50, 50 (b) 1.00, 99.00 (c) 0.25, 99.75 (d) 99.75, 0.25 |
(c) 0.25, 99.75
|
|
|
Elements _____________________ exhibit similar physical and chemical properties.
(a) with similar atomic masses (b) in the same group of the periodic table (c) in the same period of the periodic table (d) with similar atomic numbers |
Elements _____________________ exhibit similar physical and chemical properties.
(b) in the same group of the periodic table |
|
|
Which of the following is a molecular (covalent) compound?
(a) KClO4 (b) NH4ClO3 (c) SO3 (d) Na2Cr2O7 |
Which of the following is a molecular (covalent) compound?
(c) SO3 |
|
|
Molecular (covalent) compound
|
elements share electrons via covalent bonds
Molecular compounds contain two or more nonmetals. Usually you can recognize you are dealing with a molecular compound because the first element in the compound name is a nonmetal. Some molecular compounds contain hydrogen, but if you see a compound which starts with "H", you can assume it is an acid and not a molecular compound. Compounds consisting only of carbon with hydrogen are called hydrocarbons. Hydrocarbons have their own special nomenclature, so they are treated differently from other molecular compounds. |
|
|
The systematic name for Fe3(PO4)2 is
(a) iron(III) phosphate (b) triiron diphosphate (c) iron(II) phosphate (d) iron(III) diphosphate |
The systematic name for Fe3(PO4)2 is
(c) iron(II) phosphate |
|
|
Given that the formula for potassium iodate is KIO3, the formula for magnesium iodite would be:
(a) MgIO2 (b) MgIO4 (c) Mg(IO)2 (d) Mg(IO2)2 |
Given that the formula for potassium iodate is KIO3, the formula for magnesium iodite would be:
(d) Mg(IO2)2 |
|
|
When the following unbalanced reaction is properly completed and balanced, the product and its coefficient are:
Cs(s) + S2(g) => ? (a) CsS2 (b) 2 CsS (c)2 Cs2S (d) Cs3S2 |
When the following unbalanced reaction is properly completed and balanced, the product and its coefficient are:
(c) 2 Cs2S |
|
|
In the balanced chemical reaction for the combustion of ethyl acetate, C/4 H/8 O/2, what is the stoichiometric coefficient for oxygen?
(a) 4 (b) 5 (c) 6 (d) 2 |
In the balanced chemical reaction for the combustion of ethyl acetate, C/4 H/8 O/2, what is the stoichiometric coefficient for oxygen?
(b) 5 |
|
|
Stoichiometric coefficient
|
The number of molecules of a reactant taking part in a reaction is known as Stoichiometric coefficient.
|
|
|
What percentage of ethanol (CH/3 CH/2 OH) is oxygen by mass?
(a) 11.1% (b) 53.3% (c) 34.8% (d) 16.0% |
What percentage of ethanol (CH/3 CH/2 OH) is oxygen by mass?
(c) 34.8% |
|
|
How many moles of oxygen are in 1.0 mole of calcium nitrate?
(a) 0.17 (b) 3.0 (c) 4.0 (d) 6.0 |
How many moles of oxygen are in 1.0 mole of calcium nitrate?
(d) 6.0 |
|
|
Sand is made up of silicon dioxide, SiO2 (molar mass=60). How many silicon atoms are in a single grain of sand weighing 55.2 μg?
a. 5.53 × 1020 atoms b. 5.53 × 1017 atoms c. 1.53 × 10-30 atoms d. 1.81 × 10-18 atoms |
Sand is made up of silicon dioxide, SiO2 (molar mass=60). How many silicon atoms are
in a single grain of sand weighing 55.2 μg? b. 5.53 × 1017 atoms |
|
|
One reaction involved in the production of steel is:
Fe2O3(s) + 2 C(s) → 2 Fe(l) + 3 CO(g) molar mass (g/mol): 159.6 12 55.8 28 How many grams of Fe2O3 must react to produce 250 g of Fe? a. 42 g b. 130 g c. 360 g d. 740 g |
One reaction involved in the production of steel is:
Fe2O3(s) + 2 C(s) → 2 Fe(l) + 3 CO(g) molar mass (g/mol): 159.6 12 55.8 28 How many grams of Fe2O3 must react to produce 250 g of Fe? c. 360 g |

