![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
39 Cards in this Set
- Front
- Back
|
Metabolism
|
Metabolism is the sum of the chemical reactions in an organism.
|
|
|
Catabolism
|
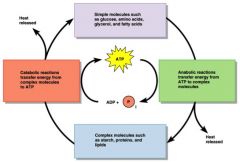
Catabolism is the energy-releasing processes.
|
|
|
Anabolism
|
Anabolism is the energy-using processes.
|
|
|
Metabolic pathway
|
A metabolic pathway is a sequence of enzymatically catalyzed chemical reactions in a cell.
Metabolic pathways are determined by enzymes. Enzymes are encoded by genes. |
|
|
Collision theory
|
The collision theory states that chemical reactions can occur when atoms, ions, and molecules collide.
|
|
|
Activation energy
|
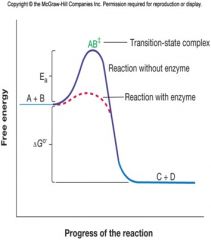
Activation energy is needed to disrupt electronic configurations.
|
|
|
Reaction rate
|
Reaction rate is the frequency of collisions with enough energy to bring about a reaction.
Reaction rate can be increased by enzymes or by increasing temperature or pressure. |
|
|
Enzymes
|
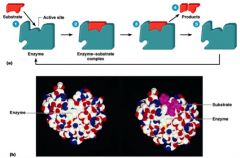
carry out reactions at physiological conditions so they proceed in a timely manner
enzymes speed up the rate at which a reaction proceeds toward its final equilibrium |
|
|
The Mechanism of Enzyme Reactions
|
A + B -> AB‡ -> C + D
AB‡ transition-state complex – resembles both the substrates and the products a typical exergonic reaction |
|
|
How Enzymes Lower Ea
|
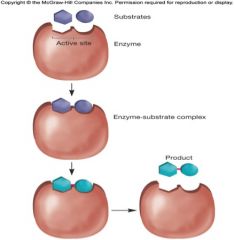
by increasing concentrations of substrates at active site of enzyme
by orienting substrates properly with respect to each other in order to form the transition-state complex two models for enzyme-substrate interaction -lock and key and induced fit |
|
|
protein catalysts
|
have great specificity for the reaction catalyzed and the molecules acted on
|
|
|
catalyst
|
substance that increases the rate of a reaction without being permanently altered
|
|
|
substrates
|
reacting molecules
|
|
|
products
|
substances formed by reaction
|
|
|
Structure and Classification of Enzymes
|
some enzymes are composed solely of one or more polypeptides
some enzymes are composed of one or more polypeptides and nonprotein components |
|
|
apoenzyme
|
protein component of an enzyme
|
|
|
cofactor
|
nonprotein component of an enzyme
*prosthetic group – firmly attached *coenzyme – loosely attached |
|
|
holoenzyme
|
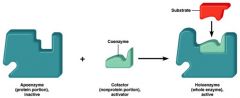
holoenzyme = apoenzyme + cofactor
|
|
|
Important Coenzymes
|
NAD+
NADP+ FAD Coenzyme A |
|
|
Coenzymes as Carriers
|
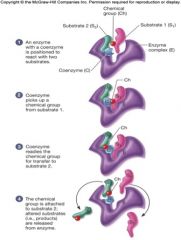
often act as carriers, transporting substances around the cell
|
|
|
Environmental Effects on Enzyme Activity
|
enzyme activity is significantly impacted by :
-substrate concentration -pH -temperature |
|
|
Effect of [substrate]
|
rate increases as [substrate] increases
no further increase occurs after all enzyme molecules are saturated with substrate |
|
|
Effect of pH and Temperature
|
each enzyme has specific pH and temperature optima
|
|
|
denaturation
|
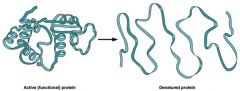
loss of enzyme’s structure and activity when temperature and pH rise too much above optima
|
|
|
competitive inhibitor
|
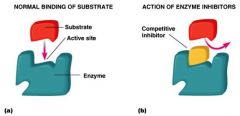
directly competes with binding of substrate to active site
|
|
|
noncompetitive inhibitor
|
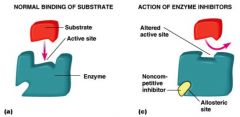
binds enzyme at site other than active site
changes enzyme’s shape so that it becomes less active |
|
|
Ribozymes
|
Thomas Cech and Sidney Altman discovered that some RNA molecules also can catalyze reactions
|
|
|
Regulation of Metabolism
|
important for conservation of energy and materials
maintenance of metabolic balance despite changes in environment |
|
|
Metabolic Regulation
|
three major mechanisms
1. metabolic channeling 2. regulation of the synthesis of a particular enzyme (transcriptional and translational) 3. direct stimulation or inhibition of the activity of a critical enzyme -post-translational |
|
|
Metabolic Channeling
|
differential localization of enzymes and metabolites
|
|
|
compartmentation
|
differential distribution of enzymes and metabolites among separate cell structures or organelles
can generate marked variations in metabolite concentrations |
|
|
Post-Translational Regulation of Enzyme Activity
|
two important reversible control measures
1. allosteric regulation 2. covalent modification |
|
|
Allosteric Regulation
|
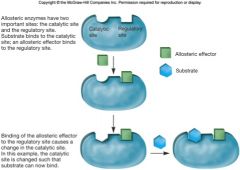
most regulatory enzymes
activity altered by small molecule |
|
|
allosteric effector
|
binds non-covalently at regulatory site
changes shape of enzyme and alters activity of catalytic site positive effector increases enzyme activity negative effector inhibits the enzyme |
|
|
Covalent Modification of Enzymes
|
reversible on and off switch
addition or removal of a chemical group (phosphate, methyl, adenyl) |
|
|
Advantages of Covalent Modification of Enzymes
|
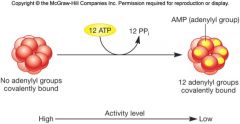
respond to more stimuli in varied and sophisticated ways
regulation on enzymes that catalyze covalent modification adds second level |
|
|
Feedback Inhibition
|
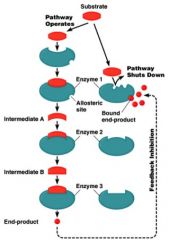
also called end-product inhibition
inhibition of one or more critical enzymes in a pathway regulates entire pathway -pacemaker enzyme each end product regulates its own branch of the pathway each end product regulates the initial pacemaker enzyme |
|
|
pacemaker enzyme
|
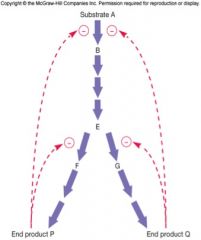
catalyzes the slowest or rate-limiting reaction in the pathway
|
|
|
isoenzymes
|
different enzymes that catalyze same reaction
|

