![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
228 Cards in this Set
- Front
- Back
|
Describe the relationship between resistance to blood flow and diameter of a blood vessel
|
resistance to flow is inversely proportional to the 4th power of the diameter (increase diameter -> increase flow -> decrease flow resistance)
|
|
|
what are the three main types of endothelial functions?
|
1. Barrier
2. Transport 3. Metabolic |
|
|
what is the most common cause of ateriosclerosis?
|
atherosclerosis
|
|
|
what type of blood vessels does atherosclerosis effect?
|
large and medium sized arteries
|
|
|
why do atherosclerotic plaques like to form at the branch points of arteries?
|
these are sites of hemodynamic turbulence
|
|
|
atherosclerosis is a disease that primarily effects which layer of a blood vessel?
|
intima
|
|
|
describe the pathogenesis and components of atherosclerotic disease
|
- endothelial dysfunction
- lipid accumulation, smooth muscle proliferation in intima - lipids cause cell injury, this attracts macrophages - macrophages scavenge extracellular lipid and release growth factors - eventual damage to endothelium causes platelets to adhere |
|
|
what could causethe dysfunction (or damage) to the arterial endothelium seen in atherosclerosis? (6)
|
- hypercholesterolemia
- mechanical injury - hypertension - immune mechanisms - toxins - viruses |
|
|
name the growth factors released by macrophages in the atherosclerotic process
(5) |
FGF
TNF IL-1 IFN TGF-B |
|
|
what are the two possible precursors to atherosclerosis (physical changes)
|
1. Fatty Streak
2. Intimal Cell Mass |
|
|
what are the pros and cons of the "fatty streak" precursor hypothesis?
|
pro-composed of foamy macrophages
cons-seen in regions where atherosclerosis is not commonly found |
|
|
What are the pros and cons of the "intimal cell mass" hypothesis?
|
pros - nodules of intimal smooth muscle at arterial branch points
cons - may only be age related change, no lipid deposition |
|
|
what is the current prevalent thought explaining the pathogenic mechanism of atherosclerosis?
|
multifactorial process
|
|
|
name the 5 most common places atherosclerosis is found
|
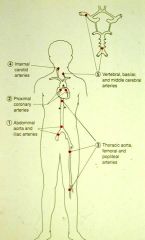
1. abdominal aorta and iliacs
2. proximal coronary arteries 3. thoracic aorta, femoral, popliteal arteries 4. internal carotids 5. vertebral, basilar, middle cerebral arteries |
|
|
name 7 possible complications of atherosclerosis
|
1. thrombosis (acute occlusion)
2. chronic stenosis (ischemia) 3. hemorrhage (into plaque) 4. aneurysm formation 5. ulceration (with atheroemboli) 5. calcification |
|
|
what are the 4 most important risk factors in the development of atherosclerosis?
|
1. hyperlipidemia
2. hypertension 3. cigarette smoking 4. diabetes mellitus |
|
|
what are the other 5 risk factors in the development of atherosclerosis?
|
1. age
2. sex (M>F) 3. genetics 4. diet 5. lifestyle (personality type, exercise habits) |
|
|
name the 5 lipid types in order of size (large to small)
|
1. chylomicron
2. VLDL 3. IDL 4. LDL 5. HDL |
|
|
what is inheritance pattern of type II familial hypercholesterolemia?
|
autosomal dominant
(mutation is on short arm of chromosome 19) |
|
|
describe the mutation subsequent effects of type II familial hypercholesterolemia
|
mutation causes malfunctioning LDL receptor
(LDLs must be processed through a receptor independant pathway) this leads to random cholesterol buildup |
|
|
name some secondary causes of hyperlipoproteinemia
|
hypothyroidism
diabetes mellitus nephrotic syndrome renal failure alcoholism obstructive liver disease pancreatitis glycogen storage disease porphyria |
|
|
name 6 possible associations/complications of hypertension
|
atherosclerosis
angina sudden cardiac death aortic dissection cerebral infarction intracerebral hemorrhage |
|
|
95% of hypertension is _____
5% of hypertension is ____ |
95% - essential (idiopathic)
5% - renal disease, endocrine related, other |
|
|
3 modifiying factors of cardiac output
|
1. Blood volume
2. heart rate 3. contractility |
|
|
3 modifying factors of TPR?
|
1. humoral factors (dilators, constrictors)
2. local factors (autoregulations) 3. neural factors (dilators, constrictors) |
|
|
what are the blood pressures for the following?
1. Normal 2. Prehypertension 3. Stage 1 hypertension 4. Stage 2 hypertension |
1. <120/80
2. 120-139/80-89 3. 140-159/90-99 4. >160/>100 |
|
|
name two renal associated causes of hypertension
|
1. chronic renal disease
2. renovascular disease |
|
|
name two aortic problems known to cause hypertension
|
1. coarctation of the aorta
2. rigidity of the aorta |
|
|
name a vessel change often caused by benign (chronic) hypertension
|
hyaline arteriosclerosis
(basement membrane thickening due to hyaline material in vessel walls) |
|
|
what are the renal complications of hyaline arteriosclerosis?
|
may cause chronic renal insufficiency via arteriolar nephrosclerosis (aka. global glomerular sclerosis)
|
|
|
name a vessel change often seen in malignant (accelerated) hypertension
|
hyperplastic arteriolosclerosis
|
|
|
what does hyperplastic arteriosclerosis look like and what are the presenting symptoms?
|
it is a fibrinous necrosis where the arterioles look like onion skin. (this is a reaction to the high pressures)
patient would present with headache, blurred vision, malaise |
|
|
what is Monckeberg's medial calcific sclerosis?
|
a degenerative calcification of the MEDIA of the artery. (intima not effected)
seen in elderly |
|
|
what is fibromuscular dysplasia?
|
- irregular wavy thickening of medium sized arteries.
- renal and carotids most often affected - women of reproductive age most often affected - may also see alpha-1-antitrypsin deficiency |
|
|
complications of fibromuscular dysplasia? (2)
|
1. renovascular HTN
2. arterial dissection |
|
|
Fibromuscular dysplasia
|

What is this disorder?
|
|
|
hyperplastic arteriolosclerosis
|
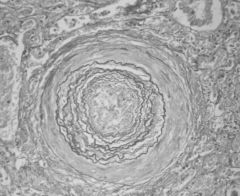
what is this?
|
|
|
hyaline arteriolosclerosis
|
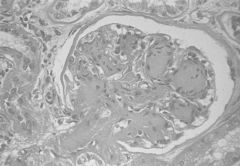
what is this?
|
|
|
name the four most common sites where atherosclerosis is found IN ORDER OF OCCURANCE
|
1. abdominal aorta
2. coronary artery 3. popliteal artery 4. carotid artery |
|
|
name 6 possible complications of HTN
|
1. CHD
2. CHF 3. LVH 4. nephropathy 5. retinopathy 6. CVA |
|
|
which portion of the blood pressure is responsible for most of the complications seen in HTN?
|
SYSTOLIC blood pressure
(pulse pressure is also partially responsible for CV events) |
|
|
what is:
1. normal BP 2. Prehypertension 3. Stage 1 HTN 4. Stage 2 HTN |
1. <120/<80
2. 120-139 / 80-89 3. 140-159 / 90-99 4. >160 / >100 |
|
|
the risk of what three events goes down when blood pressure is lowered?
|
1. Stroke (40%)
2. MI (25%) 3. Heart Failure (50%) |
|
|
in which two conditions should we change our target (goal) BP from <140/90 to <130/90?
|
1. Diabetes Mellitus
2. Renal Impairment |
|
|
describe short term control of BP when the BP is low
|
low BP = decreased stretch of vessel wall
decreased baroreceptor activity leads to decreased inhibition of vasomotor center increased SNS discharge = vasoconstriction, increased CO |
|
|
describe short term control of BP when the BP is high
|
high BP = increased stretch of vessel wall
baroreceptor stimulation inhibits SNS output less SNS - vasodilation, decreased CO |
|
|
what is most responsible for long term control of BP?
|
the kidney
(RAAS) |
|
|
when RAAS is activated, what is the result?
|
Na+ and volume retention
increase in SNS activity |
|
|
function of renin?
|
converts Angiotensinogen to AT1
|
|
|
function of ACE?
|
converts AT1 to AT2
(also breaks down bradykinin) |
|
|
result when AT1 receptors are stimulated? (6)
|
1. vasoconstriction (coronary, renal, cerebral)
2. aldosterone release 3. direct Na+ retention 4. water retention (vasopressin release) 5. renin supression (negative feedback) 6. SNS activation |
|
|
result when AT2 receptors are stimulated? (5)
|
1. CV protection
2. release of NO (vasodilation) 3. antidiuresis 4. antiproliferative 5. apoptosis |
|
|
where are the four major mechanisms of action for antihypertensive drugs?
|
1. Volume depleters (diuretics)
2. Vasodilators - direct and CCBs 3. Adrinergic inhibitors 4. RAAS inhibitors |
|
|
three classes of diuretics used in treatment of HTN?
|
1. Thiazides
2. Loop 3. K+ sparing |
|
|
what is the diuretic of choice in uncomplicated HTN?
|
HCTZ
|
|
|
which diuretic is used when more diuresis is required than can be obtained with HCTZ?
|
Loop (most often Furosemide)
|
|
|
when would K+ sparing diuretics be used?
Name three. |
they would be prescribed along with other diuretics to conserve K+
1. Amiloride 2. Spironolactone 3. Triamterene |
|
|
MOA of thiazides?
|
block Na+ and Cl- reabsorption in distal convoluted tubule
(elimate 5-10% of filtered Na+, relatively weak) |
|
|
Describe the actions of thiazides on the following ions:
1. K+ 2. Ca++ 3. Mg+ 4. uric acid |
1. K+ wasting
2. decreased excretion of Ca++ 3. increased excretion of Mg+ 4. decreased excretion of uric acid |
|
|
MOA of loop diuretics?
|
reduce Na+ resorption from the ascending limb of the loop of Henle
(eliminates 20-25% of filtered Na+ -> more potent diuresis than HCTZ, but not as good as lowering BP) |
|
|
compare the onset and duration of action of Loop vs Thiazide diuretics
|
onset - Loop diuretics fastest
duration of action - Thiazides longest |
|
|
adverse effects of diuretics?
|
1. decreased serum ion concentrations of K+, Mg+, Na+
2. Hypovolemia 3. Hyperuricemia (watch in gout) 4. Hyperlipidemia 5. mild hyperglycemia (watch in DM) 6. hypercalcemia (thiazides) |
|
|
MOA of K+ sparing diuretics?
|
inhibit reabsorption of Na+ is distal convoluted tubule and collecting duct. (Weak - <5% of filtered Na+)
**inhibit exchange of Na+ for K+ or H+; therefore reduce K excretion |
|
|
differences in MOA between
1. spironolactone 2. triamterene, amiloride |
1. competitive antagonist of aldosterone
2. block Na+ channel |
|
|
what do alpha-1 receptors do?
|
mediate vasoconstriction
(Beta-2 mediate vasodilation) |
|
|
effect of beta blockers on CV system?
1. HR 2. contractility 3. renin release 4. BP 5. AV conduction |
1. decreased HR
2. decreased contractility 3. reduced renin release 4. reduced BP 5. reduced AV conduction |
|
|
one adverse effect of beta blockers is unopposed a-adrinergic stimulation. what is the consequence of this?
|
vasoconstriction and ischemia
|
|
|
other adverse effects of beta blockers? (5)
|
1. bronchospasm
2. can mask hypoglycemia s/s 3. decrease in peripheral circulation 4. decrease in exercise tolerance 5. decrease in HDL |
|
|
contraindications of beta blockers? (5)
|
1. asthma
2. cardiogenic shock 3. AV block 4. acute CHF 5. decreased HR |
|
|
why must the abrupt withdrawal of beta blockers be avoided?
|
abrupt withdrawal causes a rebound increase in HR and contractility (could be disastrous)
therefore we must taper over 1-2 wks. whenever possible |
|
|
name 3 alpha-1 blockers used in the treatment of HTN
|
1. doxazosin
2. prazosin 3. terazosin |
|
|
blocking the alpha-1 receptors results in? (2)
|
1. reduction of arteriolar resistance
2. increase in venous capacitance |
|
|
what is the "first dose phenomenon" in relation to a-1 blockers?
|
initial dose causes:
orthostatic hypotension sudden syncope |
|
|
two other adverse effects of a-1 blockers?
|
1. fluid retention
2. vertigo |
|
|
besides lowering BP: 3 other benefits of a-1 blockers?
|
1. lower LDLs and TGs, raise HDL
2. may help Reynaud's (although CCBs preferred Tx) 3. reduction of urinary retention symptoms in BPH |
|
|
name the 2 most commonly used centrally acting a-2 agonists in tx of HTN?
|
Clonidine
methyldopa |
|
|
effect of centrally acting a-2 agonists on CO?
|
unchanged or mildly decreased
|
|
|
MOA of centrally acting a-2 agonists?
|
cross BBB
decrease SNS outflow reduce catecholamine release results in decrease of PVR and HR |
|
|
methyldopa is a prodrug -what is it metabolized into?
MOA? |
alpha-methylnorepinephrine
potent peripheral vasoconstrictor, but main action is in CNS: reduces CNS sympathetic output to periphery |
|
|
MOA of reserpine?
|
inhibits peripheral NE, dopamine and serotonin uptake - results in vasodilation, decreased HR and contractility
|
|
|
adverse effects of reserpine?
|
bradycardia
possible CHF increased gastric acid |
|
|
two classes of CCBs?
|
non-dihydropyridines
dihydropyridines |
|
|
name the two non-dihydropyridines
|
1. dilitazem
2. verapamil |
|
|
name the two most commonly used dihydropyridines
|
1. amlodipine
2. nifedipine |
|
|
describe the CCBs MOA as it relates to Ca++
|
CCBs block L type Ca++ channels
-this decreases entry of Ca++ into the cell (also decreases intracellular[Ca++] concentration) |
|
|
on which type of muscle does Ca++ have the most effect on?
|
smooth muscle
|
|
|
effect of CCBs on arteries?
|
vasodilation
(decrease afterload on heart) |
|
|
do CCBs decrease preload?
|
NO - CCBs don't really venodilate
|
|
|
which type of CCBs have greater coronary and peripheral vasodilation properties?
|
dihydropyridines
nifedipine amlodipine |
|
|
where do verapamil and diltiazem have their greatest effect?
|
on the heart
negative inotropes decrease AV conduction decrease HR |
|
|
why is reflex tachycardia observed when dihydropyridines are used?
|
dihydropyridines vasodilate:
this reflexively activates the SNS, resulting in tachycardia |
|
|
adverse effects of DHP CCBs?
|
1. flushing, diziness, headaches (due to vasodilation)
2. hypotension 3. pedal edema |
|
|
adverse effects of non-DHP CCBs?
|
1. cardiotoxic effects (too low of HR, neg. inotropy; too much AV blockade)
2. constipation (verapamil) |
|
|
besides HTN, what else can CCBs be prescribed for?
|
angina
Reynauds syndrome African Americans |
|
|
relation between mortality and CCBs?
|
increased mortality seen with short acting CCBs
|
|
|
MOA of ACEIs?
|
inhibit ACE
- reduces levels of AT2 - prevents inactivation of bradykinin (potent vasodilator) - renin release is increased b/c of loss of feedback inhibition |
|
|
why do ACEIs not completely block the effects of AT2?
|
peripheral enzymes are still converting AT1 to AT2
|
|
|
two ACEI side effects that bradykinin is responsible for?
|
1. cough
2. angioedema |
|
|
three MAJOR indications for ACEI use?
|
1. HTN
2. CHF 3. diabetic renal disease |
|
|
effect of ACEIs on serum [K+]?
|
increase serum [K+]
|
|
|
cautions/contraindications of ACEIs?
|
pregnancy (2nd/3rd trimester)
renal disease or failure |
|
|
benefits of ARBs over ACEIs? (2)
|
1. ARBs inhibit RAAS more completely than ACEIs
2. ARBs don't increase bradykinin levels |
|
|
MOA of ARBs?
|
block AT1 receptors leading to decreased BP.
No ACE activity |
|
|
Both ACEIs and ARBs can be combined with what HTN therapy to have an additive effect?
|
HCTZ
|
|
|
Adverse reactions of ARBs? (4)
|
1. hyperkalemia
2. fetal renal toxicity 3. reduced renal function 4. rare angioedema (less than ACEIs) |
|
|
K+ and Mg++ excretion is an effect of aldosterone that can cause?
|
arrythmias
|
|
|
effects of aldosterone? (6)
|
1. edema (due to Na+ and H2O retention)
2. endothelial fibrosis 3. increased blood clotting and platelet activation 4. cytokine activation resulting in vascular inflammation 5. LV dysfunction/hypertrophy/fibrosis 6. arrythmias |
|
|
name of aldosterone receptor antagonist used to treat HTN?
|
spironolactone
|
|
|
MOA of spironolactone?
|
aldosterone receptor antagonist
- binds at aldosterone dependant Na+/K+ exchange site in distal convoluted tubule - increases Na+ and H20 excretion, increases K+ reabsorption WEAK DIURETIC |
|
|
side effects of spironolactone? (4)
|
progestational and antiandrogenic side effects
1. gynecomastia 2. sexual dysfunction 3. irregular menses or amenorrhea 4. postmenopausal bleeding |
|
|
name of drug that is a selective aldosterone blocker (SAB) that is better tolerated compared to spironolactone?
|
eplernone (INSPRA)
used for HTN, post MI heart failure |
|
|
why is SAB better tolerated than spironolactone?
|
SAB has less affinity for androgen/progesterone receptors
|
|
|
which HTN drug is not a first line agent but may be used in resistant HTN?
|
Hydralazine
|
|
|
MOA of hydralazine?
|
direct acting arterioloar vasodilator, decreases PVR
(acetylated by liver) |
|
|
adverse effects of hydralazine?
|
1. reflex sympathetic activity
2. Na+ retention (need for a diuretic) 3. DRUG INDUCED LUPUS (dose related) 4. peripheral neuritis (interferes with B6 metabolism) |
|
|
Why is Monoxidil, a potent direct acting vasodilator useful in resistant HTN, seldom used?
|
many adverse effects
- hirsutism - pulmonary fibrosis - some cardiac myocyte damage - marked Na+ and H20 retention |
|
|
In multiple agent therapy of HTN: what drug is almost always one of the drugs used?
|
HCTZ
|
|
|
a localized dilation of an artery is known as?
|
an aneurysm
|
|
|
where exactly in the blood vessel is the weakness that is seen in aneurysms?
|
in the media
can be a genetic or acquired weakness |
|
|
name 5 etiologies of aneurysms
|
1. atherosclerosis
2. syphilis 3.mediastinal/retroperitoneal fibrosis 4. berry 5. mycotic |
|
|
two shape types of aneurysms
|
1. fusiform
2. saccular |
|
|
syphilitic aneurysms are most commonly seen in which population?
|
males 40-55 yrs
|
|
|
where are syphilitic aneurysms most commonly observed?
|
ascending aorta
(occasionally descending and abdominal) |
|
|
describe the pathogenesis of a syphilitic aneurysm
|
1. T. pallidum casues obliterative endarteritis of the vasa vasorum
2. this leads to ischemia of media and adventitia 3. weakens aortic wall, get aneurysm |
|
|
possible complications of syphilitic aneurysm?
|
aortic root dilation -> causes aortic regurgitation -> possible death from heart failure or rupture
- also: coronary ostial stenosis, severe aortic atherosclerosis and fibrosis ("tree bark") |
|
|
5 risk factors for an atherosclerotic aneurysm?
|
1. hypertension
2. smoking 3. genetic factors (20% w/ first degree relative) 4. age (MC 6-9th decade) 5. sex (M>>F) |
|
|
why is the abdominal aorta the most common location of atherosclerotic aneurysms?
|
no vasa vasorum below orfices of renal arteries
|
|
|
pathogenesis of an atherosclerotic aneurysm?
|
destruction and weakening of media by severe atherosclerosis. Increased collagenase, elastase activity in aneurysm
|
|
|
clinical manifestations of atherosclerotic abdominal aortic aneurysms?
|
**75% are asymptomatic**
- pulsatile mass w/ pain - bruit - rupture: severe pain (most often L flank), hypotension |
|
|
in the case of an AAA - why is the lumen size of the aorta often not enlarged?
|
most of the dilation is due to a thrombus that has formed
|
|
|
mortality rate of a ruptured AAA?
|
90%
|
|
|
5 possible complications of atherosclerotic aneurysm?
|
1. rupture
2. aorto-enteric fistula 3. aorto-caval fistula 4. infection 5. embolization |
|
|
where are Berry aneurysms most commonly found?
|
in the Circle of Willis
- most often in the bifurcations (Anterior commucnicating a. with anterior cerebral a.) |
|
|
pathophysiology of Berry aneurysms?
|
**most are congenital**
high BP contributes |
|
|
pathophysiology of mycotic aneurysm?
|
weakening of arterial wall secondary to an infection (usually occurs in patients with known sepsis)
|
|
|
is a dissecting aortic aneurysm a true or false aneurysm?
|
false aneurysm (just involves a tear in the intima)
|
|
|
risk factors for an aortic dissection?
|
1. hypertension (70%)
2. Marfans (defect in fibrillin) 3. Ehlers-Danlos (defect in collagen) 4. Pregnancy (increased plasma volume) 5. coarctation of aorta 6. aortic valve malformation 7. trauma 8. copper deficiency (causes defect in lysyl oxidase) 9. cystic medial degeneration |
|
|
where is the most common location of an aortic dissection (within the blood vessel layers)
|
between outer 1/2 and inner 2/3 of aortic wall
|
|
|
presenting symptoms of aortic dissection:
|
1. severe, tearing chest pain radiating to back
2. +/- loss of pulses (due to pinched branch vessels) 3. +/- aortic insufficiency |
|
|
fatality rate of aortic dissections if untreated?
|
90%
|
|
|
possible complications of a ruptured aortic dissection? (3)
|
1. cardiac tamponade
2. bleeding into pleural cavity, peritoneal cavity, mediastinum, retroperitoneum 3. double-barreled aorta:(bleeding back into true lumen) |
|
|
list some situations that can lead to infective arteritis (5)
|
1. bacterial pneumonia
2. TB 3. intracardiac infection 4. septic microemboli 5. arterial trauma |
|
|
possible complication of infective arteritis?
|
mycotic aneurysm
|
|
|
eitologic agents, diagnosis, management of infective arteritis?
|
same as IE
MC etiologic agents = staph, strep |
|
|
MC cause of infective phlebitis?
|
IV catheters (nosocomial)
|
|
|
1. MC etiologic agents in superficial veins? (2)
2. MC etiologic agents in pelvic and portal veins? (3) 3. MC etiologic agents in intracranial venous sinuses? (3) |
1. S. aureus, Gram negative aerobic bacilli
2. Bacteroides, Streptococci, E. coli 3. H. influenzae, S. pneumoniae, S. aureus |
|
|
Symptoms of infective phlebitis?
(pelvic, portal, intracranial) |
pelvic - nausea, vomiting
portal - jaundice intracranial - headache |
|
|
What is the most common cause of myocarditis in the US?
|
infections, mainly viruses
|
|
|
name 5 bacteria known to cause myocarditis
|
1. Clamydiae
2. Rickettsiae 3. Corynebacterium diptheria 4. Neisseria meningitidis 5. Borrelia burgdorferi (Lyme) |
|
|
what is a characteristic finding in lyme disease carditis?
|
bandlike collection of lymphoid cells in endocardium
(also see lymphocytes, plasma cells, macrophages in myocardium) |
|
|
pericarditis is usually secondary - when it is primary what is the most common etiology?
|
viral
(also occasionally from TB) |
|
|
MC bacterial cause of infective endocarditis?
|
Streptococcus viridans
(followed by Staph aureus) |
|
|
most common bacteral cause of IE in IVDUs?
|
Staph aureus
|
|
|
bacteria seen causing IE in the presence of colon cancer?
|
Streptococcus bovis
|
|
|
differentiate between the symptoms seen in acute vs. subacute IE
|
acute - high fever, toxicity, death in a few days or weeks
subacute - low grade fever, night sweats, weight loss, weeks to months |
|
|
1. acute IE most often caused by?
2. subacute IE? |
1. S. aureus (MORE VIRULENT ORGANISM)
2. Streptococcus (LESS VIRULENT) |
|
|
risk factors for native valve IE (3)
|
1. congenital heart disease
2. acquired abnormalities (RF, mitral valve prolapse, degenerative valve lesions) 3. idiopathic |
|
|
most common colonizing bacteria in native valve IE?
|
S. viridans
(alpha hemolytic) |
|
|
differentiate between the types of bacterial infection seen in early vs. late prosthetic valve IE
|
early (<60 days) - most often nosocomial
late (> 60 days) - most often community aquired |
|
|
bacteria types in an early prosthetic valve IE? (2)
|
S. epidermidis
S. aureus |
|
|
bacteria types in a late prosthetic valve IE?
|
Streptococcus, HACEK group
(Haemophilus, Actinobacillus, Cardiobacterium, Eikenella, Kingella) |
|
|
most common valve infected in general IE?
|
mitral
followed by aortic |
|
|
most common valve infected in IVDU IE?
|
tricuspid
followed by aortic |
|
|
side of heart infected in IVDU IE?
|
right
|
|
|
MC infective organism in IVDU IE?
|
S. aureus (often MRSA)
|
|
|
two most common bacteria in nosocomial IE?
|
S. aureus
Enterococcus |
|
|
four things always observed in the pathogenesis of IE?
|
1. altered endothelium OR valvular abnormalities (facilitates colonization)
2. Transient bacteremia 3. Bacterial adherence (formation of vegetation) 4. Host defenses (immune complexes and compliment cause peripheral effects |
|
|
components of sterile vegitations? (2)
|
1. platelets
2. fibrin |
|
|
three clinically important pathogens belonging to the Staph family?
|
1. S. aureus (very destructive)
2. S. epidermidis 3. S. saprophyticus (seen in UTIs) **2 and 3 are coagulase negative: 1 is coag. (+) |
|
|
8 virulence factors seen in Staph?
|
1. adherence
2. resistance to host defenses 3. toxins (alpha, beta, gamma) 4. Coagulase 5. Leukocidin 6. Staphylokinase 7. Proteases 8. Superantigens |
|
|
regarding virulence factors of Staph:
1. mechamism of resistance to host defenses 2. function of beta toxin? 3. function of coagulase? 4. function of leukocidin 5. function of staphylokinase? 6. 2 examples of superantigens released by Staph |
1. capsule, peptidoglycan
2. degrades sphingomyelin 3. converts fibrinogen to fibrin 4. damages host cell membrane 5. plasminogen activator 6. enterotoxin, TSST |
|
|
are strep catalase + or -?
|
catalase negative
|
|
|
three ways to classify strep via hemolysis?
|
1. alpha hemolytic (green)
2. beta hemolytic (clear) 3. gamma hemolytic (none) |
|
|
give the species name(s) of:
1. group A strep 2. Group B strep 3. Group C strep 4. Group D strep |
1. S. pyogenes
2. S. agalactiae 3. S. equi 4. Enterococcus (E. faecalis, E. faecium) AND Non-enterococcus (S. bovis) |
|
|
two types of strains that are not Lancefield classified?
WHY are they not classified? |
S. viridans
S. pneumoniae both have no capsule |
|
|
7 virulence factors seen in all Strep?
|
1. extracellular dextran
2. adhesin 3. liptechoic acids 4. M protein 5. compliment C5a peptidase 6. Capsule 7. toxins |
|
|
regarding Strep virulence factors:
1. function of extracellular dextran? 2. function of lipotechoic acid? 3. function of M protein? 4. composition of capsule? |
1. adherence
2. bind laminin 3. prevents phagocytosis 4. hyaluronic acid |
|
|
6 toxins released by streptococci
|
1. streptolysin S (leukocidin)
2. streptolysin O (leukocidin) 3. Hyaluronidase (spreading factor, destroys CT) 4. streptokinases 5. proteases 6. pyrogenic exotixins (SPEs) |
|
|
possible damage/complications from IE? (7)
|
1. vegetations can embolize
2. persistent bacteremia 3. circulating immune complexes 4. cytokine release 5. new or changing heart murmur 6. splenomegaly 7. fever |
|
|
describe the circulating immune complexes (seen in infective endocarditis) that are deposited
|
1. Roth spots in retina
2. splinter hemorrhages 3. Osler's nodes (painful) on hands and feet 4. Janeway lesions (painless) on hands and feet 5. glomerulonephritis (with hematuria and RBC casts) 6. arthritis 7. petechiae |
|
|
two major Duke's criteria for diagnosis of IE?
|
1. 2 positive blood cultures
2. endocardial involvement |
|
|
minor Duke's criteria for diagnosis of IE? (6)
|
1. predisposing condition of IVDU
2. Fever >100.4 3. vascular phenomena 4. immunologic phenomena 5. echocardiogram 6. + blood culture |
|
|
regarding Duke's criteria in diagnosis of IE: what would constitute a definite diagnosis?
|
1. pathology of vegetations
2. 2 major 3. 1 major, 3 minor 4. 5 minor |
|
|
which abx. would be used in the treatment of staph induced IE?
|
beta lactam + gentamycin
|
|
|
which abx. would be used in the treatment of strep induced IE?
|
penicillin + aminoglycoside
|
|
|
which heart valve looks like a mercedes-benz sign?
|
Aortic valve
|
|
|
LAD - see hypertrophy and dilation as a result
|
Which coronary is occluded?
|
|
|
describe forward left sided heart failure
|
decreased contractility = decreased CO -> reduction in renal perfusion, systemic perfusion
|
|
|
describe backwards left sided heart failure
|
decreased compliance of LV places increased load on LA - LA dilates, pulmonary edema and pulmonary venous HTN common
|
|
|
MCC of R sided heart failure?
|
L sided heart failure
|
|
|
describe backwards R sided heart failure
|
decreased CO -> increased systemic venous pressures -> congestive hepatomegaly, splenomegaly,
peripheral edema, fluid accumulation in pericardial, pleural and peritoneal spaces |
|
|
myocardial adaptations to CHF? (6)
|
1. beta adrinergic stimulation
2. increased cytosolic [Ca++] 3. increased protein synthesis 4. cellular hypertrophy 5. change in protein isoforms (myosin) and oncogene expression 6. increased interstitial collagen formation |
|
|
changes in blood volume in response to CHF? mechanism?
|
- increase in volume
- increase preload - renal conservation of sodium and water |
|
|
negative aspects of increasing peripheral vascular tone in response to CHF?
|
- increase afterload and decrease perfusion
|
|
|
4 common causes of heart failure?
|
1. ischemic heart disease (>80%)
2. hypertensive heart disease 3. valvular heart disease 4. primary myocardial diseases (cardiomyopathies, myocarditis) |
|
|
in cor pulmonale:
1. look of R ventricle? |
1. dilated R ventricle with hypertrophy
|
|
|
causes of cor pulmonale?
|
1. pulmonary embolus
2. emphysema 3. pulmonary HTN 4. chronic bronchitis 5. some congenital heart disease (L->R shunts) |
|
|
major risk factors for IHD? (4)
|
smoking
DM HTN hypercholesterolemia |
|
|
minor risk factors for IHD? (7)
|
obesity
age male gender family history OCPs sedentary lifestyle personality |
|
|
regarding critical stenosis:
___ reduction in blood flow? ___ reduction in area ___ reduction in diameter |
90% reduction in blood flow
75% reduction in area 70% reduction in diameter |
|
|
which type of plaques are at a higher risk of rupture?
|
lipid rich (soft) plaques
|
|
|
in an MI: the window period is?
|
reversible time (first 30 min)
- 1-2 min: loss of contractility - 10 min: 50% depletion of ATP - sarcoplasmic and mitochondrial swelling by EM |
|
|
in an AMI: how long until irreversible myocardial damage occurs?
- mechanisms of irreversible damage? |
20-40 minutes
- loss of membrane integrity (leakage on myoglobin, LDH, creatine kinase, potassium) - chromatin clumping and disrupted sarcolemma |
|
|
gross and microscopic pathology after an AMI?
1. 24h 2. 2-3d 3. 4-9d 4. 10-14d 5. 2-4 wk 6. >4 wk |
1. G-> pale / M-> myocyte eosinophilia, contraction bands
2. G-> soft yellow tan / M-> neutrophils, nuclear diseintegration 3. G-> soft yellow tan / M-> inflammatory debris, macrophages 4. G-> yellow grey / granulation tissue, early collagen 5. firm, red-grey / scar formation 6. G-> firm, grey / mature scar |
|
|
what is Dressler's syndrome?
|
autoimmune rxn. to pericardium 6-8 weeks after an MI
see - pericarditis, fever, joint pain, friction rub |
|
|
some complications of AMI? (7)
|
1. cardiogenic shock
2. arrythmias 3. rupture 4. pericarditis 5. sudden death 6. ventricular aneurysm 7. mural thrombus and embolization |
|
|
complication that is most common cause of death after an AMI?
|
arrythmias
|
|
|
when would rupture happen as a result of an AMI?
3 MC places of rupture? |
most common 3rd-7th day
- free wall, septum, papillary muscle |
|
|
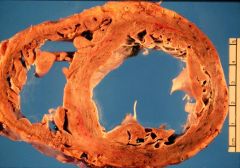
coronary arterial dissection
|
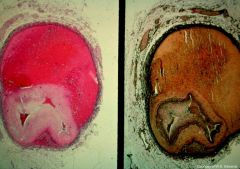
what is this?
|
|
|
18-24 hrs
no nuclei, see beginnings of coagulative necrosis |
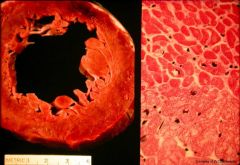
How long after an AMI?
|
|
|
when would rupture happen as a result of an AMI?
3 MC places of rupture? |
most common 3rd-7th day
- free wall, septum, papillary muscle |
|
|
know
|
know
|
|
|
know
|
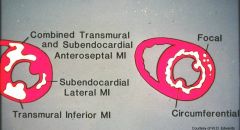
know
|
|
|
circumferential subendocardial MI
|
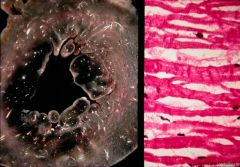
type of MI?
|
|
|
Left main coronary occlusion
HUGE MI! |
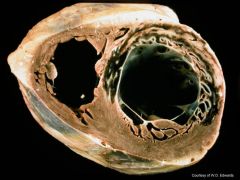
occluded vessel?
|
|
|
Right main coronary artery
-posterior 1/2 of septum occluded |
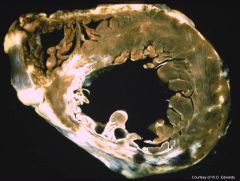
occluded vessel?
|
|
|
Right main coronary artery
-posterior 1/2 of septum occluded |
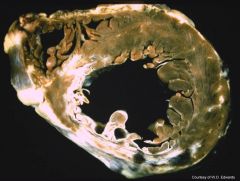
occluded vessel?
|
|
|
Four common causes of Left Sided Heart Failure?
|
1. Ischemia (>80%)
2. Aortic or Mitral Valve disease 3. Systemic HTN 4. Myocardial disease |
|
|
Five common effects of Left sided heart failure?
|
1. Dyspnea
2. Cough 3. Prerenal azotemia 4. Hypoxic encephalopathy 5. sodium overload and systemic dependent edema (hypoperfused kidneys) |
|
|
Explain the mechanism behind Paroxysmal Nocturnal Dyspnea
|
patient lies down and fluid redistributes in the body, result is pulmonary edema
(total body hypoxia also contributor) |
|
|
what causes the cough seen in left sided heart failure and what HF cause is it most commonly seen in?
|
cough results from left atrium pushing on bronchus
- most commonly seen in mitral valve disease |
|
|
Two major common causes of Right sided heart failure?
|
1. Any disease interfering with lung ventilation (Emphysema, CF, etc.)
2. Left sided heart failure |
|
|
what is the mechanism behind R sided HF due to pulmonary disease?
|
increased pulmonary vascular resistance due to fibrosis and/or the hypoxic vascular response
|
|
|
effects of R sided HF? (4)
|
1. Splanchnic congestion (hepatosplenomegaly, (+) hepatojugular reflex)
2. JVD 3. edema (from incr. venous hydrostatic pressure) 4. effusion (pleural) |
|
|
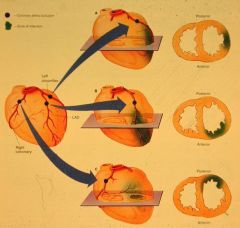
which area of the left ventricle does the Left circumflex coronary artery supply?
|
lateral wall
|
|
|
which area of the left ventricle does the LAD supply?
|
anterior wall and apex
|
|
|
which area of the left ventricle does the R coronary artery supply?
|
posterior wall
|
|
|
in hypertensive heart disease most of the cardiac changes are due to increased cardiac work. Name three changes and the general result of these changes.
|
1. cardiomegaly
2. LV hypertrophy 3. cardiac myocyte hypertrophy **leads to increased myocardial oxygen demand** |

