![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
28 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Describe 3 important functions of cholesterol. |
- Vital component of cell membranes - Precursor to steroid hormones (testerone, cortisol, etc) - Precursor to bile acids which are crucial to digestion |
|
|
|
What is cholesterol's relationship with heart disease? |
Deposition in arteries associated with atherosclerosis and heart disease. |
|
|
|
Describe the biosynthesis of Mevalonate. (3 steps) What does it ultimately become? |
1. 3 units of acetate 2. 1 unit HMG-CoA 3. HMG-CoA converted via HMG reductase to mevalonate
Part of cholesterol synthesis. |
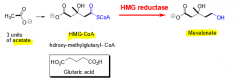
|
|
|
What is the function of HMG-reductase? |
Mediates the rate-determining step in cholesterol biosynthesis. (HMG-CoA ----HMG reductase---> Mevalonate) |
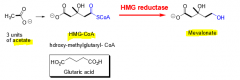
|
|
|
Describe the conversion of mevalonate to famesyl pyrophosphate (C-15). (3 steps) |
1. Mevalonate converted into C5 unites ("isoprene"). 2. Combines via carbo-cation intermediates to C10 3. Add another isoprene (C5) to give C-15. |
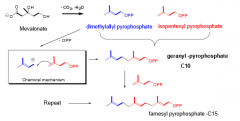
|
|
|
Describe the conversion of C15 unites to cholesterol. (3 steps) |
1. Two C15 unites are combined to give C30 (squalene) 2. Squalene is oxidized to squalene epoxide. 3. Squalene epoxide is cyclized and additional reactions convert it to cholesterol |
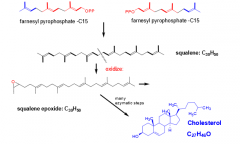
|
|
|
How does one inhibit cholesterol biosynthesis? |
Interfere with the rate determining step by inhibiting the enzyme HMG reductase.
This can be done by using a molecule with a similar structure to HMG-Co (which reductase binds to) |
|
|
|
What is mevastatin? |
- HMG-CoA reductase inhibitor - Isolated from penicillium mold - Japanese scientist Akiro Endo |
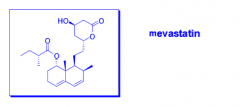
|
|
|
How does mevastatin's structure compare to HMG-CoA |
- The lactone (ester) ring is closed |
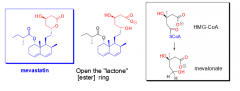
|
|
|
Why did mevastatin not become a drug? |
Showed toxicity |
|
|
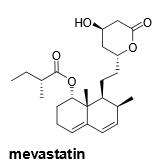
What part of the mevastatin structure is crucial to its function? Why? |
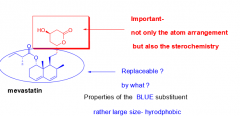
|
|
|
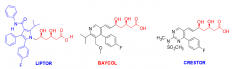
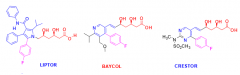
Describe the structure relationships to: Natural HMG-CoA reductase inhibitors + each other. |
- All have the open lactone ring (no longer open but cyclized like in HMG) - All have benzene ring with F attached to N-ring structure |
|
|
|
What is Lipitor used for? What kind of drug is it? How successful? |
- Statin - Used for cholesterol reduction - Best selling drug in history |
|
|
|
What is an important adverse side effect of Baycol? |
Baycol = statin Rhabdomylolsis -- skeletal muscle wasting + kidney failure - Relatively rare but caused Baycol to be withdrawn in 2001 |
|
|
|
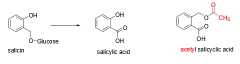
What is the active ingredient isolated from willow bark? What is it used for? |
- Salicin - Pain, fevers, inflammation |
|
|
|
By 1860, it was realized that salicin could be converted into ________ to result in more effective treatment. |
Salicylic acid Note: More side effects! |
|
|
|
What is ASA? |
Acetyl Salicylic Acid More tolerated/effective version of salicylic acid; sold as Aspirin. |
|
|
|
What are the positive effects of ASA? (4) |
- Pain relief (analgesic) - Fever reduction (antipyretic) - Reduces inflammation - Reduces blood clotting |
|
|
|
What is a negative side effect of ASA? |
Gastric lesions |
|
|
|
Describe the synthesis of ASA from a phenol. |
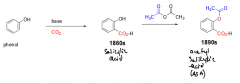
1. Phenol converted to salicylic acid with a base and CO2 2. Salicylic acid converted to ASA thru acetylation. |
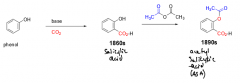
|
|
|
What is ASA's overall mechanism of action? |
- Inhibits formation of prostaglandins from archadonic acid (polyunsaturated fatty acid) - Does so by inhibiting COX |
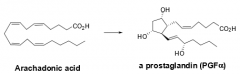
|
|
|
How is archadonic acid converted to a prostaglandin? |
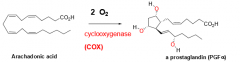
1. Reacts with O2 and COX |
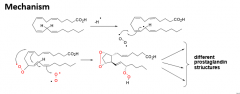
|
|
|
What is the difference between COX-1 and COX-2 enzymes? |
COX-1: Mediates formation of prostaglandins associated with renewal of stomach/esophagus lining
COX-2: Mediates prostaglandin formation associated with inflammation. |
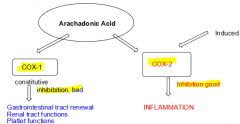
|
|
|
What is IC50? |
Concentration (of a drug) that inhibits 50% of the activity of a substance. |
|
|
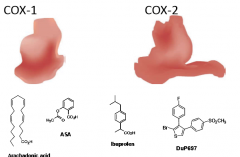
Which of the following compounds would be most selective against COX-2? |
DuP697 -- most resembles shape of COX-2 receptor. |
|
|
|
Why was Vioxx withdrawn from the market in 2004? |
- Study showed that long-term use of Vioxx for prevention of colon cancers increased risk of heart attack or stroke by factor of 2 |
|
|
|
Why was Vioxx re-introduced to the market? (2 main) |
- Benefits outweighed risks for some patients (colon cancer vs heart problems) - Cardiovascular risks for Vioxx seemed no worse than Ibuprofen |
|
|
|
Why is Celebrex considered to be 'safer' than Vioxx? |
- Vioxx more strongly inhibits CO-2 - Speculation: COX-2 does more than just mediate inflammation! May also be present in heart tissues (i.e. cardioprotective function?) |
|

