![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
87 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Topic: Biosafety
Name 3 reasons why we wear personal protective equipment |
1) Protects outer clothing for becoming of contamination 2) Prevents outer layer of clothing from introducing contamination into cultures 3) Protects from being splashed by stains, spills etc. 4) gloves protects from level 2 bacteria |
|
|
|
Topic: Biosafety
Why do we wipe down the top of benches |
Free of dust-borne microorganisms that could contaminate cultures and protects against any pathogens that could be present
Remember: >10s contact time with solution |
|
|
|
Topic: Biosafety
Why do we use electric incinerators? |
-Prevents aerosols that could increase risk of contamination during sterilization of the loop - used when working with potential pathogen in place of a Bunsen burner - wait 10 minutes for it to reach optimal sterilizing temperature - Avoid scraping the sides with loop |
|
|
|
Topic: Biosafety
Define Biosafety Level 2 |
- can cause human or animal disease, but are unlikely to be a serious hazard to laboratory workers, community or the environment - rarely cause infection --> disease - laboratory has biohazzard signs on the door, HEPA filtered class BSC, PPE and proper disposal |
|
|
|
Topic: Biosafety
What is a Class II BSC? |
- BSC = Biosafety Cabinet - provide safe working area for class 2 pathogens |
|
|
|
Topic: Biosafety
Name the 4 different type of fires and type of exinguisher used |
Types of fires: A - ordinary combustibles B - flammable liquids C - electrical fires D - combustible metals
Types of exinguisghers Water and Soda type = for type A [not C] CO2 = ABC [not D] Dry chemical = ABC |
|
|
|
Topic: Biosafety
Operation of fire extinguisher |
PASS P = point A = aim S = squeeze S = sweep |
|
|
|
Experiment 1
Aerobic Plate Count Enumeration Limitations |
1) Will not grow injured cells and thus does not grow all viable cells (this is why we use CFU) 2) Not M.O. will grow due to nutrient, oxygen, incubation temperature requirements
|
|
|
|
Experiment 1
Why do we use peptone water? |
Peptone water contains nutrients (source of C and N) Note: you want to maintain an initial dilution of 1:10 |
|
|
|
Experiment 1
Why do we add Tween 80 to the stomacher bag? |
Tween 80 solubilizes fats and spreads out the M.O. |
|
|
|
Experiment 1: Determination of Aerobic Plate Count of Bacteria from Food
What is the purpose of this experiment? |
The purpose was to determine the total number of aerobic bacteria CFU/g in room temperature extra lean ground beef |
|
|
|
Experiment 1:
What is an advantage of using the Aerobic Plate Count Method (APC method)? |
Does not grow dead cells |
|
|
|
Experiment 1
How do you find the number of Colony Forming Units (CFU) |
N = A x D where N is the number of colony forming units A is the average count per plate D is the dilution factor |
|
|

Experiment 1: Using the values in this image, calculate CFU.
|
( 70 + 66) / 2 = 68
N = 68 x 10, 000 = 680, 000 CFU/g |
Remember your counting rules, only use counts between 20-200 |
|
|
Experiment 2
Name 4 Characteristics of Coliforms |
1) Aerobic and facultative anaerobic 2) Gram-negative 3) Non-sporeforming rods 4) Fermenting lactose with acid and gas formation
Note: Coliforms do not have to be part of the Enterobacteriaceae group |
|
|
|
Experiment 2: Determination of Coliforms in Foods
What was the purpose of this Experiment |
The purpose of this experiment was to determine the number of coliforms in a hamburger sample using presumptive and confirmed tests using the Most Probable Number and Violet Red Bile Agar Method |
|
|
|
Experiment 2
What does the MPN tell us? |
The MPN tells us the number of coliforms per 100mL (or 100g) of test material corresponding to the number of gas-postive tubes in the coliform test |
|
|
|
Experiment 2:
What does the VRBA method tell us? |
The VRBA method estimates the number of viable coliforms per g of product (CFU) |
|
|
|
Experiment 2:
What are the advantages of using the MPN method? |
MPN method is useful when there are low concentrations of lactose fermenting organisms in a sample |
|
|
|
Experiment 2:
What are the advantages of using the VRBA method? |
We are able to distinguish between different microorganisms that exist in a sample which can be seen metabolically or through morphology |
|
|
|
Experiment 2:
What are the disadvantages of using the MPN method? |
1) More statistical variability because only 5 tubes are looked at for gas production (small sample size) 2) Cannot distinguish bacteria type as only gas information can be collected |
|
|
|
Experiment 2:
What are the disadvantages of using the VRBA method? |
VRBA method may not completely inhibit Gram-positive organisms and will allow growth of other Gram-negative bacilli other than Enterobacteriaceae |
|
|
|
Experiment 2:
We use LST broth for the presumptive MPN of coliforms. What does LST stand for and list it's characteristics. |
LST = Lauryl Sulfate Tryptose Broth
1) Contains sodium lauryl sulfate which is selective for coliforms 2) Contains lactose, which is utilized by fermenting MOs to produce acid and gas |
|
|
|
Experiment 2:
What does VRBA stand for? Name the components of the media which allows it to select for coliforms. |
VRBA = Violet Red Bile Agae
1) Contains bile salts that inhibit Gram-positive but can grow fermenting bacteria that are not coliforms (Gram-negative lactose fermenters) |
|
|
|
Experiment 2:
Why do we overlay the VRBA plate with 2 x VRBA? |
Used during the presumptive test as an overlay to create anaerobic conditions. |
|
|
|
Experiment 2:
What does BGLB stand for?
What components make it selective for coliforms?
Is this media used for the confirmation test for both the MPN and VRBA method? |
BGLB = Brilliant Green Lactose Bile
Contains oxbile and brilliant green which inhibit Gram-postive and many Gram-negatives (other than coliforms) and allows lactose fermentation
Yes, this media is used for the confirmatory tests in both methods of experiment 2. |
|
|
|
Experiment 2:
Why do we multiply the MPN/100 g number from the table by 10? |
We only used 1/10 of the amount of hamburger in the analysis, so we multiply by 10 to get the actual per 100g of test material |
|
|
|
Experiment 2:
How much gas in the Durham tube is considered to be a positive result? |
10% gas |
|
|
|
Experiment:
What is the formula to calculate the MPN? |
MPN per 100 mL = number of M.O x dilution factor of the middle set of the dilutions |
|
|
|
Experiment 2:
How do we determine the confirmed number of coliforms using the VRBA method? |
Confirmed CFU/g = % of BGLB positive tubes x presumptive # |
|
|
|
Experiment 2:
What would be the expected results of E. aerogenes, Staphylococcus, E.coli and salmonella for VRBA, LST broth and BGLB broth? |

|
|
|
|
True or false?
For calculating MPN, use the dilution factor of the middle of the set of the three dilutions |
True |
|
|
|
True or false?
For MPN, if only 3 dilutions were made, use the results of those 3 dilutions to compute the MPN. |
True |
|
|
|
True or false?
If more than 3 dilutions are used, use only the results of 3 consecutive dilution.
Select the highest dilution in which all 5 tubes are positive and the 2 subsequent higher dilutions |
True |
|
|
|
True or false?
If more than 3 dilutions are made for the MPN method have all of 5 of the tubes +ve, use the first 3 dilutions. |
True |
1 - 5 positive 0.1 - 5 positive 0.01 - 4 positive 0.001 - 0 positive
|
|
|
True or false?
If a positive tube occurs in the dilution higher than the 3 chosen to the rule, the number of such positive tubes are added to those of the next lower dilution |
True |
1 - 5 positive 0.1 - 4 positive 0.01 - 1 positive 0.001 - 1 positive |
|
|
True or false?
If the tubes of all sets of a dilution series are positive, choose the 3 highest dilutions of the series used and indicate by using a ">" that the MPN is larger than what was calculated |
True |
1 - 5 positive 0.1 - 5 positive 0.01 - 5 positive 0.001 - 5 positive |
|
|
Experiment 3: Determination of Staphylococcus in Foods
What was the purpose of this experiment? |
The purpose of this experiment was to determine the number of CFU of coagulase positive S. aureus in a sausage sample. This was achieved by using an enumeration presumptive test in which BPA selected for S. aureus and was confirmed using a coagulase test to determine if the strain is capable of producing enterotoxins. |
|
|
|
Experiment 3:
What are enterotoxins? |
A protein that is excreted extracellulary which target epithelial cells of the intestinal cell wall |
|
|
|
Experiment 3:
Name 3 reasons why we examine foods for S. aureus |
1) To confirm that this organism may be the causative agent of food borne illness 2) to determine whether a food or food ingredient is a potential source of of Staphylococcal food poisoning 3) To demonstrate post-processing contamination, which is usually due to human contact with processed food or inadequate sanitization practices
|
|
|
|
Experiment 3:
True or false?
The presence of large numbers of S. aureus in foods is not sufficient enough to incriminate a food vector of food poisoning |
True |
|
|
|
Experiment 3:
Name 5 characteristics of S. aureus. |
1) Ability to grow in the presence of specific concentrations of selectively toxic chemicals; can withstand high salts 2) Form and appearance of colonies 3) Microscopic morphology 4) The capacity of metabolites produced by S. aureus to hydrolyze substrates like egg yolk or DNA 5) Production of a substance that coagulates the plasma of humans and other species |
|
|
|
Experiment 3:
What does BP agar stand for?
What are the advantages of using this media? |
Baird-Parker agar
Adv: 1) Allows heat-damaged staph and inhibits coagulase-negative cocci 2) high [salt] 3) Telleride enrichment resulting in a colouration of black/grey 4) contains yolk enrichment (hydrolyzed by S. aureus) |
Black halos are most important. What causes this to occur? |
|
|
Experiment 3:
What are the limitations of using BP agar? |
1) Large variation of colony sizes, easily missed 2) A. faecalis, P. vulgaris and Bacillis look phenotypically similar on telluride media 3) Clearing zones + yolk precipitate are not identifiabe characteristic of S. aureus |
|
|
|
Experiment 3:
How do you calculate the presumptive S. aureus on BP Agar? |
Presumptive CFU = average colonies x 5 x dilution factor |
We multiply by 5 because we used 0.2 mL and not 1 mL of the sausage solution. |
|
|
Experiment 3:
Why was a simple stain conducted? |
A simple stain was conducted to determine the morphology. Only cocci were inoculated into BHI for the confirmatory test. |
|
|
|
Experiment 3:
What does BHI stand for?
What are the advantages and limitations of using this media? |
Brain Heart Infusion Broth
Suitable for the cultivation of fastidious strains of bacteria
Due to nutritional variation, some strains may be encountered that grow poorly or fail to grow on this medium |
|
|
|
Experiment 3:
Describe the advantages and limitations of the coagulase confirmatory test? |
Adv: coagulase result of 4+ indicates the identification of S. aureus
limitations: Only isolates with 4+ clotting results in identification.
Isolates may be weakly-coagulase producing strains or a different type of Staphylococcus |
|
|
|
Experiment 3:
Name a few additional tests that could be conducted to confirm or reject S. aureus identification (that got a 1+ to 3+ coagulase result). |
Nuclease production Lysostaphin sensitivity Accuprobe Enterotoxin assay Latex aggulatination Rapid ID test |
|
|
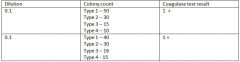
Example: Calculate the presumptive and confirmed CFU
|
1030 CFU presumptive S. aureus
0 CFU confirmed S. aureus |
|
|
|
Experiment 4: Isolation and identification of Salmonella from foods
What was the purpose of this experiment? |
- isolate Salmonella to determine its presence in poultry - achieved through a series of steps including the pre-enrichment step, selective enrichment, selective plating, purification, biochemical screening and serological identification - selective enrichment broths, SC and TBG, and selective plating agards, BS and BGS, were used to isolate salmonella based on metabolism - biochemical screening and serological identification was conducted for final confirmation |
|
|
|
Experiment 4:
Why do we have a Pre-enrichment step? |
Non-selective media is used to allow damaged cells to grow and increase the detection of Salmonella
Fatty foods are a problem, so we use a wetting agent anion tergitol to dispurse and emulsify the fat |
|
|
|
Experiment 4:
Why do we use Selective enrichment?
What medias are used? |
Selective enrichment is influenced by the proportion of inoculum to broth and the duration and temperature of the incubation period
Other M.O.s are capable of competing with Salmonella, so sub-culturing into fresh medium increases yield of Salmonella
Selenite cysteine and tetrathionate brilliant green broth |
|
|
|
Experiment 4:
Why do we use selective plating?
What medias are used and why? |
Bismuth sulfite and Brilliant Green Sulfa agar
Presumptive salmonella should be lactose dependent, except in bismuth sulfite which responds to H2S production |
|
|
|
Experiment 4:
Why is MacConkey Agar used for purification?
Wns?hat are the advantages and limitatio |
- Selective for Gram-negative bacteria and differential for entero bacteria - Contains Natural Red dye which turns red when lactose is utilized
- salmonella appears colourless as it does not use lactose or sucrose |
|
|
|
Experiment 4:
What does TSI stand for?
What is being tested with TSI media? |
Triple sugar agar
Dextrose (Butt) - turns yellow when +ve
Lactose/sucrose (slant) - turns yellow +ve
Iron - blackening; H2S when +ve
Gas formation - fermentation +ve |
|
|
|
Experiment 4:
What does LIA stand for?
What is being tested with LIA? |
Lysine Iron Agar
Lysine decarboxylase - butt remains purple when +ve, turns yellow -ve
Lysine deaminase activity - slant turns wine red +ve; unchanged -ve
H2S production, positive |
|
|
|
Experiment 4: What is being tested with urea broth? |
Positive reaction, slant turns red
bacteria with protease activity may cause a positive reaction |
|
|
|
Experiment 4:
Selenite cystine
which step? Limitations? Adv? Salmonella? |
Selective enrichment Inhibits Gram-positive and most enteric Gram-negative bacilli
Salmonella is gram-negative and is able to grow |
|
|
|
Experiment 4:
Tetrathionate Brilliant Green Broth
Which step? Limitations? Adv? Salmonella? |
Selective enrichment contains ox-bile and brilliant green which inhibit Gram-positive
Salmonella is Gram-negative and able to grow |
|
|
|
Experiment 4:
Bismuth Sulfite
Which step? Limitations? Adv? Salmonella? |
Selective plating Inhibits Gram-positive and most enteric Gram-negative bacilli
Salmonella produces H2S P. vulgaris can be distinguished as green with dark halos |
|
|
|
Experiment 4:
Brilliant Green Sulfa Agar
Which step? Limitations? Adv? Salmonella |
Selective plating
Contains Sodium sulfapyrine and brilliant green which inhbit Gram-positive and some Gram-negative
Phenol red makes Salmonella appear purple-pink (because they do not use the lactose) |
|
|
|
Experiment 4:
MacConkey Agar
Which step? Limitations? Adv? Samonella? |
Purification Selective for Gram-negative bacteria and differential for enteric bacteria
Turns red from Natural red indicator during fermentation
Samonella appears colourless as it is unable to utilize lactose and sucrose |
|
|
|
Experiment 4:
Triple Sugar Agar
limitations? salmonella? |
False positives for lactose and sucrose test because of low acid and oxygen environment as media contains x 10 more dextrose
Salmonella uses dextrose, but not lactose/sucrose, Has H2S production and no gas |
|
|
|
Experiment 4:
Lysine Iron Agar
Salmonella? |
Produces H2S, has lysine decaboxylase activity and no lysine deaminase activity |
|
|
|
Experiment 4:
Urea Broth
Salmonella? |
Salmonella does not have urease activity. |
|
|
|
Experiment 4:
What are the advantages and disadvantages of using serological testing for identification of Salmonella? |
Positive result - agglutination Somatic and flagellar antiserum
Able to react with serovars listed in the Kauffman-White Schema, but do not react with all serovars.
Possible to get false-positive results. |
|
|
|
Why do we use sterilization? |
Sterilization is a process that destroys or inactivates living organisms on an item.
it is important as the media and equipment that we use may contaminate samples thus affecting final results |
|
|
|
Name some sterilization methods.
Which process is most commonly used in the Biology department and why? |
Moist and dry heat, ethylene oxide, ionizing radiation and filtration
Moist Heat more commonly used in the department because it is more effective in killing MO than dry heat. |
|
|
|
True or false.
An autoclave performs sterilization process. |
True.
The autoclave uses high pressure saturated steam at 121 degrees Celcius.
Even will kill endospores (depends on duration of the cycle)
Decimal reduction in numbers occurs in 4-5 mins. |
|
|
|
What is Tyndallization? |
Steam at atmospheric pressure for 30 minutes for 3 days
New vegetative cells are killed from the second heating, the third heating is for safety.
Sterilizes sugar solutions and elemental sulfur. |
|
|
|
True or false
Bismuth Sulfate is boiled for sterilization as autoclaving would destroy selective characteristics |
True
Any spores that may be present may not be killed, but the medium will select against Gram-positives and will not germinate |
|
|
|
True or False
Most autoclaves are the downward-displacement type, and depend on the difference in density between the air and steam. |
True |
|
|
|
Name the three cycles of an autoclave |
1) Slow exhaust (liquid cycle) 2) fast exhaust (dry cycle) 3) fast exhaust with dry (wrapped goods)
They vary in exhaust portions of the cycles |
|
|
|
If you want to prevent boil-over of media in the autoclave, which cycle should you put it on? |
Slow exhaust |
|
|
|
If you want to prevent filters from cracking during sterilization, which cycle should you put it on? |
Slow exhaust |
|
|
|
When do we use fast exhausting cycles on the autoclave? |
When we don't mind if evaporation of liquid will occur Ex, used plates, tubes |
|
|
|
True or False?
Fast and dry cycles are the same as fast exhaust, except that a drying period occurs after the exhaust |
True
Used for supplies that are wrapped |
|
|
|
True or False
Ethylene oxide is explosive by itself and leaves residues that are mutagenic. |
True.
It is effective against all vegetative cells and spores |
|
|
|
Gaseous formaldehyde is a good surface and space steriliant that is useful against... |
bacteria (including intracellular pathogens like rickettsiae), fungi, viruses, insects and animal life. |
|
|
|
Name a disadvantage for using formaldehyde as a disinfectant. |
sterilized areas have to be neutralized with ammonia gas to minimize personnel exposure |
|
|
|
What are the limitations of using radiation for sterility? |
- low penetrance of electrons - UV has limited germicidal use because of poor penetration - Gamma rays have better penetration and are used for heat sensitive items - Vegetative bacteria are most sensitive to Gamma rays, followed by molds, yeasts, endospores and viruses |
|
|
|
True or false?
Filtration is used for oil emulsions or for heat-sensitive substances. |
True |
|
|
|
How does filtration sterilization work? |
liquid is passed through a sterile filter into a sterile vessel.
The filter traps MOs in the pores |
|
|
|
Describe depth filters |
- fibrous or grandular - flow of channels that have a continuous structure
|
|
|
|
Describe membrane filters |
- retention of contaminants due to absorption and mechanical entrapment in the matrix (based on pore size) - used to sterilize media |
|
|
|
Compare routine and investigational sampling |
Routine: detecting levels of contamination that may require action and may lead to further examination
Investigational: conducted under many different circumstances in which subjective judgement depends on the number and factors which contribute to the amount of hazard |
|

