![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
68 Cards in this Set
- Front
- Back
|
Lipids
|
- have a methyl (CH3) group and hydroxyl (-OH) on either end
- are either completely or partially hydrophobic |
|
|
Fatty Acids
|
- simplest lipid
- hydrophobic (due to it being most hydrocarbon w/ a carboxyl group) - sometimes they can have double bonds, which therefore changes structure and therefor function |
|
|
Saturated Fatty Acids
|
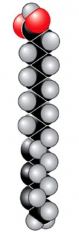
- has no carbon-carbon double bonds, which allows them to have the maximum amount of hydrogens
|
|
|
Unsaturated Fatty Acids
|
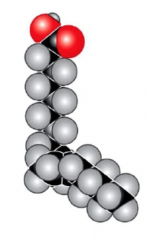
- has one or more carbon-carbon double bonds
- the double bonds mean its structure is in a plane shape, making it more rigid - main function is to provide energy |
|
|
Two Possible Double Bonds of Unsaturated Fatty Acids
|
Cis and Trans
|
|
|
Cis Unsaturated Fatty Acid
|
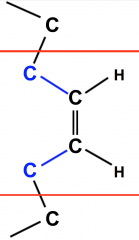
- carbons connected to the double bonded central carbons on same side, with hydrogens on the other side
- found in nature |
|
|
Trans Unsaturated Fatty Acid
|
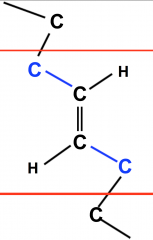
- carbons are found on either side of the double bonded central carbons, with the hydrogens mirroring the same way
- are a by product of partial hydrogenation, such as fine processing |
|
|
Omega Number
|
- tells the position of the first double bond in a fatty acid
you count the carbons that aren't double bonded starting from the methyl end, entail you reach the first double bonded carbon Omega 3's (which are unsaturated) reduce risk of heart disease by lowering cholesterol |
|
|
Dietary Recommendations
|
- consume unsaturated fats (omega-3), not saturated (omega-6)
- eat LESS saturated and trans fats, and MORE mono and poly unsaturated fats |
|
|
Fats
|
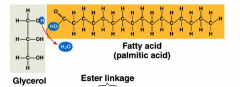
- made from 3 fatty acids and 1 glycerol (which is an alcoh-ol)
- joined by the means of dehydration reactions - are hydrophobic |
|
|
Functions of Fats
|
1. store/provide energy (stores the fatty acids, which makes fat)
2. cushion organs 3. insulation |
|
|
3 Energy Nutrients
|
carbs 4 Kcal/g
protein 4 Kcal/g fat 9 Kcal/g Fats provide more energy than carbs and proteins combined! |
|
|
Saturated Fats
|
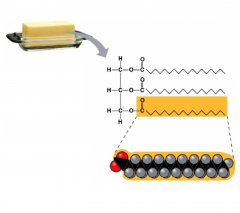
- contain saturated fatty acids, aka animal fats
- solids at room temperature, because it is tightly packed due to no cis double bonds - since they have saturated fatty acids, they do not have double bonded carbons |
|
|
Unsaturated Fats
|
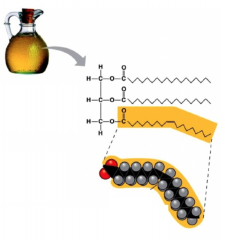
- contain at least 1 unsaturated fatty acid, aka plant and fish fats
- liquid at room temperature (oil), due to its looser packed form because of cis double bonds - since they have unsaturated fatty acids, they contain double bonded carbons, which gives it the kink |
|
|
Partial Hydrogenation
|
- makes vegetable oil more solid
ex: margarine and shortening - produces trans fats as a by product |
|
|
Cis Fatty Acid
|
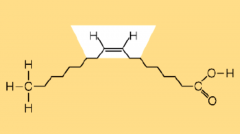
|
|
|
Trans Fatty Acid
|
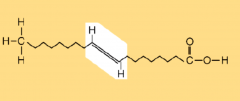
|
|
|
Heart Disease
|
- #1 cause of death in US
- includes all diseases of heart and blood vessels - caused by atherosclerosis, which is the accumulation of plaques on the inner walls of the arteries |
|
|
LDL
|
low density lipoproteins
- transports cholesterol from liver to other cells - causes plaques |
|
|
What increases LDL cholesterol?
|
1. dietary cholesterol
2. dietary saturated fats 3. trans fats |
|
|
Phospholipids
|
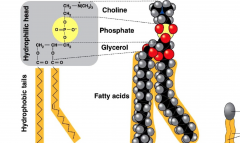
- surrounds the cell, with hydrophobic tails and a hydrophilic head
|
|
|
What are phospholipids made from?
|
2 Fatty Acids
1 Glycerol 1 Phosphate 1 Additional Chemical |
|
|
Phospholipids in water
|
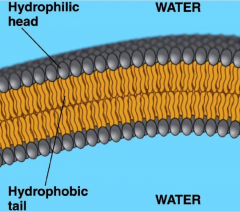
a film could develop on the surface, sometimes a second layer forms underneath to protect, making a barrier surrounding the cell
- joins tail to tail with heads on the outside as protection |
|
|
Steroids
|
- a type of lipid
- hydrophobic |
|
|
Structure of Steriods
|
4 rings
Ex: cholesterol (an alcoh-ol) |
|
|
What does cholesterol make?
|
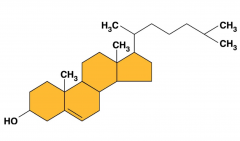
- steroid hormones
- bile: made in liver stored in gallbladder , consumes/emulsifies fats, since it is like a phospholipid it can break up fats like in the intestines - vitamin D: one fat soluble vitamin, hydrophobic (vitamin C and B are water soluble). Used to make coenzymes - also in cell membrane |
|
|
Proteins
|
- are built from 20 different amino acids (polymers and monomers)
- are one or more polypeptides, folded into a specific conformation - always have 'pep' in name |
|
|
Functions of Proteins
|
1. energy/carbon source
2. storage 3. structural support 4. intercellular signaling 5. movement 6. transport 7. defense 8. enzymes |
|
|
Two Functional Groups in Amino Acids
|
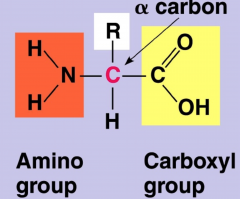
amino group, and carboxyl group, which is then attached to a central carbon and a hydrogen atom
- it's a variable group = "side chains" (20 different ones) - the amino acids are charged, but R groups can change its charge so ignore both carboxyl and amino groups when looking at it's charge, and only look at the R group |
|
|
Amino Acid Examples
|
1. alanine: hydrocarbon
2. serine: carboxyl 3. aspartic: acidic R group, negatively charged |
|
|
Three R Groups
|
1. polar: uncharged R group, but polar (ex: hydroxyl)
2. charged: has a full positive or negative charge on R group 3. nonpolar: uncharged and nonpolar |
|
|
Polar R Group
|
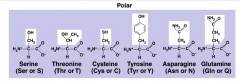
7 in total
|
|
|
Charged R Group
|
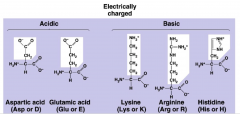
5 in total
2 Acidic 3 Basic DNA is negative, therefore an acid |
|
|
Nonpolar R Group
|
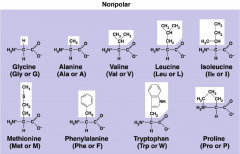
9 in total
|
|
|
Polypeptide
|
- contains many different amino acids, with different ones having different sequences
- most complex of all known molecules - formed by peptide bonds - no positive or negative charges - always an -OH on the end |
|
|
Polypeptide Backbone
|
1. made of a -N-C-C from each amino acid
2. Oxygens of -C=O 3. Hydrogens of -N-H (good for hydrogen bonding) 4. N and C termini - the C&H bond is non polar |
|
|
Polypeptide Size
|
- 129 amino acids; 14,400 Daltons
|
|
|
Why Protein Folding?
|
- it gives it its shape when in a cell
|
|
|
Lysozyme
|
- hydrolyzes the peptidoglycan cell wall (saliva)
|
|
|
Amino Acids → Proteins
|
- the process involves 3 levels of structure
|
|
|
Primary Structure
|
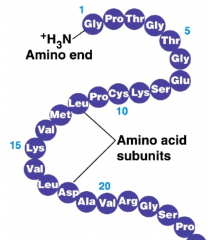
- determines which amino acids are present and in what order, ultimately determining structure
- results from covalent bonding b/w amino acids - the order of amino acids are determined by the order or nucleotides in its gene - a protein folds in a particular way, based on primary structure! - structure of polypeptide → covalent bonding |
|
|
Secondary Structure
|
- characterized by local protein folding
- results from h-bonding between backbone atoms (O and H), holds it all together - found in all proteins, the backbone atoms hold it all together - localized folding → hydrogen bonding |
|
|
Two types of secondary structure
|
1. coils (alpha helices)
2. sheets (beta pleated sheets both founded by linus pauling |
|
|
Alpha helices
|
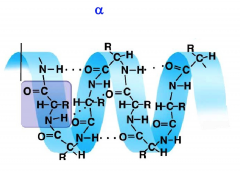
C-C-N backbone
|
|
|
Beta sheets
|
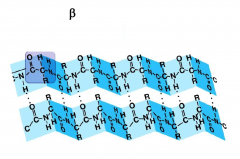
C-C-N backbone
|
|
|
Tertiary Structure
|
- overall 3D structure formed by a polypeptide
- has both alpha helices and beta sheets - R groups hold it all together, by the means of all 4 weak interactions - the nonpolar amino acids are found in the core of the folded protein, b/c it wants the nonpolar in its folds, where they cluster in the interior of the proteins - they interact with each other by hydrophobic interactions -overall conformation → all 4 weak interactions |
|
|
Disulfide Bonding/Bridges
|
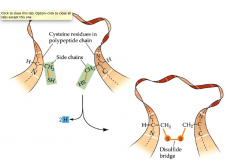
- two cysteines in a protein can become covalently bonded
- (the only polar one to do this) - this provides extra stability - excreted proteins have disulfide bonds |
|
|
Protein Folding
|
- occurs spontaneously based on primary structure
|
|
|
Environmental Factors affecting Conformation
|
- water
-pH - temperature |
|
|
Denature
|
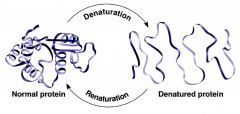
unfolding of protein in effect of extreme environmental factors
|
|
|
Renature
|
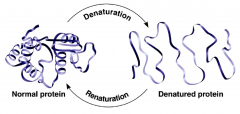
refolding of protein (usually by cooling)
|
|
|
Lysozyme
|
1 polypeptide
|
|
|
Hemoglobin
|
4 polypeptides
|
|
|
Quaternary Structure
|
- more than 1 polypeptide coming together
- held together by all 4 weak interactions and disulfide bonds |
|
|
Protein
|
- have tertiary and/or quaternary structure
- can be denatured and renatured |
|
|
Functions of Nucleic Acids
|
1. information storage (genes) → polynucleotides
2. enzymes → polynucleotides 3. molecular adaptor → polynucleotides 4. energy source (ATP) → nucleotides 5. cell signaling (2nd messengers) → nucleotides 6. chemical runs (coenzymes) → nucleotides |
|
|
Nucleotides
|
- 4 different ones (including monomers)
- pentose sugar (vary based on 2nd carbon) - at least 1-3 phosphates - a nitrogen base (rings, pyrimidine and purine) |
|
|
Purines
|
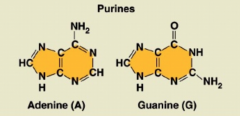
2 rings; adenine and guanine
|
|
|
Pyrimidines
|
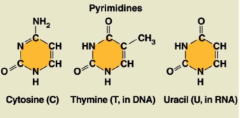
1 ring; cytosine, thymine, and uracil
|
|
|
Structure of Nucleotides
|
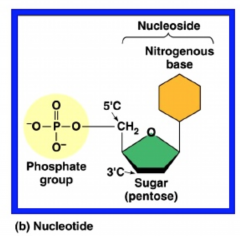
- the nitrogen bases are connected to the first carbon on the sugar, the phosphate group is attached to the 5th carbon, and if more than 1 phosphate group, it'll attach to the other phosphate groups
|
|
|
Types of Pentose Sugars
|
Ribose and Deoxyribose
|
|
|
Deoxyribose
|
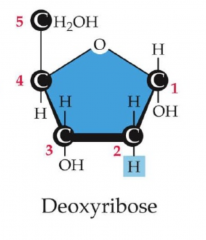
has just an H
|
|
|
Ribose
|
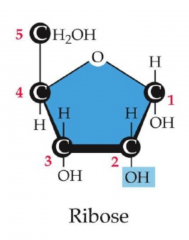
has an OH
|
|
|
Polynucleotide
|
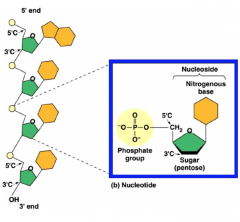
- contains different nucleotides
- different ones have different sequences |
|
|
Polynucleotide Backbone
|
1. -sugar-P from each nucleotide
2. negatively charged 3. two ends, 5' and 3' - repeating units of sugar→ phosphate - on the hydroxyl group, another polynucleotide can be added (the 3' end) |
|
|
Difference Between DNA and RNA
|
Thymine → DNA
Uracil → RNA Ribose → RNA Deoxyribose → DNA RNA = 1 chain DNA = 2 chains, held together by H bonding |
|
|
Gene
|
a unit of inheritance consisting of a region of DNA
- the order of nucleotides in a gene determines the order of amino acids in a protein, aka its primary structure |
|
|
DNA→RNA→Protein
|
- DNA and RNA remain intact the whole process to a protein (they're just copied by the gene)
|

