![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
23 Cards in this Set
- Front
- Back

|

|
|
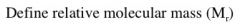
|
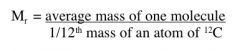
The sum of the relative atomic masses of each of the atoms in a molecule.
|
|
|
What does the avagadro number mean?
|
The number of particles in one mole of substance. 6.022 x 10^23
|
|
|
What do the same numbers of moles of different substances have in common?
|
The same number of particles.
|
|
|
Define the mole.
|
The number of particles in 12g of Carbon-12.
|
|
|
Define the term molar mass.
|
Mass per mole of substance (kg mol^-1). Same numerically as M^r.
|
|
|
What is the formula for calculating moles (n)?
|

|
|
|
What is the ideal gas equation?
|

|
|
|
Define the term empirical formula.
|
The simplest, whole number, ratio of elements in a compound.
|
|
|
Define the term molecular formula.
|
The exact number of atoms of each element in the formula of a compound.
|
|
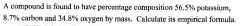
|

|
|
|
How do you find the molecular formula?
|
Relative molecular mass/relative mass of empirical formula.
|
|

|

|
|
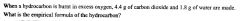
|

|
|

|

|
|

|

|
|

|

|
|
|
What is the formula for calculating concentration.
|

|
|
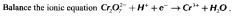
|

|
|
|
What is the formula for calculating atom economy?
|

|
|
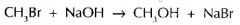
Calculate the atom economy for the methanol in the following equation.
|

|
|
|
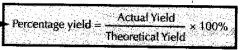
What is the formula for calculating percentage yield?
|
|
|
|
State the basic assumptions about ideal gas equation.
|
>Made up of identical particles in continuous random motion
>Particles do not react when they collide >Collisions between particles are perfectly elastic >Particles have no intermolecular forces |

