![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
16 Cards in this Set
- Front
- Back
|
What are three factors that the attractive force depends on?
|
1. Distance
2. charge on ions 3. magnitude of the diapole |
|
|
What type of intramolecular force occurs between oxygen and water molecules?
|
diapole/induce diapole
|
|
|
What type of intramolecular force occurs between i2 and CH4?
|
Induce-Induce
|
|
|
What intramolecular force is found in almost all chemical bonding?
|
London dispersion force
|
|
|
List the intramolecular forces from strongest to weakest.
|
Ion-diapole, diapole-diapole, hydorgen bonding, diapole-induce diapole, induce diapole-induce diapole
|
|
|
The higher the mass the ____ the boiling point.
|
higher
|
|
|
The point at which there is no distinction between liquid and gas is no as the ______
|
critical point
|
|
|
The point at which all three phases coexist is known as the _______.
|
triple point
|
|
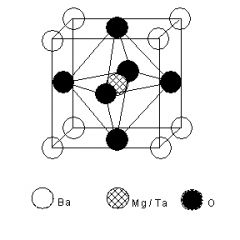
Base on this unit cell, what is the formula?
|
BaMg03
|
|
|
What are some characteristics property of pure crystalline solids?
|
1. melt at specific temperature
2. form well-define crystals with smooth flat faces |
|
|
What are some characteristics property of pure crystalline solids?
|
1. melt at specific temperature
2. form well-define crystals with smooth flat faces |
|
|
what phase will this chemical be at 50 psi and 150 degree Fahrenheit?
|
liquid
|
|
|
What are some characteristics property of pure crystalline solids?
|
1. melt at specific temperature
2. form well-define crystals with smooth flat faces |
|
|
What phase will this chemical be if it is at 50 psi and 135 degree fahrenheit
|
liquid
|
|
|
what cause the water molecules to form circular droplets?
|
The cohesive force cause by hydrogen bonding.
|
|
|
What are 3 common cell symmetries?
|
1. simple cubic
2. body center cubic 3. Face-centered cubic |

