![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
21 Cards in this Set
- Front
- Back
|
Clarify Hazards |
|
|
|
Define Hazard |
Hazard is the potential for harm from a chemical, biological or physical agent. Itis the intrinsic capability of an agent to cause an adverse effect on health. Theeffect may develop within a short period or may not show for many years. |
|
|
Risk |
Risk is the probability of an adverse effect occurring. It is the likelihood of harmoccurring under the actual circumstances of use. This depends on the degree towhich people are exposed to the hazard and the amount that is absorbed into thebody (the dose). |
|
|
Anticipation, recognition, evaluation and control |

|
|
|
Routes of entry into the body |
If it is impossible for the hazardous substance to enter the body, there is usuallyNO risk to health. In other words, the initial questions we must ask are Can the substance be inhaled into the lungs as a gas, vapour, fume or dust? Can the substance be ingested (eaten/swallowed) and absorbed from thedigestive tract? Can the substance be absorbed through the skin into the bloodstream or causeirritation to skin by direct contact? |
|
|
Physical forms of hazardous substances - how may they exist in the workplace |

a solid (including dusts and fumes) a liquid (including airborne sprays, aerosols and mists), or a gas (including vapours). |
|
|
Assessing risk |

|
|
|
Monitoring strategy Initial appraisal Basic survey Detailed survey |
Initial appraisal Normally follow walk through. May need a hand held direct reading vapour detector. If peak levels low - go no further if higher basic survey or detailed survey Basic survey Includes measurement of worker exposure using personal monitoring techniques. Samples over full shift. If results of monitoring low, below 1/4 exposure limit unlikely detailed ix needed Detailed survey\ If exposures elevated (1/2 exposure standard) more detailed ix warranted. Personal sampling may be supplemented by static (fixed position) sampling. A larger proportion of exposed population would be monitored for more than one shift. |
|
|
Measuring gases and vapours Define True gases examples Common vapours How to detect |
Gases exist as molecules in the environment over a wide range of temperature and pressure Examples: Hydrogen Carbon monoxide Methane Sulphur dioxide Ozone Traditionally measured by bubbling air through a liquid absorbent solution that is held in an impinger (or bubbler) which is taken back to the lab Commonly acquired vapours: Mercury Isocyanates Solvents (cleaners, degreaser, fuels, alcohols) Common method for organic compounds is to draw air through a glass tube packed with activated charcoal or silica gel which absorbs the sample ready for laboratory analysis, which is commonly gas chromatography (GC) or high pressure liquid chromatography (HPLC) |
|
|
Indicator tubes - alternative monitoring |
snap shot Draw backs Tubes are specifically made for each contaminant, which requires you tohave the right tube available. (You can buy them at short notice). They are of limited accuracy, usually quoted as about ± 20% of the truelevel. They may be cross sensitive to other chemicals in the environment (thecolour development is based on fairly crude chemistry which reduces thespecificity of the tube). They have a relatively short shelf life, sometimes only 6-12 months. Tubes are not available for all contaminants. |
|
|
Measuring particulates |
Dusts - particles up to 100microm in diameter Fumes are <1microm in diameter and produced by condensation of vapour or less commonly chemical reaction Particulates (dusts) drawing air through a filter that collects the particles for subsequent analysis. Weigh the deposit which leads to total inhalable particulate More toxic species (heavy metals) are determined by laboratory analysis (atomic abrorptin) <7microm may be deposited in lungs - respirable fraction Cyclone is used for dusts like silica |
|
|
Air sampling and the law CAD COSHH |
CAD - Chemical agents directive The CAD sets out a framework comprising two types of values: 1 Indicative occupational exposure limit values (IOELV’s), 2 Binding occupational exposure limit values. In the UK, the COSHH Regulations state that exposure of employees should be monitored: To demonstrate that adequate control of exposure is being maintained Whenever it is required to protect the health of employees For certain substances listed in the Regulations such as vinyl chloride andchromium from plating baths. |
|
|
OELs Occupational exposure limits |
Upper limit on acceptable concentration of hazardous substance in workplace air May be set by national bodies, or self imposed |
|
|
Occupational exposure limits in the UK Workplace exposure limits (WELs) |
Occupational exposure limits set by COSHH in order to help protect health of workers WELs are concentrations of hazardous substancesin the air, averaged over a specified period of time referred to as a time-weightedaverage (TWA). Two time periods are used: long term (8 hours) and short term(15 minutes). Short-term exposure limits (STELs) are set to help prevent effects,such as eye irritation, which may occur following exposure for a few minutes |
|
|
Older limits OES and MEL OES - occupational exposure standards MEL - maximum exposure limits |
The key difference between the two types of limit was that an OES was set at alevel at which there was no indication of risk to health; for an MEL, a residual riskmay have existed and the level set took socio-economic factors into account. Mel set if no-adverse-effect-level could be established and where effects of overexposure (known carcinogens) serious Changed to WEL due to concerns if used and if understood |
|
|
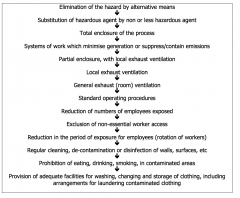
Hierarchy of controls |
|
|
|
Ventilation General exhaust ventilation |
GEV known as dilution ventilation Removes contaminated air from room and allows clean air to enter thereby diluting pollutant levels Only adopted when pollutants are low toxicity and release rate is constant Found in laundries (remove heat and vapour) Office remove people ordours Workshops prevent build up of welding fumes/solvent vapours Limited use when people exposed directly to hazardous materials More value is LEV |
|
|
Ventilation Local exhaust ventilation |
Contaminants are captured and removed close to where it s generated (before dispersed in workplace) Achieved by suction hood of some type and can be very effective Basic components An extraction hood, where the contaminant is drawn in. This can take manyforms, from a slot to a laboratory fume cupboard Ductwork to connect the system together An air cleaner (optional) if the contaminant cannot be safely discharged outside A fan A discharge point (e.g. a stack). |
|
|
LEV testing |
Smoke tests. Smoke can be released into the airflow to ensure that it iscaptured. Hand held smoke tubes are readily available. Tyndall beam lamps can be used to visualise whether particulates are beingcaptured (see: MDHS 82). Face velocities. The speed at which air is drawn into an exhaust hood can bemeasured using an anemometer. Needs test every 14 months by legislation |
|
|
PPE |
Bottom of list Be uncomfortable to wear; Make working difficult; Create a secondary risk not identified in the original assessment. Forexample, some items of personal protective equipment can hinder the body’snatural cooling mechanisms by preventing evaporation of perspiration; and It can also be difficult to monitor the level of protection provided by thepersonal protective equipment. |
|
|
RPE |
Useful short term solution Air-purifying: A respirator with an air-purifying filter, cartridge, or canister thatremoves specific air contaminants by passing ambient air through the air-purifyingelement. or Supplied Air: A respirator that supplies the user with breathing air from a source independent of the ambient atmosphere. |

